What Is The H3o+ Of Pure Water At Body Temperature

Imagine your body as a bustling city, full of tiny citizens doing important jobs. We're talking about cells, muscles, and all the amazing parts that keep you running. Now, what do you think is the most abundant, essential, and often overlooked resident of this incredible metropolis? You guessed it – good old water!
But water isn't just sitting there, doing nothing. It's a super-active participant in everything that goes on inside you. It's the ultimate multitasker, acting as a delivery service, a cleaning crew, and even a little bit of a dance floor for tiny molecules.
When we talk about pure water, we're not just talking about something you'd find in a fancy bottle. We're talking about the stuff that makes up about 60% of you! Yes, you're practically a walking, talking water balloon, and that's a good thing.
Now, here's where things get a little magical. Even in the purest water, things aren't as still as they seem. It’s like a perpetual party where molecules are constantly mingling and shaking things up.
The main character in this tiny party is the water molecule itself, which we often represent as H₂O. Think of it as a tiny Mickey Mouse head, with an oxygen atom as the big round face and two hydrogen atoms as its ears. Cute, right?
But sometimes, these little Mickey Mouse heads get a little excited. They bump into each other, and sometimes, one of the hydrogen "ears" decides to go on a little adventure.
This adventurous hydrogen ear, now a positively charged little fellow, floats off and finds a neighborly water molecule to cling onto. It's like a shy friend asking to join a group hug.
![SOLVED: For Kw, the product of [H3O+] and [OH-] for pure water is 1.0](https://cdn.numerade.com/ask_images/8bdde4ea9a5f41928bcff1038a44d42f.jpg)
When this happens, a special new character emerges. This is where we meet the famous H₃O⁺. It’s basically a water molecule that has adopted an extra, positively charged hydrogen atom. We affectionately call it the hydronium ion.
So, in pure water, you have lots of regular H₂O molecules hanging out. But you also have a few of these special H₃O⁺ buddies, and they're constantly being made and unmade.
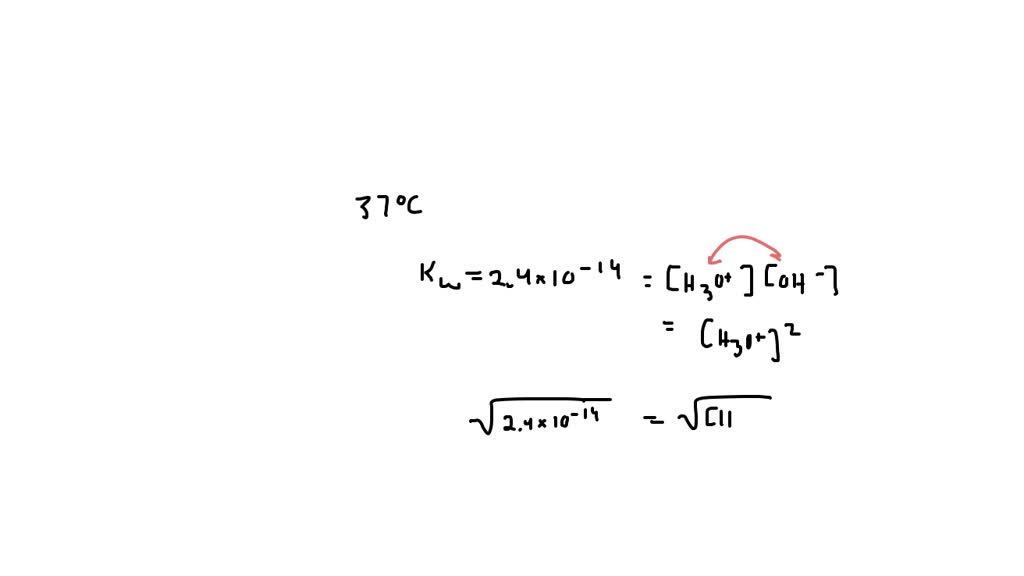
Now, let’s bring this party to your body temperature. Think about when you feel perfectly comfortable, not too hot and not too cold. That's around 37 degrees Celsius, or about 98.6 degrees Fahrenheit. It's your happy place temperature!
