What Is The Molar Solubility In Water Of Ag2cro4

Hey there, coffee buddy! So, you wanna chat about silver chromate and its solubility in water? You know, that pretty, yellowish stuff that looks like it belongs in a fancy paint palette? Well, pull up a chair, because we're about to dive into the wonderfully weird world of chemistry, and it's not nearly as scary as it sounds, promise!
We're talking about Ag2CrO4, which is just a fancy way of saying silver chromate. Think of it like this: it's two silver ions (Ag+) doing a little dance with one chromate ion (CrO42-). It’s a bit of a power couple, really. And when you toss this couple into water, what happens? Do they just… dissolve? Like sugar? Spoiler alert: not exactly.
So, what's this "molar solubility" thing we're yammering on about? It’s basically how much of our solid little silver chromate dudes can actually sneak into the water before they throw up their little chemical hands and say, "Nope, we're full!" It’s like trying to cram too many people into a tiny elevator. Eventually, it just gets too crowded, and nobody else fits. Molar solubility tells us the maximum number of moles of a substance that can dissolve in a liter of water. Pretty neat, huh?
Now, silver chromate? It’s not exactly known for being a super-friendly traveler in water. It’s more of a… bashful guest. You know, the kind who brings a whole suitcase for an overnight stay and then barely unpacks? Yeah, that’s Ag2CrO4. It doesn't really want to hang out in water much. It prefers to stick with its own kind, forming a solid chunk. It’s a bit of a homebody, you could say.
So, when we talk about the molar solubility of Ag2CrO4 in water, we're talking about a very, very small number. Like, ridiculously small. We’re not talking cups and cups dissolving here. We’re talking pinpricks. A whisper. A fleeting thought. You’d be hard-pressed to see any real difference in your water if you dumped a bucket of it in. It’s almost like it just… waves goodbye and then immediately clumps back together. Dramatic, isn't it?
Why is it so grumpy about water, you ask? Well, it all boils down to something called the solubility product constant, or Ksp. Ooh, fancy acronym! The Ksp is like a scorekeeper for how much stuff can dissolve. A low Ksp means the substance is a real trooper at staying solid. A high Ksp? That's your soluble superstar, like salt! Our Ag2CrO4 has a super, super low Ksp. It’s practically bragging about how little it dissolves.
Let's get a little more science-y, but keep it chill, okay? When Ag2CrO4 does decide to dip its toes into water, it dissociates, meaning it breaks apart into its ions: two silver ions (Ag+) and one chromate ion (CrO42-). This is usually written as a little equation:
Ag2CrO4(s) ⇌ 2Ag+(aq) + CrO42-(aq)
See the little (s) and (aq)? That just means solid and aqueous (fancy word for dissolved in water). The double arrow means it’s an equilibrium – it’s not a one-way street. Some of it dissolves, and then some of it un-dissolves right back. It’s a constant back-and-forth, like a tennis match, but with ions.
The Ksp expression for this little shindig is:
Ksp = [Ag+]2 [CrO42-]
Notice the little '2' next to the [Ag+]? That's because for every one chromate ion, we get two silver ions. It’s like a package deal, but with unequal numbers. Chemistry, am I right?
Now, the actual value of Ksp for silver chromate at, let’s say, room temperature (around 25°C) is roughly 1.1 x 10-12. Whoa. See that exponent? Negative twelve. That means there are twelve zeros after the decimal point before you get to the '11'. That’s a number so small, it makes a microscopic dust bunny look like a giant redwood. It’s practically invisible solubility.
So, if we want to find the molar solubility, let’s call it 's', of Ag2CrO4, we can use that Ksp value. Remember our equilibrium equation?
Ag2CrO4(s) ⇌ 2Ag+(aq) + CrO42-(aq)
If 's' is the molar solubility of Ag2CrO4, that means 's' moles of Ag2CrO4 dissolve per liter of water. Following our equation, this means we get 2s moles of Ag+ ions and s moles of CrO42- ions dissolved.

