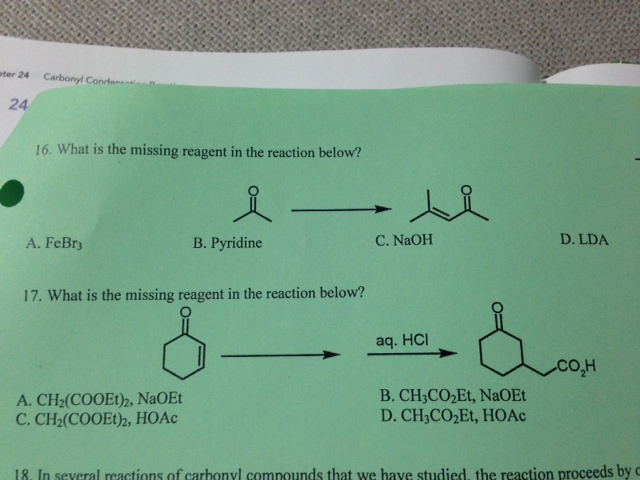
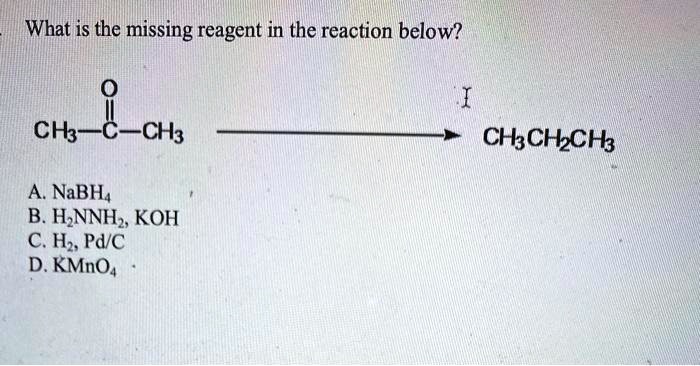
What Is The Missing Reagent In The Reaction Below
Alright, gather 'round, you magnificent bunch of brainiacs (or just people who appreciate a good mystery, which is basically everyone, right?). We’ve got a little puzzle here, a chemical whodunit that’ll make Sherlock Holmes’s deerstalker hat spin. Imagine this: we’re in our super-secret, slightly-sticky laboratory – let’s call it “The Beaker’s Brew Cafe” – and we’ve got a reaction that’s gone… well, let’s just say it’s missing its opening act. It’s like a play with no protagonist, a band with no lead singer, a taco Tuesday with no tacos! Tragic, I know.
So, what’s the deal? We’ve got this equation, looking all official and important:
2 Na + 2 H₂O → 2 NaOH + ?
See that question mark? That’s our culprit, our elusive phantom, the ingredient that’s playing hide-and-seek with our precious sodium and water. And let me tell you, when you mix sodium, that glittery, intensely reactive metal that would probably high-five lightning if it could, with plain old water, things tend to get… lively. It’s not just a gentle fizz; it’s more like a tiny, controlled explosion doing a samba. Don’t try this at home unless you have a really good insurance policy and a fire extinguisher the size of a small car. Seriously.
Now, let’s break down what we do know. On the left side of our equation, we’ve got our reactants. We’ve got sodium (Na). This stuff is so reactive, it has to be stored under oil to keep it from spontaneously combusting just by looking at the air. It’s like the James Bond of the periodic table – always ready for action, a bit dangerous, and incredibly cool (when contained, of course). Then we have water (H₂O). You know water, right? That stuff that makes up most of you, that keeps your houseplants from staging a dramatic wilting protest, that’s the same H₂O.
When these two amigos get together, they have a serious heart-to-heart. Sodium, with its surplus of electrons practically begging to be shared, sees water’s structure and thinks, “Ooh, I can make some friends here!” It’s a classic tale of attraction and electron transfer. This interaction, as I mentioned, is a bit… enthusiastic. It releases a good amount of energy, often enough to make the hydrogen gas produced by the reaction ignite with a satisfying pop.

On the right side, we have our products – the result of this energetic shindig. We’ve got sodium hydroxide (NaOH). This is also known as lye, and if you’ve ever made soap, you’ve probably met this stuff. It’s a strong base, meaning it’s the opposite of an acid. Think of acids as grumpy teenagers and bases as overly enthusiastic cheerleaders – they’re both intense, but in different ways. Sodium hydroxide is what’s left after the sodium has done its thing and the water has been… well, a bit rearranged.
We also have our mystery guest, represented by that tantalizing question mark. This isn’t just any random element; it’s a product of this reaction. It’s what emerges from the chemical chaos, a byproduct of the sodium-water dance. Think of it as the confetti that falls after a spectacular fireworks show.
So, what could this elusive attendee possibly be?
Let’s channel our inner detectives. We started with sodium (Na) and water (H₂O). Water is made of hydrogen (H) and oxygen (O). The sodium is eager to lose an electron, and the water molecule, bless its little heart, is willing to oblige. The sodium essentially kicks one of the hydrogen atoms out of the water molecule, steals its electron, and forms a happy little bond with the remaining OH group.
This leaves behind the kicked-out hydrogen atom. But hydrogen atoms don't like to hang out alone, especially when they’re in their atomic form. They’re like that one person at a party who doesn’t know anyone and is awkwardly checking their phone. So, what do they do? They buddy up! Two of these lonely hydrogen atoms find each other and form a molecule. And what do you get when two hydrogen atoms join forces?
You get hydrogen gas (H₂)! It’s a diatomic molecule, meaning it likes to exist in pairs. It’s the same stuff that made those Hindenburg blimps go up in flames, albeit in a much less dramatic fashion in our lab. It’s light, it’s flammable, and it’s the perfect candidate for our missing reagent!
So, the complete, glorious, and slightly explosive reaction looks like this:

2 Na + 2 H₂O → 2 NaOH + H₂
Bam! Mystery solved! The missing reagent is none other than hydrogen gas (H₂). Our question mark has been unmasked, revealed in all its gaseous glory.
Why is this so cool (besides the potential for tiny explosions)?
Well, first off, it shows us how elements and compounds can transform. It’s like alchemy, but, you know, real science. We’re taking simple ingredients and creating something new. Secondly, it highlights the energetic nature of some chemical reactions. This isn’t a gentle simmering; it’s a vigorous exchange that releases energy. This energy can be harnessed, though perhaps not in this particular uncontrolled fashion. Imagine the power we could have if we could just bottle that pop!

And a fun fact for your next trivia night: Sodium metal is so reactive that it can even react with alcohol! Imagine trying to have a drink with sodium – it would probably try to steal the carbon from your cocktail and leave you with plain old water. Not exactly the life of the party.
So, there you have it. Our missing reagent, the star player we were looking for, was hydrogen gas. It’s the little byproduct that makes this reaction go from a simple observation to a full-blown, energetic chemical event. It’s a reminder that even in the seemingly mundane world of chemistry, there are always exciting transformations happening, and sometimes, the most important things are hiding in plain sight, or, in this case, in the form of a gas just waiting to make its grand exit.
Next time you see a chemical equation with a question mark, remember our little café mystery. It’s a quest for knowledge, a pursuit of the missing piece, and maybe, just maybe, a good laugh at the expense of overly enthusiastic metals. Cheers to science, and cheers to never forgetting the little guys, or in this case, the little gas molecules, that make it all happen!
