What Is The Hybridization Of The Central Atom In Brf5
Imagine a world where molecules, those tiny building blocks of everything around us, have their own personalities. They’re not just boring arrangements of atoms; they’re like little families, each with its own unique way of interacting and shaping up. Today, we're going to peek into the cozy, slightly quirky, and surprisingly strategic world of a molecule called Bromine Pentafluoride, or BrF5 for short. And the star of our show, the central figure holding this whole gang together, is the bromine atom. We’re going to uncover a little secret about our friend bromine: its hybridization!
Now, "hybridization" might sound like something out of a science fiction movie, or perhaps a particularly challenging level in a video game. But in the world of molecules, it's a bit like giving your atoms a makeover. Think of it as our bromine atom deciding to wear a different outfit, or perhaps a special blend of outfits, to best get along with its new neighbors – those five zesty fluorine atoms.
Our central character, bromine, is a bit like a seasoned parent. It has a certain number of "slots" or "spaces" where it can hang out with its fluorine "kids." Normally, it might just use its regular atomic "rooms" to say hello. But with five energetic fluorines demanding attention, our bromine atom needs to be extra resourceful. It needs to create more welcoming spaces, more opportunities for connection.
This is where the magic of hybridization comes in. Instead of just using its pre-existing atomic rooms (which, in chemistry lingo, are called atomic orbitals), the bromine atom decides to get creative. It takes some of its own "rooms" and mixes them up, like a baker combining different flours to make the perfect dough. It’s not creating new rooms from scratch, mind you, but rather blending and reshaping the ones it already has.
For BrF5, our bromine atom is a bit of a social butterfly. It has five fluorine friends to greet, and a little bit of quiet time for itself, too. So, it decides to blend one of its "s" rooms with three of its "p" rooms. This creates four brand new, equally sized, and perfectly shaped rooms. These are its sp3 hybridized orbitals. Think of these as specially designed, multi-purpose rooms, perfect for entertaining guests. Each of these rooms is ready to welcome a fluorine atom.

But wait, there's more! Even after creating these four sp3 rooms, our bromine atom still has a couple of "rooms" left over. One is a lone "d" room, a bit more spacious and perhaps a little less tidy than the others. This "d" room doesn't get mixed in with the others. It stays as it is, contributing to the overall shape and feel of the molecule. So, in total, for its hybridization, our bromine atom uses one 's' orbital, three 'p' orbitals, and one 'd' orbital. This leads to a total of five hybridized orbitals – perfect for hosting its five fluorine visitors. This specific blend of orbitals is often described as sp3d hybridization. It’s like having a special set of five "connector pieces" that perfectly fit the needs of the fluorine atoms.
It's not about the atoms changing their fundamental nature, but rather about them adapting their forms to create the most stable and comfortable arrangements. It's a testament to nature's ingenuity, finding clever ways to make things work!
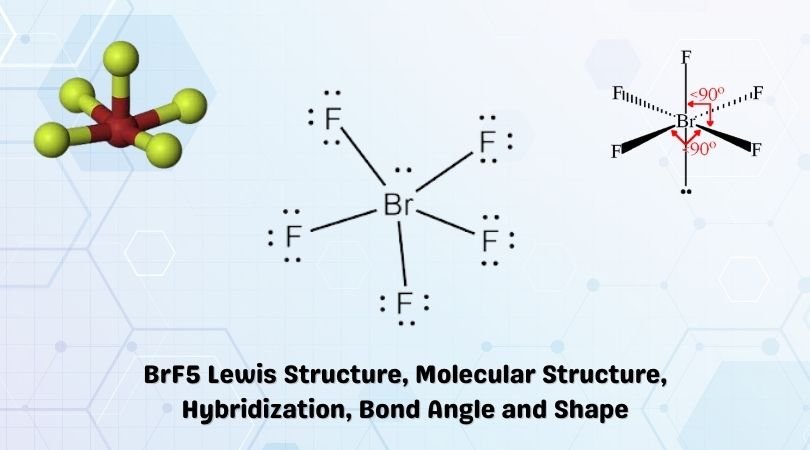
BrF5 Lewis structure, Molecular geometry, Bond angle, Hybridization
Now, what about the rest of the "rooms"? Remember that spare "d" orbital that didn't get mixed? It turns out that this unused room is quite important. It’s not used for bonding with the fluorine atoms directly. Instead, it becomes a little "storage room" for a pair of electrons that don't have a partner. These are called lone pair electrons. They’re like the quiet observers in the family, not directly interacting but still influencing the overall atmosphere.
This combination of five bonding pairs (one for each fluorine) and one lone pair of electrons gives BrF5 a rather interesting shape. It's not perfectly symmetrical. Imagine a square, but with one corner slightly pushed down or pulled up. This shape is called a square pyramidal shape. It’s a bit like a charming little house with a slightly quirky roofline. The bromine atom is at the peak, with four fluorines forming the base of the pyramid, and the fifth fluorine sitting directly above one of the base fluorines, or perhaps slightly off-center due to the presence of that lone pair.
So, the hybridization of the central bromine atom in BrF5 is a story of adaptation and resourcefulness. It’s about an atom understanding its needs and its surroundings, and then creatively rearranging its "space" to forge strong, stable connections. It’s a beautiful example of how even the smallest components of our universe are constantly working, adapting, and creating structures that are both functional and, in their own molecular way, quite elegant.

