What Is The Name Of The Molecular Compound Cbr4

Hey there, science explorer! So, you've stumbled upon this mysterious little chemical formula: CBr4. Looks kinda like a secret code, right? Like, what do you even call this thing? Is it like a superhero name? "It's... Carbon Tetrabromide, fighting crime one molecular bond at a time!" Or maybe it's a fancy dish? "Tonight, I'm serving Carbon Tetrabromide with a side of… well, let's not go there just yet. Stick with me, and we'll crack this code together. No lab coat required, promise!
First off, let's break down the players in this molecular drama. We've got 'C' and 'Br'. You probably already know 'C' is for our good old friend, carbon. This is the element that forms the backbone of so many cool things, from your smartphone to that delicious chocolate bar you might be eyeing. Carbon is like the ultimate builder, always ready to link up with other atoms.
Then we have 'Br'. Now, this little guy stands for bromine. Think of bromine as a bit more… dramatic. It's a halogen, and halogens are known for being quite reactive. They like to grab onto electrons. So, bromine atoms are always looking for a partner, and they're not shy about it!
Now, let's look at the numbers. That little '4' next to the 'Br' is super important. It tells us we have four bromine atoms for every one carbon atom. Imagine a carbon atom sitting in the middle, looking a little like a nervous host at a party, and then four bromine atoms are enthusiastically surrounding it, all eager to hold its hand. It's a pretty cozy arrangement, wouldn't you say?
So, when we put it all together, CBr4 tells us we have one carbon atom bonded to four bromine atoms. And the name? Drumroll, please! The official, totally-not-made-up-by-a-mad-scientist name for CBr4 is carbon tetrabromide. See? Not so scary after all! It's like giving a nickname to your pet rock. "Oh, that's not just a rock, that's Reginald!"
Let's dissect that name a bit. "Carbon" is straightforward – that's our central atom. Then we have "tetra". Ever heard of a tetrahedron? Or maybe a quartet of singers? That "tetra" prefix always means four. So, it's like saying "carbon with four..."
And then, "bromide". This is where we acknowledge our bromine buddies. When bromine atoms are part of a compound like this, their name changes slightly. It's like when your friend goes on vacation and you refer to them by their vacation nickname, "Hey, where's 'Sunny'?" Bromine becomes "bromide" when it's joined up with another element. So, carbon tetrabromide literally means "carbon with four bromides." Pretty logical, if you ask me!
It’s kind of like building with LEGOs. You have your big carbon brick, and you snap on four smaller bromine bricks. And the name just describes exactly what you've built! Easy peasy, lemon squeezy, right?
Now, you might be wondering, "Okay, okay, I know its name. But what does this CBr4 thing actually do?" Well, that's where things get a little more interesting. Carbon tetrabromide isn't exactly something you'd find in your kitchen pantry, unless you're hosting a very, very avant-garde cooking show. It's more of an industrial chemical.

Historically, carbon tetrabromide has had a few different jobs. In the past, it was actually used in things like fire extinguishers. Imagine that! This seemingly simple molecule helping to put out fires. It's like the quiet hero who doesn't seek the spotlight but is always there when you need it.
It was also used as a refrigerant. Think of it as a tiny, invisible ice pack. However, like many older refrigerants, its use has declined due to environmental concerns. It’s a good reminder that even the simplest compounds can have complex impacts on our planet.
These days, you're more likely to find carbon tetrabromide being used in organic synthesis. This is basically the art and science of creating new carbon-based molecules. Chemists use it as a reagent, which is like a special ingredient that helps to create other, more complex chemicals. It's like being a master chef, and carbon tetrabromide is one of your secret spices!
It can also be used as a solvent in certain chemical reactions. A solvent is something that dissolves other substances. Think of it like water dissolving sugar. Carbon tetrabromide can dissolve certain things that water or other common solvents can't, making it useful for specific chemical processes. It’s the go-to for those stubborn solutes!

