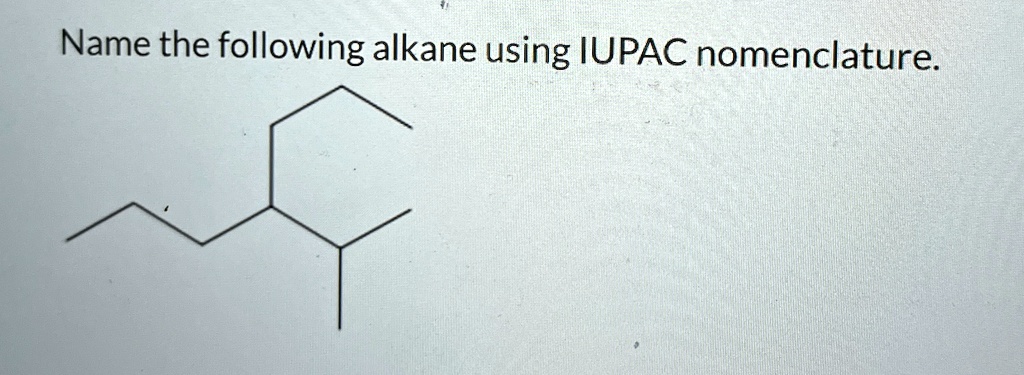
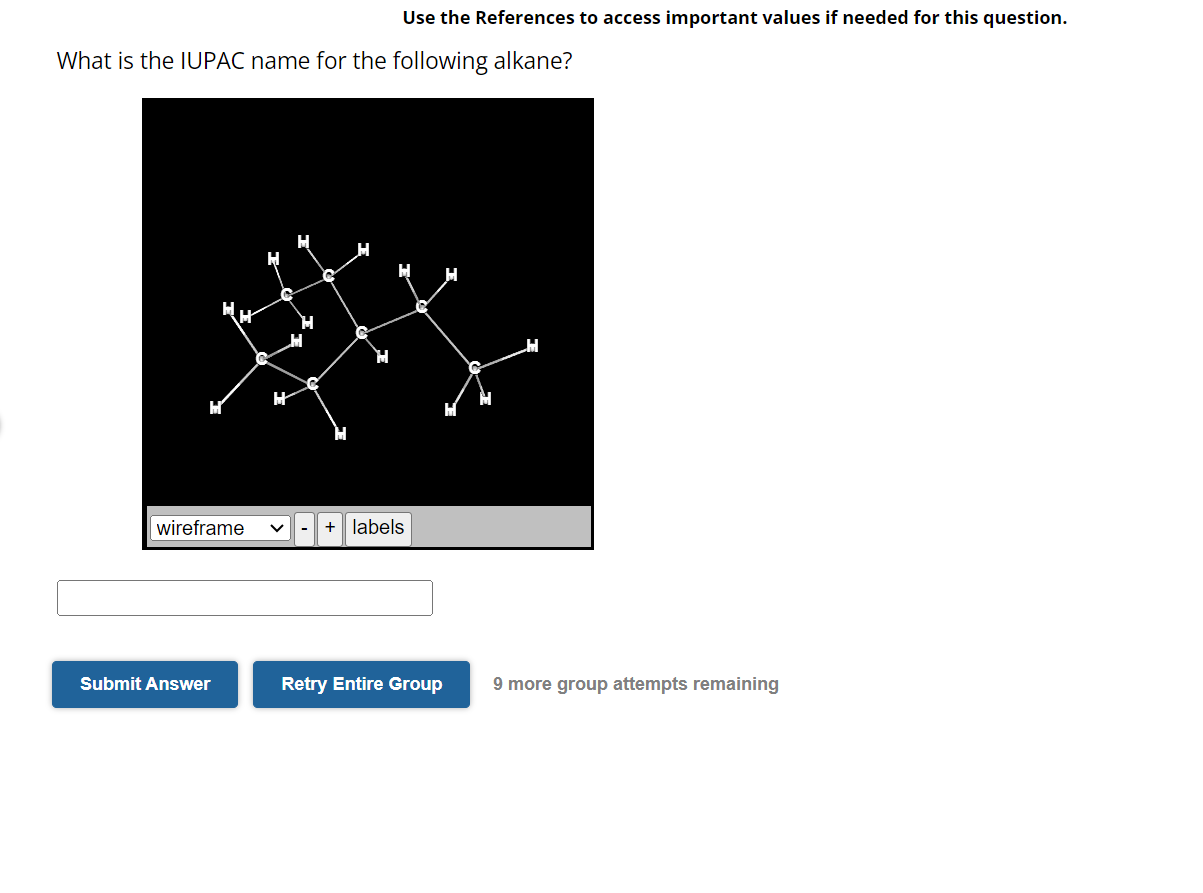
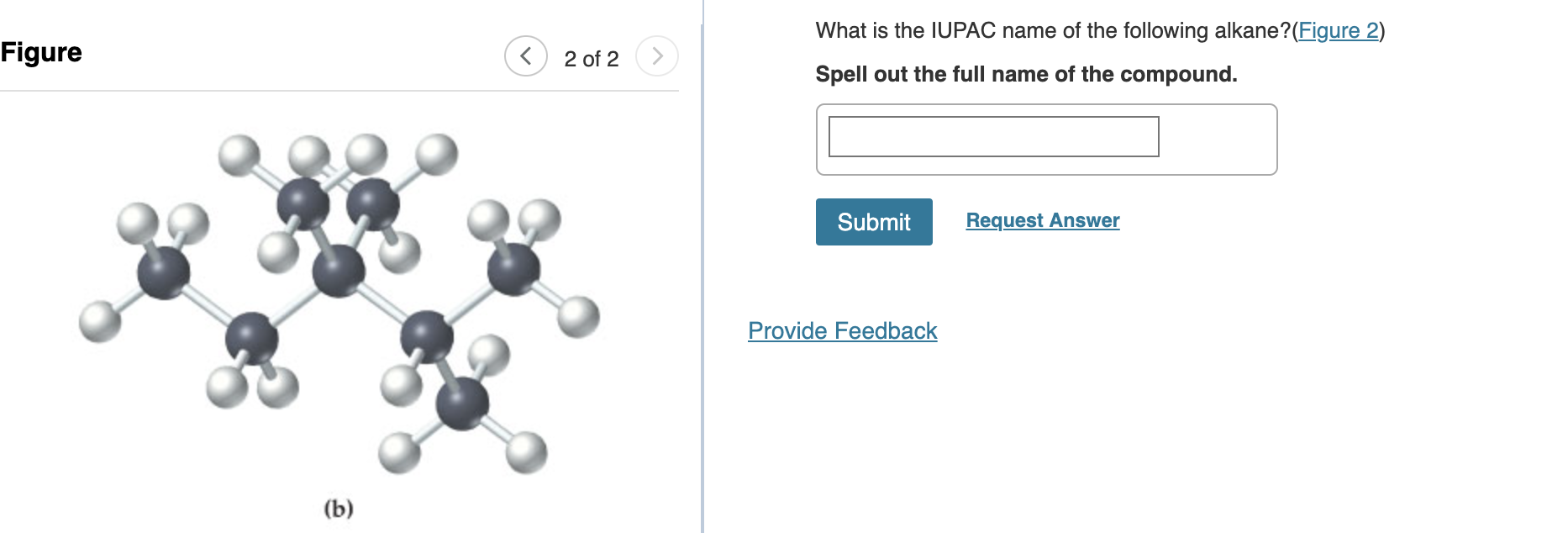
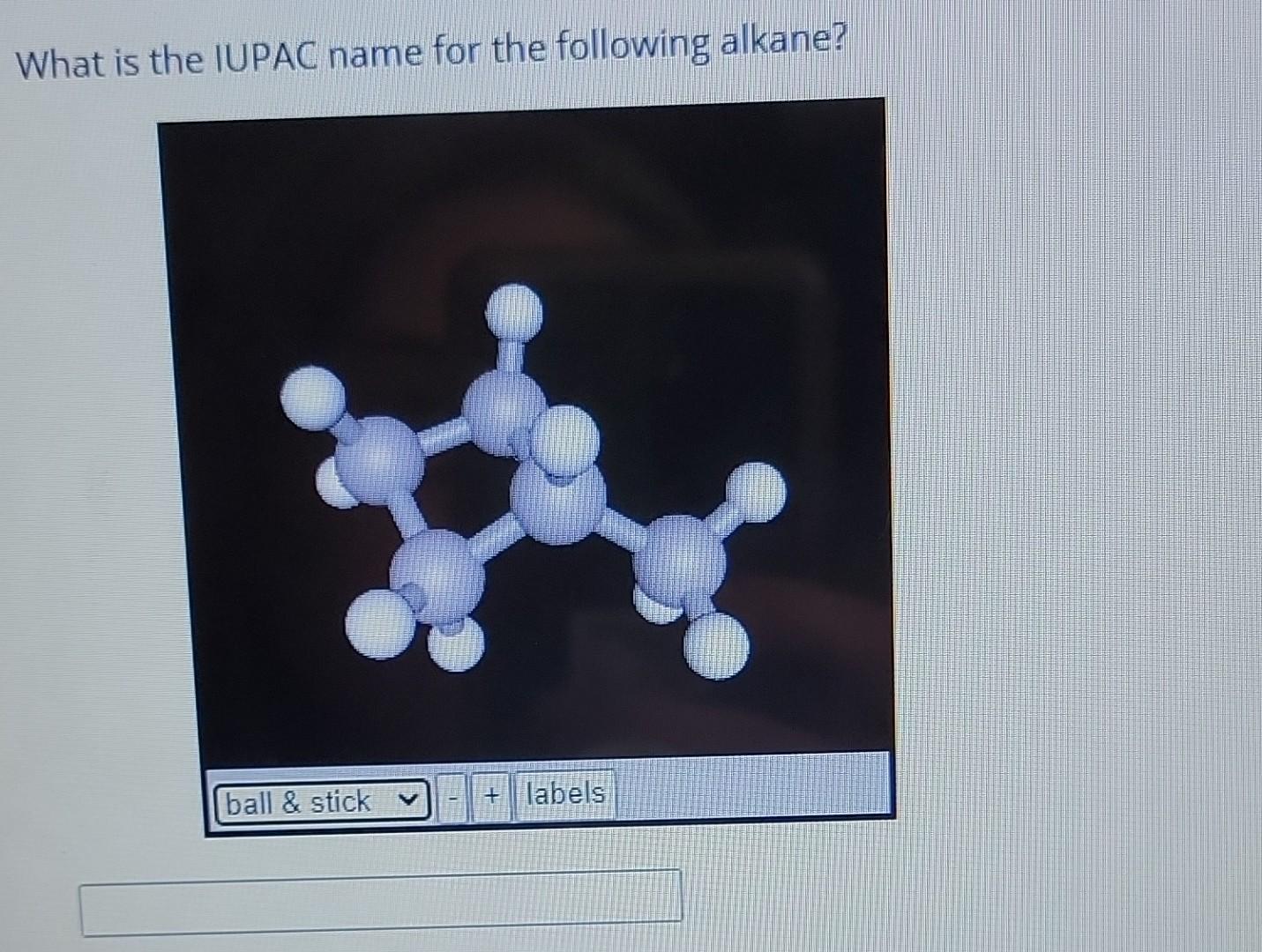
What Is The Iupac Name For The Following Alkane
Ever found yourself staring at a long, complicated string of letters and numbers in a science textbook or on a product label and thought, "What in the world is that?" Well, my friends, today we're going to demystify one of those seemingly intimidating things: the IUPAC name for a specific type of molecule called an alkane. Think of it as the super-official, no-nonsense nickname for these fundamental building blocks of so much around us.
