What Is The Name Of An Alcohol Derived From Hexane
Ever found yourself staring at a chemistry textbook, or perhaps just pondering the mysteries of the universe while sipping a beverage, and wondered about the names of things? It’s a bit like a linguistic treasure hunt, isn't it? We often encounter words and concepts that sound intriguing, like they belong to a secret society of scientists. Today, we're going on an adventure into the world of organic chemistry to uncover the name of a specific type of alcohol. This isn't just about memorizing terms; it’s about understanding how the building blocks of nature connect to create the world around us, and sometimes, even the flavors we enjoy.
The reason this particular question is fun is because it highlights a fundamental principle in chemistry: how we name compounds tells us a lot about their structure. It’s like a chemical ID card! And knowing this helps us understand the properties and potential uses of different substances. Plus, let's be honest, sounding a little knowledgeable about the molecular world can be a great party trick. So, let's dive in and demystify the name of an alcohol derived from a very common molecule.
The Building Blocks of Alcohol
Before we get to the grand reveal, let’s lay some groundwork. Alcohols are a class of organic compounds that are characterized by the presence of a hydroxyl group (OH). This hydroxyl group is bonded to a carbon atom, which is itself part of a larger carbon-based chain or ring. Think of it like adding a special tag to a basic carbon skeleton. The type of carbon skeleton determines the fundamental name of the alcohol.
Now, you might have heard of familiar alcohols like ethanol, which is the alcohol in alcoholic beverages (think wine, beer, and spirits). Ethanol has two carbon atoms in its backbone. But what if we have a longer carbon chain? This is where our investigation into hexane comes in.
Hexane: The Six-Carbon Skeleton
Hexane itself is a hydrocarbon, meaning it's composed solely of hydrogen and carbon atoms. It's part of a family called alkanes, which are saturated hydrocarbons. The "hex" prefix in hexane tells us something very important: it has a chain of six carbon atoms. A straight-chain hexane looks like this: C-C-C-C-C-C. You can also have branched forms of hexane, but for the basic naming of alcohols derived from it, we’re thinking about that six-carbon backbone.

Hexane is commonly found as a component of gasoline and is often used as a solvent in laboratories and industries due to its ability to dissolve greases, oils, and other nonpolar substances. It’s a workhorse molecule in many practical applications.
Adding the Hydroxyl Group: The Birth of an Alcohol
So, we have our six-carbon chain, hexane. To turn it into an alcohol, we need to attach a hydroxyl group (-OH) to one of those carbon atoms. When we do this, the name of the compound changes to reflect this new functional group.
The naming convention in organic chemistry is quite systematic. The suffix of the parent alkane name is changed to "-ol" to indicate that it's an alcohol. So, if our parent chain is hexane, and we add an -OH group, we’re looking at something related to "hexanol".
