What Is The Most Stable Monatomic Ion Formed From Aluminum

You know, I remember this one time, way back when I was knee-deep in chemistry homework that felt like trying to decipher ancient hieroglyphs. My roommate, bless his oblivious soul, was trying to make a peanut butter and jelly sandwich. He’d meticulously spread the peanut butter, then confidently… well, he’d kind of just plopped the entire jar of jelly right in the middle. It was a glorious, sticky mess. He looked at me with this sheepish grin and said, "I guess it didn't really… spread."
It sounds silly, right? A sandwich analogy for something as serious as atomic stability. But bear with me, because in a weird, roundabout way, it actually hit on something fundamental. We’re talking about how atoms like to arrange themselves, how they achieve a state of… well, let's call it 'sandwich perfection' for the elements.
And when we talk about aluminum, this is where things get really interesting. Aluminum, that shiny, lightweight metal we see everywhere from soda cans to airplane parts, has a story to tell when it comes to forming ions. And today, we’re going to unravel the mystery of its most stable, most perfectly “spread” ionic form.
The Quest for Stability: Atoms Aren't Exactly Fans of Being Unbalanced
So, what makes an atom "stable"? Think of it like a really comfortable armchair. Atoms, much like us after a long day, crave comfort. This comfort, in chemical terms, comes from having a full outer electron shell. It's like having all your essential snacks within reach – no need to go searching for more. The most stable electron configuration for most atoms is one that mimics the noble gases, those aloof, incredibly unreactive elements like Neon or Argon. They've got their outer shells jam-packed.
Atoms achieve this fullness in a couple of ways. They can either gain electrons to fill their outer shell, or they can lose electrons to expose a pre-existing, full inner shell. This is where the whole "ion" thing comes in. When an atom gains or loses electrons, it develops an electrical charge, becoming an ion. And these charged little guys are the stars of our story today.
It's like a perpetual game of electron hopscotch, and every atom is trying to land on a perfect square. Some are good at snatching up electrons, others are eager to shed them. And then there’s aluminum, with its own unique strategy.
Aluminum's Electron Circus: A Tale of Three Potential Performers
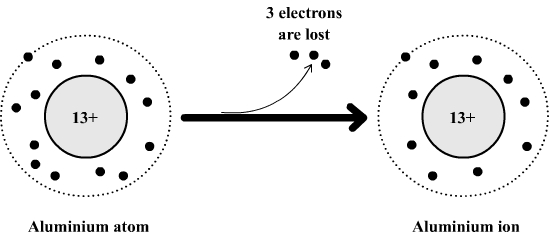
Let's zoom in on our friend, aluminum. The symbol for aluminum is Al, and its atomic number is 13. This means it has 13 protons in its nucleus and, in its neutral state, 13 electrons buzzing around. Now, the electron configuration of aluminum is 2, 8, 3. This tells us it has two electrons in its first shell, eight in its second, and importantly, three in its outermost shell, the valence shell.

So, aluminum is faced with a choice. It can try to gain five more electrons to fill that third shell and get a nice, stable 8 in there. Five electrons! That's like trying to collect five extra sprinkles to finish off your ice cream cone. It’s a lot of effort, and honestly, it’s not its natural inclination.
Alternatively, and this is where things get interesting, it can lose those three valence electrons. If aluminum loses these three electrons, what’s left? The second electron shell, which already has a full eight electrons! Voila! It achieves that coveted noble gas configuration.
This is the crucial point, the "aha!" moment, if you will. Losing three electrons is a much more energetically favorable process for aluminum than gaining five. Think about it: which is easier, carrying three light shopping bags, or lugging five incredibly heavy ones? For aluminum, shedding those three is the clear winner.
When aluminum loses those three electrons, it becomes an ion with a charge of +3. Why +3? Because it started with 13 protons (positive charges) and 13 electrons (negative charges), a neutral balance. When it loses 3 negative charges (electrons), the positive charges (protons) now outnumber the negative charges by three. So, we get the aluminum ion, denoted as Al3+.
This Al3+ ion is the most stable monatomic ion formed from aluminum. It’s the perfectly balanced peanut butter and jelly sandwich. It’s the atom that has achieved its ultimate state of electrochemical bliss.