So, plugging that into our Ksp expression:
Ksp = (2s)2 (s)
Which simplifies to:
Ksp = 4s3
Now, we just rearrange to solve for 's':
s3 = Ksp / 4

s = 3√ (Ksp / 4)
Let's plug in our Ksp value:
s = 3√ (1.1 x 10-12 / 4)
s = 3√ (2.75 x 10-13)
And after some fancy calculator work (or just knowing how to do cube roots of tiny numbers, which, let’s be honest, I leave to the pros), we get something like:
s ≈ 6.5 x 10-5 M
M stands for molarity, which is moles per liter. So, the molar solubility of Ag2CrO4 in water is about 6.5 x 10-5 moles per liter. That’s 0.000065 moles. Still a teensy, tiny number. We're talking about dissolving less than a millionth of a mole in a whole liter of water. Mind-bogglingly low, right?
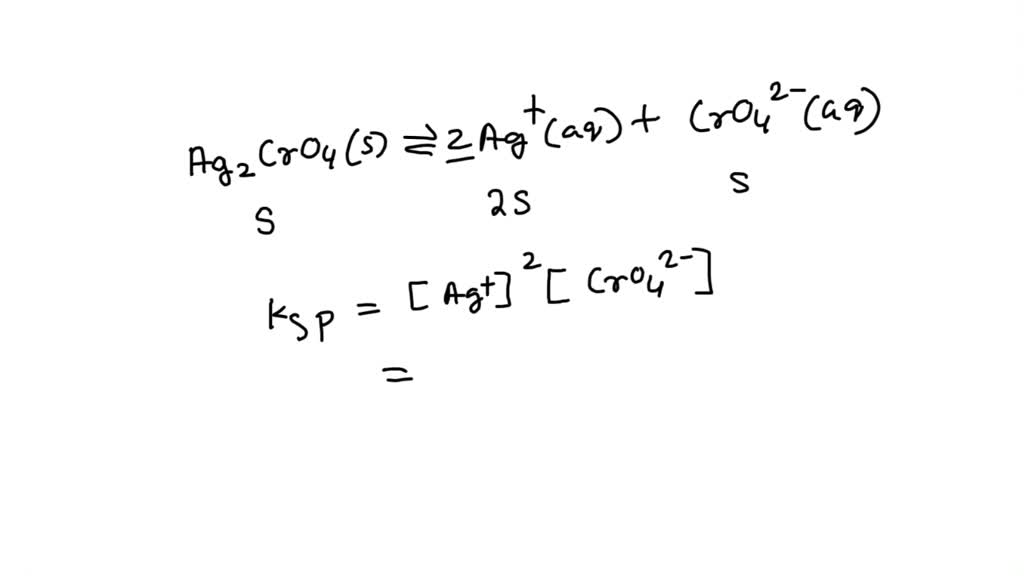
It's like trying to fill a bathtub with a single drop of water. You'd be there for a very long time, and the tub would still be mostly empty. That's our silver chromate for you. It’s just not a big fan of taking a dip in the H2O swimming pool.
Now, this value can change a tiny bit depending on the temperature, because, you know, everything in chemistry is so predictable. Higher temperatures sometimes make things dissolve a little better, but for something this insoluble, it's not going to be a dramatic jump. We're still talking "barely dissolves" territory, even if you crank up the heat.
Think about it this way: if you had a liter of water and added exactly one mole of silver chromate, almost none of it would dissolve. Most of it would just sit at the bottom, looking smug and solid. It's like bringing a bowling ball to a pool party – it just doesn't quite fit the vibe.
So, what does this mean in the real world? Well, it means you’re not going to be making any “silver chromate flavored water” anytime soon. It’s also why when you mix solutions that could form silver chromate, you often see a precipitate. That’s just fancy science talk for “it makes a solid that sinks to the bottom.” It’s like the ions are saying, “Nope, too crowded, we’re forming our own little solid club!”
It’s a really important concept in analytical chemistry, though. If you’re trying to figure out if there are silver ions or chromate ions in a solution, adding them together and seeing if you get a yellow precipitate is a dead giveaway. It’s like a chemical “aha!” moment. “Aha! There must be silver and chromate in there because we made yellow stuff!”
And you know, the color is important too! That vibrant yellow is a signature of silver chromate. It’s not just some boring white powder. It’s got personality! It's like the toddler at a party who insists on wearing the brightest, most obnoxious outfit. That yellow is its way of screaming, “Here I am! I’m silver chromate, and I’m definitely not dissolving very well!”
So, to recap our little coffee chat: the molar solubility of Ag2CrO4 in water is extremely low. We’re talking in the ballpark of 10-5 M. It’s a substance that really prefers to stay in its solid form. It’s a bit of a chemist’s puzzle, but a really cool one. It just goes to show you, even in the simplest of substances, there's a whole lot of interesting behavior happening. Makes you wonder what else is out there, right? Now, who needs a refill?