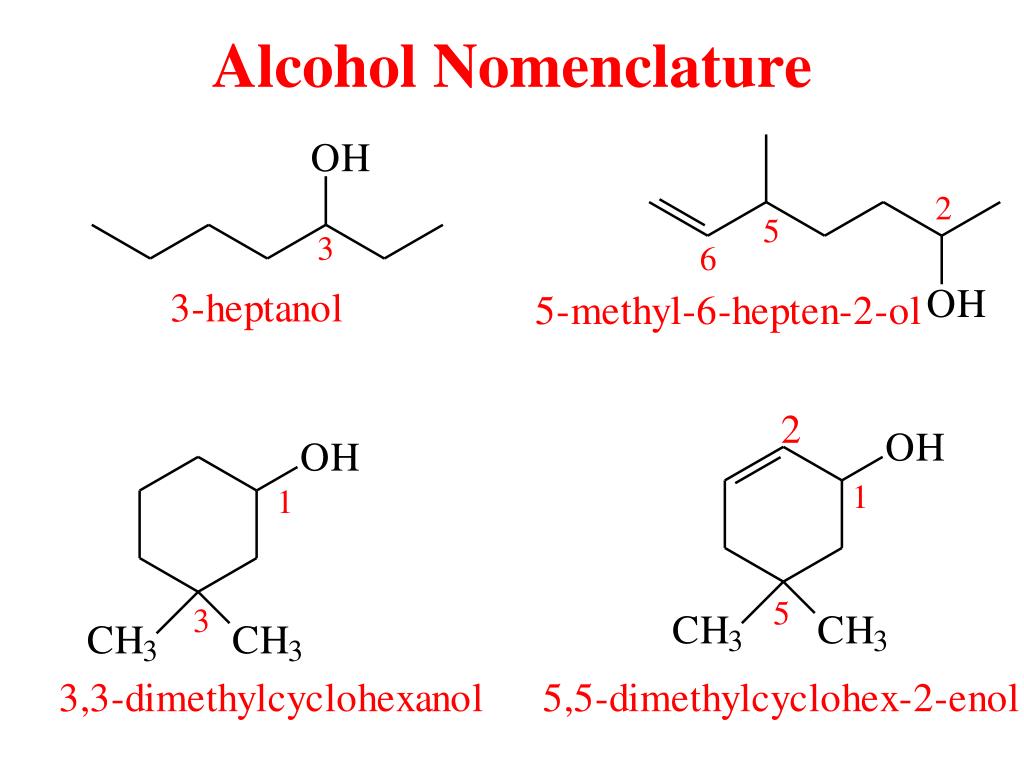
The name of an alcohol derived from hexane is simply hexanol.
"The name 'hexanol' directly tells us that the molecule has a six-carbon backbone and an alcohol functional group."
It's as straightforward as that! The "hex-" prefix signifies the six-carbon chain, and the "-ol" suffix signifies the presence of the hydroxyl group, making it an alcohol.
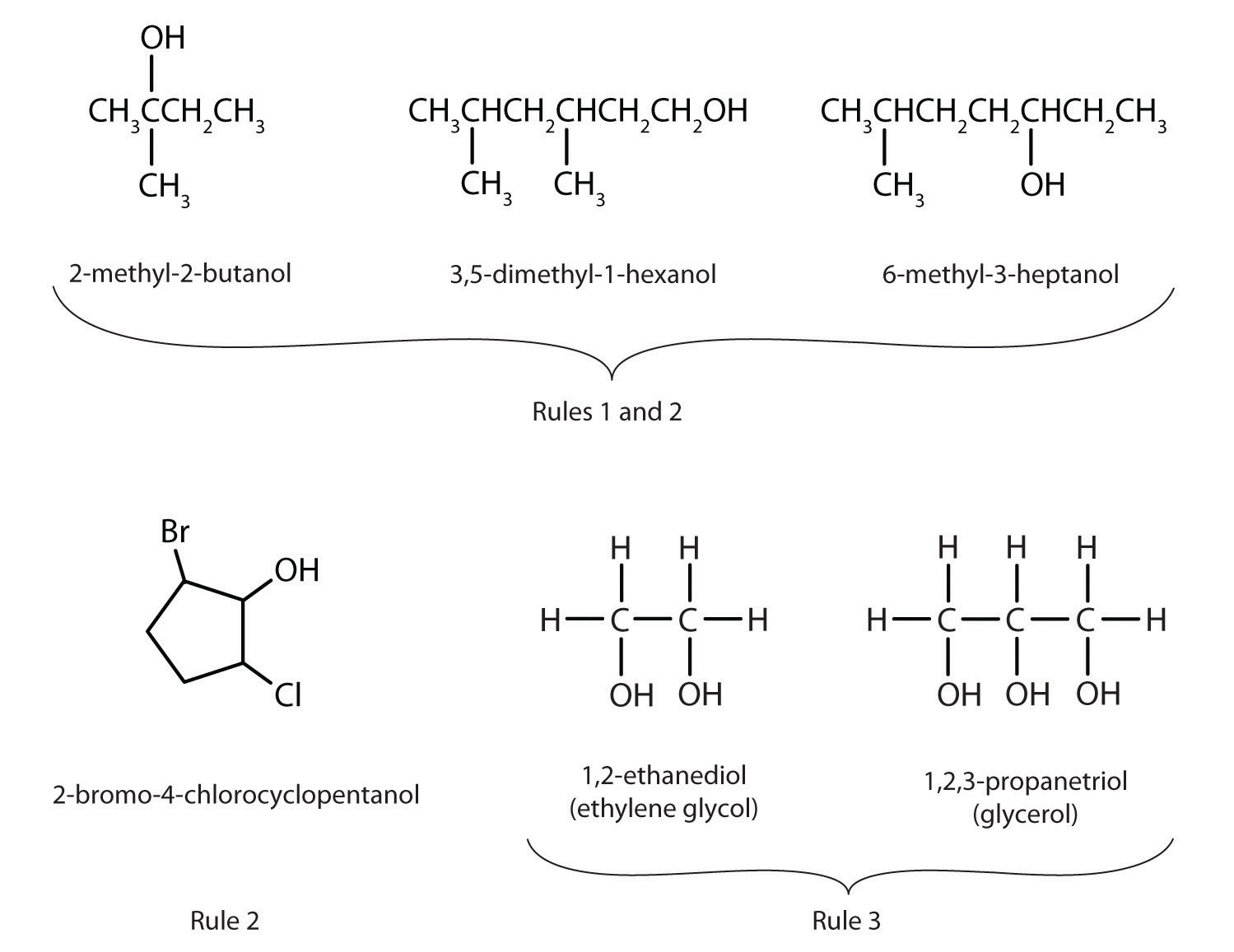
Variations and Positions: It Matters Where You Put It!
Now, here's where it gets a little more interesting and scientifically precise. A hexane molecule has six carbon atoms. The hydroxyl group can attach to different carbon atoms along this chain. This leads to different structural isomers, and their specific names reflect the position of the hydroxyl group.
For example, if the hydroxyl group is attached to the very first carbon atom in a straight chain of six carbons, it's called 1-hexanol. If it's attached to the second carbon atom, it's called 2-hexanol. If it's attached to the third carbon atom, it's called 3-hexanol. The numbers indicate the position of the -OH group on the carbon chain, starting from one end.
These positional isomers, while all being hexanols, can have slightly different properties. For instance, their boiling points, solubilities, and reactivity can vary. This is a fundamental concept in organic chemistry: the arrangement of atoms matters!

The Purpose and Benefits of Hexanol
So, why do we care about hexanol? These six-carbon alcohols are not just theoretical concepts; they have practical applications. Hexanols are often used as:
- Solvents: Like other alcohols, they can dissolve a variety of substances. Their specific solubility properties depend on the position of the hydroxyl group.
- Intermediates in Chemical Synthesis: They are building blocks for creating other, more complex chemicals. This could include fragrances, plasticizers (substances added to plastics to increase their flexibility), and pharmaceuticals.
- Components in Lubricants and Hydraulic Fluids: Their properties make them useful in certain industrial applications.
- Fragrance and Flavor Compounds: Some hexanol isomers have distinct odors and tastes, and they are used in the food and cosmetic industries. For example, cis-3-hexenol has a strong, grassy, leafy aroma and is a key component in the scent of freshly cut grass.
The benefits of understanding hexanol, and indeed alcohols in general, lie in our ability to predict and harness their properties. When chemists see the name "hexanol," they immediately know it's a molecule with a six-carbon backbone and an -OH group. This knowledge allows them to design experiments, create new materials, and understand existing ones.
It’s a testament to the elegance of chemical nomenclature that a simple name can unlock so much information. So, the next time you hear the word "hexanol," you'll know you're talking about a versatile six-carbon alcohol, a useful molecule with a name that perfectly describes its chemical identity. It’s a small piece of the vast and fascinating puzzle that is chemistry, and a reminder that even everyday substances have intriguing scientific stories to tell.
