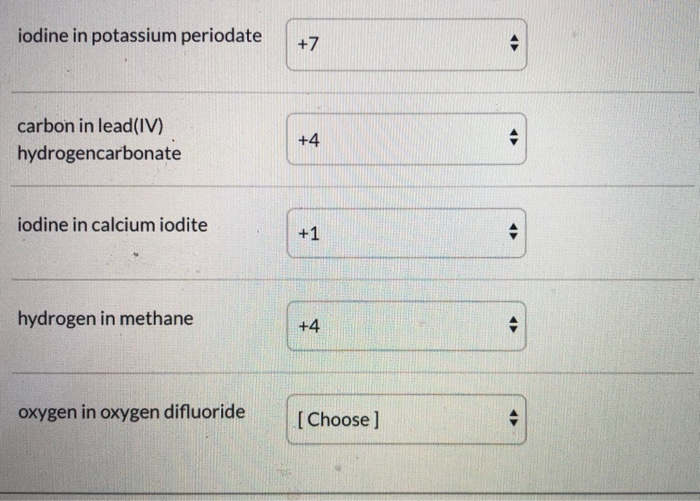
What Is The Oxidation Number Of Hydrogen In Kh

Okay, so picture this: I’m staring at a chemistry textbook, right? It’s late, I’ve had way too much coffee, and my brain is basically mush. And there it is, staring back at me, this simple question: “What is the oxidation number of hydrogen in KH?”
My first thought, naturally, was: “Hydrogen? Usually it’s +1, right? Like in H₂O, where oxygen is the boss and gives hydrogen a little pat on the back, saying ‘Here’s an electron, kid.’” But then I saw the KH, and my internal chemistry alarm bells started dinging. KH. That’s Potassium Hydride. And suddenly, the simple answer felt… wrong. Like wearing socks with sandals – just not the vibe.
This is the kind of thing that keeps me up at night (okay, maybe not literally up at night, but you get the drift). Those seemingly small details that can totally flip the script on what you thought you knew. So, let’s dive into this KH mystery, shall we? Because understanding the oxidation number of hydrogen in this particular compound is actually pretty darn fascinating, and it teaches us a valuable lesson about not jumping to conclusions.
The Usual Suspects: Hydrogen's Typical Oxidation Numbers
Before we get too deep into the KH drama, let's refresh our memories about hydrogen's usual gigs in the chemical world. Most of the time, our friend Hydrogen likes to be the giver, the benevolent electron donor. In compounds with more electronegative elements (think oxygen, halogens like chlorine or fluorine), hydrogen happily hands over its single electron. This gives it an oxidation number of +1.
Water (H₂O) is the classic example, as I mentioned. Oxygen is way more electronegative, so it pulls electrons towards itself. Hydrogen ends up with a partial positive charge, hence, +1. Hydrochloric acid (HCl) is another good one. Chlorine is a greedy electron-grabber, leaving hydrogen with its +1 status.
Even in things like ammonia (NH₃), nitrogen is more electronegative, so hydrogen is rocking the +1 oxidation number. It’s like hydrogen’s default setting, its comfort zone. It’s the friendly neighbor who always lends a hand (or an electron).
But here’s the kicker: chemistry, much like life, is full of exceptions. And the exceptions are often the most interesting parts, right? They’re the plot twists that keep you engaged. So, what happens when hydrogen encounters an element that’s even less electronegative than itself?
Enter the Metal Mavericks: Alkali Metals and Their Influence
This is where Potassium (K) struts onto the scene. Potassium is an alkali metal. If you’ve ever messed with alkali metals, you know they are super reactive. They’re like the rock stars of the periodic table – they love to lose an electron and become positively charged ions.
In fact, alkali metals are among the least electronegative elements out there. They have a very strong tendency to give away their electron. So, when hydrogen comes knocking on the door of potassium, who’s the electron-giver and who’s the electron-receiver?
Think about it: Potassium is practically begging to lose an electron to achieve a stable electron configuration. Hydrogen, while often a +1, can sometimes play a different role. In the presence of a metal that’s even more eager to shed an electron than hydrogen typically is to donate, the roles can get… swapped.
So, in KH, potassium is the dominant force. It’s the alpha dog of this particular chemical partnership. It’s going to grab that electron from hydrogen.
The Shocking Revelation: Hydrogen as the Hydride Ion
And this, my friends, is where we get to the heart of the matter. In potassium hydride (KH), potassium has an oxidation number of +1. It loses its electron to become K⁺. Now, what does hydrogen do?
Instead of giving away its electron and becoming H⁺, hydrogen accepts an electron from potassium. This means hydrogen gains an electron. When an atom gains electrons, its oxidation number decreases. Since hydrogen started with zero charge and gained one electron, it now has a charge of -1.
So, the oxidation number of hydrogen in KH is -1. This form of hydrogen is called the hydride ion (H⁻).