Now, before you start picturing mad scientists in bubbling beakers, let's get real. Alkanes are actually the simplest kind of organic molecules. They're like the plain white t-shirts of the chemical world – foundational, versatile, and surprisingly important. They're made up solely of hydrogen and carbon atoms, linked together in chains. No fancy functional groups, no double bonds, just good old single bonds holding tight.
So, why should you, a person who likely spends their day navigating traffic, choosing between Netflix shows, or trying to remember where you put your keys, care about the IUPAC name of an alkane? Because these simple molecules are the backbone of fuel. Yes, that gasoline in your car? That natural gas heating your home? That propane powering your grill? They're all packed with alkanes!
Let's imagine for a second. You're getting ready for a backyard barbecue. You grab your propane tank, right? That propane is a specific alkane. If we didn't have a standardized way to name it, imagine the chaos! "Pass me that flammable gas, the one that smells a bit like burnt toast." Not exactly ideal for a smooth grilling experience. The International Union of Pure and Applied Chemistry (IUPAC) is like the ultimate naming committee for chemicals, ensuring everyone, from a seasoned chemist to a backyard griller, is talking about the same thing.
Now, let's get down to the nitty-gritty of naming. It's actually not as scary as it sounds. The IUPAC system for alkanes is wonderfully logical, almost like a set of very organized rules for building with LEGOs. You've got a prefix that tells you how many carbon atoms are in the main chain, and a suffix that tells you it's an alkane. Simple as that!
Let's start with the shortest ones. If you have just one carbon atom, it's called methane. Think of it as the single, solitary carbon atom. Methane is the primary component of natural gas, the stuff that keeps you warm in the winter and allows you to cook your dinner. Imagine a tiny, lonely carbon atom, happily doing its job, providing warmth and sustenance. It's the intro to the alkane party!
Next up, with two carbon atoms linked together, we have ethane. This is like having two LEGO bricks snapped together. Ethane is also found in natural gas and is used in the production of plastics. So, that cool toy your kid loves? It might have started its life as a couple of carbon atoms in ethane!
Then comes propane, with three carbon atoms. This is our barbecue buddy! Remember that propane tank? That's what it is. Propane is a fuel gas used for heating, cooking, and in vehicles. It’s the perfect example of a simple alkane having a very practical, everyday use. You can almost hear the sizzle of burgers just thinking about it.
Moving on to butane, with four carbon atoms. This one is often found in lighter fluid and aerosol propellants. So, the next time you flick on a lighter, you're interacting with butane! It’s the little guy that gets things started, in a literal sense.
Now, things start to get a little more interesting as the chains get longer. For five carbon atoms, it's pentane. For six, it's hexane. For seven, heptane. And for eight, octane. You're probably familiar with octane, especially if you’ve ever filled up your car. The octane rating of gasoline tells you how resistant it is to knocking or pinging, which is basically the engine complaining. Higher octane means a smoother, more powerful engine performance. So, octane is like the smooth operator of fuels!
The prefixes continue: nonane for nine, decane for ten, and so on. It’s like a systematic naming convention that scales perfectly. Think of it like building a chain of paper dolls. Each doll represents a carbon atom, and the name just tells you how many dolls are in your chain.
What about when the carbon chain isn't just a straight line? What if it branches out like a tree? That's where things get a little more involved, but the IUPAC system is designed to handle it with elegance. For instance, if you have a main chain of carbon atoms, and a shorter chain hanging off it like a little side branch, that branch gets its own name and is attached to the main chain at a specific numbered position. It's like giving your house a main address, and then having a specific apartment number if it's a multi-unit building.