Now, a little word of caution. While the name is fun, and its uses are interesting, carbon tetrabromide isn't something to play with casually. It can be a bit of a grumpy molecule if not handled properly. It's not toxic in the same way a snakebite is, but it can be irritating to skin and eyes, and it's definitely not something you want to breathe in regularly.
Think of it like a powerful tool. A hammer can build amazing things, but if you're not careful, you can also whack your thumb. So, when chemists work with carbon tetrabromide, they do it with the respect and safety precautions it deserves. It's all about knowing your ingredients and how to use them wisely.
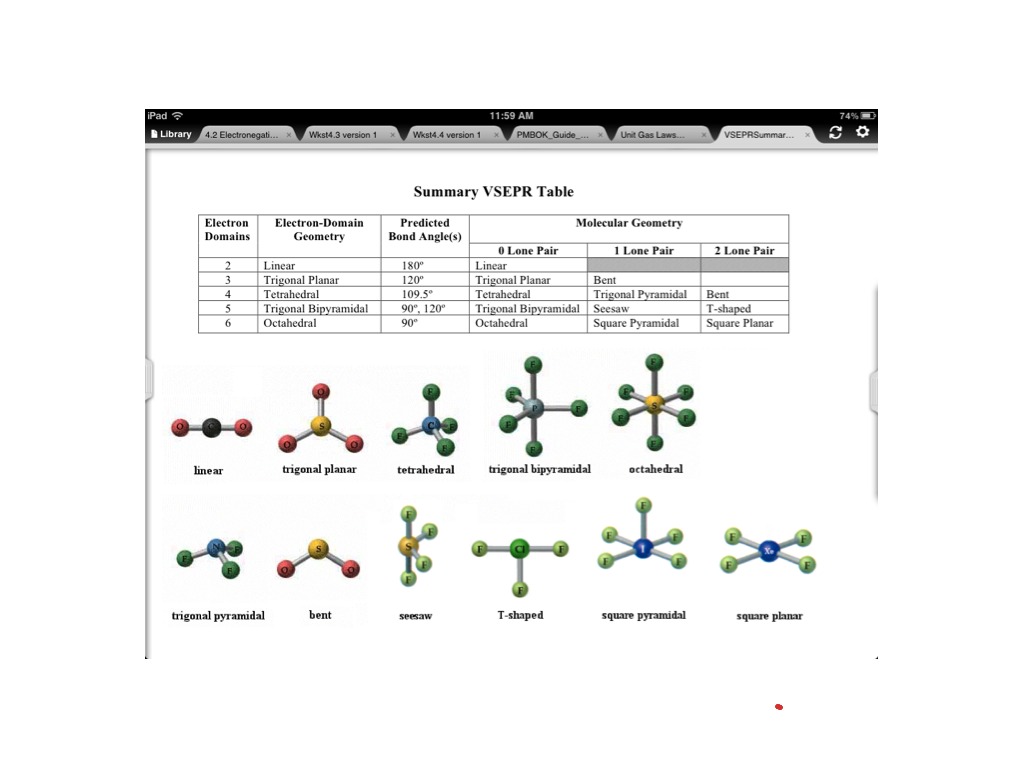
The structure of carbon tetrabromide is actually quite neat. The carbon atom sits at the center, and the four bromine atoms are arranged around it in a tetrahedral shape. Imagine a pyramid with a flat base and three triangular sides meeting at a point. Now, flip it upside down, and the carbon atom is at the bottom, with the bromine atoms forming the three points of the base and the single top point. Or, even simpler, picture a tripod with a ball balanced on top. The carbon is the ball, and the bromine atoms are the legs of the tripod, spread out evenly.

This tetrahedral arrangement is very common in molecules. It's a way for atoms to get as far away from each other as possible, minimizing those pesky electron repulsions. Nature likes things to be neat and tidy, and this shape is a perfect example of that.
So, there you have it! CBr4 is carbon tetrabromide. It’s a molecule that might sound a bit intimidating at first, but it’s just a carbon atom hanging out with four bromine atoms in a rather symmetrical fashion. It’s played a role in fighting fires, keeping things cool, and is now helping chemists create new wonders.
Isn't it amazing how a few letters and numbers can represent something so specific, something with its own history and purpose? It just goes to show that the world of chemistry is full of fascinating characters, each with their own unique story. From the simplest atom to the most complex compound, there’s a whole universe of discovery waiting for you.
And the best part? You don't need a fancy degree to start appreciating it. Just a little curiosity, a willingness to explore, and maybe a willingness to remember that "tetra" means four! Keep asking questions, keep exploring, and who knows what other amazing molecular compounds you'll discover. The universe is a giant chemistry lab, and you're invited to be a part of the experiment. So go forth, be curious, and always keep that smile of discovery on your face!