The "Why" Behind the +3 Charge: It's All About the Octet Rule (Mostly)
Now, you might be asking, "But why is losing electrons always better than gaining?" Well, it’s not always, but for aluminum, it absolutely is. This concept is deeply rooted in something chemists call the octet rule. As we touched upon, atoms tend to gain, lose, or share electrons to achieve a full outer shell of eight valence electrons, much like the noble gases (Helium is the exception with two).
Aluminum’s neutral electron configuration is 2, 8, 3. To reach an octet in its outermost shell by gaining electrons, it would need to acquire five more. That’s a significant energy investment. Imagine trying to convince five new friends to join your already packed party. It’s a tough sell!
On the other hand, by losing its three valence electrons, aluminum reveals its second electron shell, which already contains a stable octet of eight electrons. This is like inviting a couple of close friends over for a chill hang-out; it’s much easier and requires less effort. The energy required to remove those three valence electrons is far less than the energy required to gain five. So, the universe, in its infinite wisdom (and efficiency), favors the +3 ion.
This stability of the Al3+ ion is why aluminum compounds are so common and generally quite stable. Think about aluminum oxide (Al2O3), the main component of rust on some metals (though not aluminum itself, it's quite resistant to corrosion!). Or aluminum chloride (AlCl3), a widely used catalyst. These compounds exist because the Al3+ ion is so darn happy and content with its electron configuration.
It's like aluminum is saying, "You know what? I'm not going to struggle to get more. I'm going to let go of what's easy, and bam, I'm stable. Done deal." And that, my friends, is a pretty smart strategy.
The Journey to the Ion: It's Not Always Smooth Sailing
It’s important to remember that this transition isn't instantaneous. When aluminum metal reacts, it undergoes a process. Electrons are pulled away, and the atom transforms into an ion. This often happens when aluminum comes into contact with reactive elements, like oxygen in the air or halogens like chlorine.
While Al3+ is the most stable monatomic ion, the formation process itself involves overcoming some energy barriers. Think of it as the effort it takes to get out of bed on a Monday morning – there's a bit of a struggle, but once you're up and moving, things are much smoother. The energy to remove those electrons, called ionization energy, increases with each electron removed.
The first ionization energy for aluminum is the energy to remove the first electron. The second ionization energy is to remove the second, and the third for the third. Each subsequent ionization energy is higher because you're removing a negatively charged electron from an already positively charged ion, which makes it harder to pull away. But, as we’ve established, the energy difference between needing three and needing five is so significant that even with these increasing ionization energies, losing three is still the path of least resistance for aluminum.
This is where the ironies of chemistry sometimes come out. Aluminum is a metal, so we think of it as shiny and conductive. But when it forms that Al3+ ion, it’s not a metal anymore. It’s a charged species, ready to bond with other ions to form ionic compounds. It’s undergone a fundamental change in its character, all in pursuit of that perfect electron configuration.
Beyond the Monatomic: What Happens in Compounds?
Now, we're talking about monatomic ions, which means ions made of a single atom. But in the grand scheme of chemistry, these ions rarely exist in isolation. They’re like social butterflies, always looking to bond with oppositely charged ions to form neutral compounds. That's why you don't typically find a pure pile of Al3+ ions just floating around.

Instead, you’ll find them paired up with anions – negatively charged ions. For example, in aluminum chloride (AlCl3), the Al3+ ions are balanced by three chloride ions (Cl-). In aluminum oxide (Al2O3), two Al3+ ions are balanced by three oxide ions (O2-). The charges have to balance out in a neutral compound.
This ability to readily form stable ionic bonds with a variety of anions is a testament to the inherent stability of the Al3+ ion. It's the dependable player on the team, always ready to form a winning combination.
It’s fascinating to think that this seemingly simple metal is governed by these complex rules of electron behavior. It’s like understanding why your roommate would put the entire jelly jar on the sandwich – it's not necessarily wrong, it’s just… not the most stable configuration. And in the world of atoms, stability is king (or queen, or monarch of the noble gas family!).
So, the next time you see an aluminum can or handle an aluminum foil, remember the atomic journey. Remember the three electrons that were so willing to depart, paving the way for the most stable monatomic ion formed from aluminum: the ubiquitous and oh-so-stable Al3+. It’s a small piece of atomic elegance, quietly powering the world around us.
And that, my friends, is the simple, yet profound, truth about aluminum's ionic destiny. It's a story of shedding the unnecessary to embrace the perfect. Just like a perfectly spread peanut butter and jelly sandwich, it’s all about finding that ideal, balanced state. Now, if you’ll excuse me, all this talk of sandwiches is making me hungry. I think I'll go make one. And this time, I promise to spread the jelly properly!