Mind. Blown. Right? It’s like finding out your quiet neighbor is actually a secret agent. It completely changes your perception of them.
This is a crucial concept: oxidation numbers aren't fixed properties of an element. They depend entirely on the environment – the other elements it’s bonded to and how electronegative they are. It’s all about the tug-of-war for electrons.
Why Does This Matter? The Big Picture
Okay, so we’ve established that hydrogen can be -1. Why should you care? Well, understanding oxidation numbers is fundamental to understanding chemical reactions. It helps us:
- Balance chemical equations: Knowing who’s gaining and losing electrons is key to making sure your equations are balanced.
- Predict reaction types: Is it a redox reaction (reduction-oxidation)? Oxidation numbers are your cheat sheet for that.
- Understand bonding: It gives us clues about how atoms are interacting and sharing electrons.
- Name compounds: Oxidation states are sometimes incorporated into systematic naming conventions.
In the case of KH, its reactivity is a direct consequence of this hydride character. Hydride ions are strong reducing agents. They’re eager to donate that extra electron they’ve acquired, making them useful in certain chemical syntheses.
So, while H₂O has hydrogen with a +1 oxidation state, and KH has hydrogen with a -1 oxidation state, both are perfectly valid and tell us different stories about how these atoms are behaving.
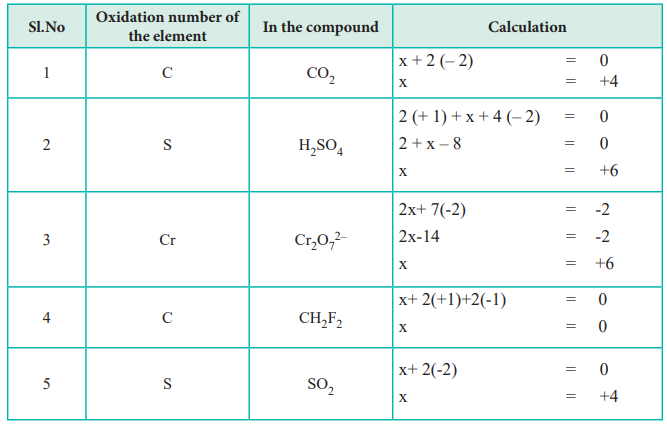
More Than Just Numbers: The Nuances of Electronegativity
Let’s talk a bit more about this electronegativity business. It’s the really important factor here. Electronegativity is a measure of how strongly an atom attracts electrons in a chemical bond. Fluorine is the queen of electronegativity, followed by oxygen, chlorine, and so on.
The general trend is that electronegativity increases as you move up and to the right on the periodic table. Metals, especially alkali and alkaline earth metals, are on the lower left, meaning they have low electronegativity. Nonmetals, especially halogens and oxygen, are on the upper right, with high electronegativity.
When hydrogen is bonded to an element that is more electronegative than it (which is most of the time!), hydrogen gets a positive oxidation number. When it’s bonded to an element that is less electronegative than it (like potassium or sodium), hydrogen gets a negative oxidation number.
It’s like a dominance hierarchy. If the element you're bonded to is stronger (more electronegative), it takes electrons and hydrogen ends up positive. If the element you're bonded to is weaker (less electronegative), hydrogen can actually take electrons and end up negative. Wild, huh?
The Exception to the Exception: Metal Hydrides
So, we’ve established that KH is an example of an ionic hydride, where we have distinct K⁺ and H⁻ ions. These are typically formed between alkali metals or alkaline earth metals and hydrogen. You’ll see similar behavior in compounds like NaH (sodium hydride) and CaH₂ (calcium hydride).
It’s important to distinguish these from covalent hydrides, like methane (CH₄) or ammonia (NH₃). In CH₄, carbon is more electronegative than hydrogen, so hydrogen has an oxidation number of +1. In NH₃, nitrogen is more electronegative than hydrogen, so hydrogen is again +1.

Then there are interstitial hydrides, often formed with transition metals. The hydrogen atoms are sort of “stuck” in the gaps (interstices) of the metal lattice. The oxidation states in these can be a bit more complex and less clearly defined, sometimes fractional, but the -1 state is generally not dominant here.
But for KH, it’s definitely in the ionic hydride camp, and that -1 oxidation number for hydrogen is the star of the show.
A Word of Caution (and Encouragement!)
The big takeaway here, for me, is that you can’t just memorize a single oxidation number for an element and expect it to apply everywhere. Chemistry is nuanced! It’s about context. It’s about understanding the forces at play.
So, next time you see hydrogen, don’t automatically assume it’s +1. Take a second. Look at what it’s bonded to. Is it a nonmetal that loves to grab electrons? Then probably +1. Is it a super-reactive alkali metal that practically gives electrons away? Then… maybe -1?
This kind of thinking is what makes chemistry so rewarding. It’s a puzzle, and the more pieces you understand, the clearer the whole picture becomes. It’s not just about rote memorization; it’s about understanding the why.
So, yeah, the oxidation number of hydrogen in KH is -1. It’s a brilliant example of how even the most common elements can surprise us. And that, my friends, is why I love chemistry. It’s always teaching us something new, even when we think we’ve got it all figured out.