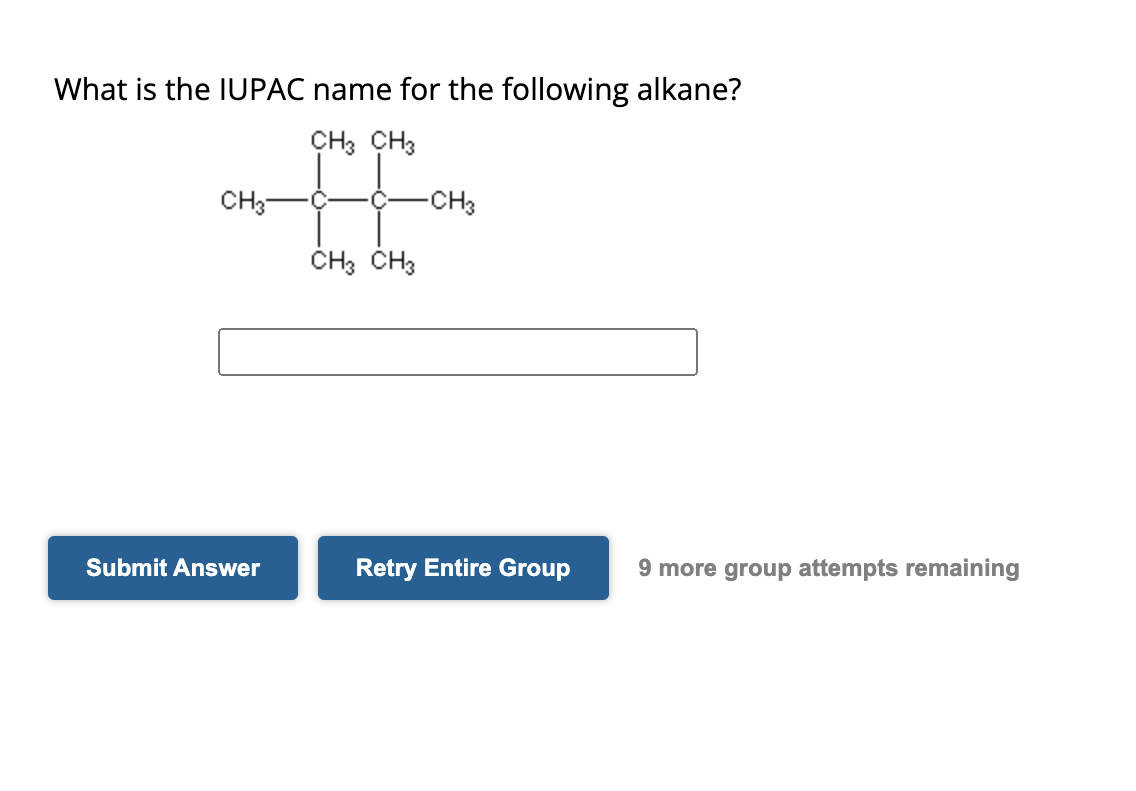
Let's take a hypothetical example. Imagine a molecule that's mostly a chain of five carbon atoms (pentane), but one of those carbon atoms has a single carbon atom branching off it. Following the IUPAC rules, we'd identify the longest continuous chain, number it, and then name the branch. This particular branched alkane, with a total of six carbon atoms but arranged with a branch, would be called 2-methylpropane (if it were a specific isomer, just for illustrative purposes – the actual naming for branched alkanes gets slightly more complex with numbering, but the principle is the same!). The "methyl" part refers to the single carbon branch, and the "2-" tells you where on the main propane chain (in this case, a propane backbone, which is three carbons long for this specific branched example, we’re getting a little ahead of ourselves, but the idea is there!) it's attached. It’s all about precision!
The beauty of the IUPAC system is that it’s universal. It doesn't matter if you're in London, Tokyo, or a small village in the Andes, when you see a specific IUPAC name, you know exactly what molecule you're dealing with. This is crucial for safety, for scientific research, and for trade. Imagine trying to order a specific chemical for a pharmaceutical company without a standardized name. It would be like trying to order a specific type of screw at a hardware store by just saying "that metal thingy."
So, the next time you see a string of letters like "hexane" or "octane," don't just gloss over it. Take a moment to appreciate the elegant simplicity and the incredible importance of these molecules. They are the silent workhorses that power our vehicles, heat our homes, and form the basis of countless products we use every day. And the IUPAC name? It's simply the key that unlocks our understanding of these fundamental building blocks of our modern world. It's the way we make sure everyone is on the same page, from the chemist in the lab to you, enjoying that perfectly grilled burger thanks to propane!
