What Is The Hybridization Of The Central Atom In No3

Hey there, science curious friends! Ever wondered about the tiny, invisible world that makes up everything around us? It's a bit like peeking behind the curtain of a magic show, and today we're going to pull back a little bit of that curtain to talk about something called "hybridization." Don't let the fancy word scare you! It's actually a super neat concept that helps us understand how atoms decide to get along and form all the stuff we see and touch. Think of it like atoms getting ready for a big party and needing to figure out the best way to mingle!
Our main star today is a little molecule called NO₃, which is the chemical formula for something called the nitrate ion. You might not know its name, but you've definitely encountered its work! Nitrates are super important in nature, playing a big role in things like plant growth – they're like the vitamins for your garden! They're also found in some of the foods we eat, like leafy greens.
So, what's up with the central atom in NO₃? Well, in NO₃, the central atom is nitrogen. Nitrogen is a pretty common element, making up a big chunk of the air we breathe. But when it teams up with oxygen atoms to form nitrate, it gets a little… complicated. Or rather, it gets clever!
Let's dive into this "hybridization" thing. Imagine you have three friends who all want to play a game of catch. Friend A is really good at throwing in one direction, Friend B in another, and Friend C in a third. Now, if they wanted to form a tight little triangle and throw a ball perfectly around in a circle, just using their original throwing styles might be a bit awkward. They'd have to twist and turn in funny ways.
Hybridization is kind of like those friends deciding to pool their talents and create a new, super-powered throwing style that's perfect for playing in that triangle. They blend their individual abilities to create something new and more efficient for the task at hand. Atoms do something similar!
Atoms have different "orbitals," which are basically like their personal space or zones where their electrons like to hang out. Think of them as different shaped rooms in a house: a round living room, a more elongated bedroom, and maybe a quirky hobby room. Before hybridization, a nitrogen atom has these distinct rooms.

Now, when nitrogen wants to bond with those three oxygen atoms in NO₃, it needs to make some space and form strong connections. It can't just stick those oxygens anywhere; they need to be arranged just right. So, the nitrogen atom decides to get a bit creative. It takes some of its electrons from those original "rooms" (orbitals) and mixes them up.
This mixing process is hybridization. For nitrogen in NO₃, it mixes one of its 's' orbitals (that's the round one) with two of its 'p' orbitals (those are the dumbbell-shaped ones). It's like taking some furniture from the living room and the bedroom and combining it to create a new, perfectly shaped playroom!
What comes out of this mixing party are three new, identical orbitals. These new orbitals are called sp² hybrid orbitals. They're like super-versatile tools that are perfect for forming those bonds with the oxygen atoms. They are also arranged in a way that makes them spread out as much as possible, which is a common theme in chemistry – things like to be spread out and happy!
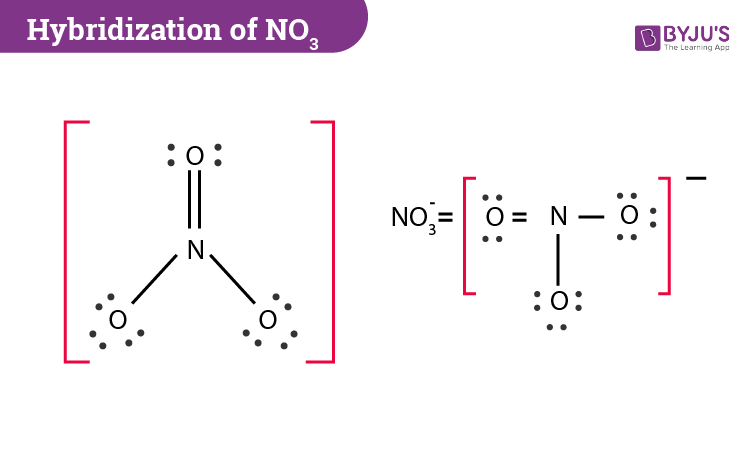
So, the nitrogen atom, now with its sp² hybrid orbitals, is ready to party with the oxygen atoms. It forms three strong connections, or bonds, with the three oxygen atoms. And guess what? Because these sp² orbitals are arranged in a specific way, the NO₃ molecule ends up with a really cool, flat, triangular shape! It's like the perfect arrangement for a game of frisbee!
Why should you care about this? Well, understanding hybridization helps us understand the shape of molecules. And the shape of a molecule is super important! It dictates how that molecule will behave. Think about a key and a lock. A key only works if its shape perfectly fits the lock. Molecules are similar; their shape determines if they can interact with other molecules.
For NO₃, its flat, triangular shape is crucial for its role in nature. It allows it to participate in important chemical reactions that help plants absorb nutrients from the soil. Without this specific shape, it might not be able to do its job as effectively, and our gardens (and the food they produce) would suffer!

It's also why certain drugs can work. The shape of the drug molecule needs to fit precisely into the shape of a receptor in your body to have its intended effect. A tiny change in shape, often due to different hybridization of the central atoms, can make a drug completely ineffective or even harmful. So, in a way, the humble act of mixing orbitals can have a big impact on our health!
Think of it like building with LEGOs. If you just have individual bricks, you can build things, but if you have specialized connector pieces that are designed to fit together perfectly, you can build much more stable and intricate structures. Hybridization is like creating those specialized connector pieces for atoms.
The central atom, nitrogen, in NO₃ uses its sp² hybridization to create three strong sigma bonds with the oxygen atoms. One of these bonds is actually a bit special; it's a double bond. This means that nitrogen shares two pairs of electrons with one of the oxygen atoms. And guess what? The electrons in that double bond aren't just stuck in one place; they can actually spread out over all three oxygen atoms! This is called resonance, and it makes the NO₃ ion even more stable.

It's like having a group of friends who are so in sync that they can finish each other's sentences, or even share a single thought! This delocalization of electrons in resonance makes the NO₃ ion very happy and resistant to breaking apart.
So, the next time you see a leafy green salad or admire a blooming flower, you can take a moment to appreciate the invisible dance of atoms, the cleverness of hybridization, and the beautiful, flat, triangular shape of the nitrate ion that makes it all possible. It’s a little peek into the incredible chemistry that’s happening all around us, all the time, making our world work.
It’s this fundamental understanding of how atoms bond and arrange themselves that forms the bedrock of so much of our modern world, from the materials in our phones to the medicines that heal us. So, even though hybridization might sound like something from a complex textbook, it’s actually a surprisingly relatable concept when you think about atoms as tiny chemists figuring out the best way to connect and build. Pretty neat, right?