At this delightful body temperature, the little water molecules are just buzzing with energy. They're lively, but not chaotically so. It’s the perfect balance for all the amazing work they do inside you.
And guess what? This perfect temperature also affects how often our little hydrogen ears go on their adventures. It turns out that at body temperature, the number of H₃O⁺ ions is quite specific.
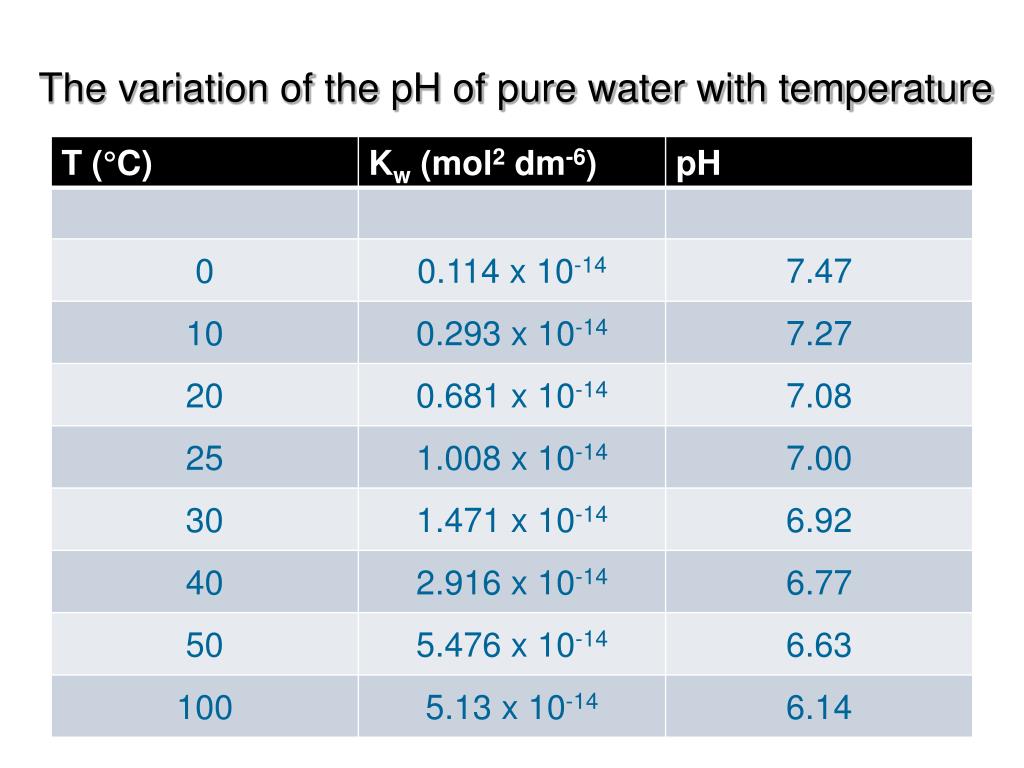
So, what is the H₃O⁺ of pure water at body temperature? It's a very, very small number. It's like finding a single, perfectly formed snowflake in a vast desert.
We’re talking about a concentration of about 1 x 10⁻⁷ moles per liter. Now, don't let that scientific-sounding phrase scare you! Think of it this way: for every ten million water molecules, only one is in the H₃O⁺ form.
That's incredibly tiny! It’s like having one super-special VIP guest at an enormous concert.
This incredibly low number is actually a good thing. It means that pure water is, well, pretty neutral. It's not trying to be an acid, and it's not trying to be a base. It’s just… water.
And this neutrality is crucial for your body. Your cells are very sensitive. Imagine them as delicate little flowers; you wouldn’t want to pour something too strong on them, right?
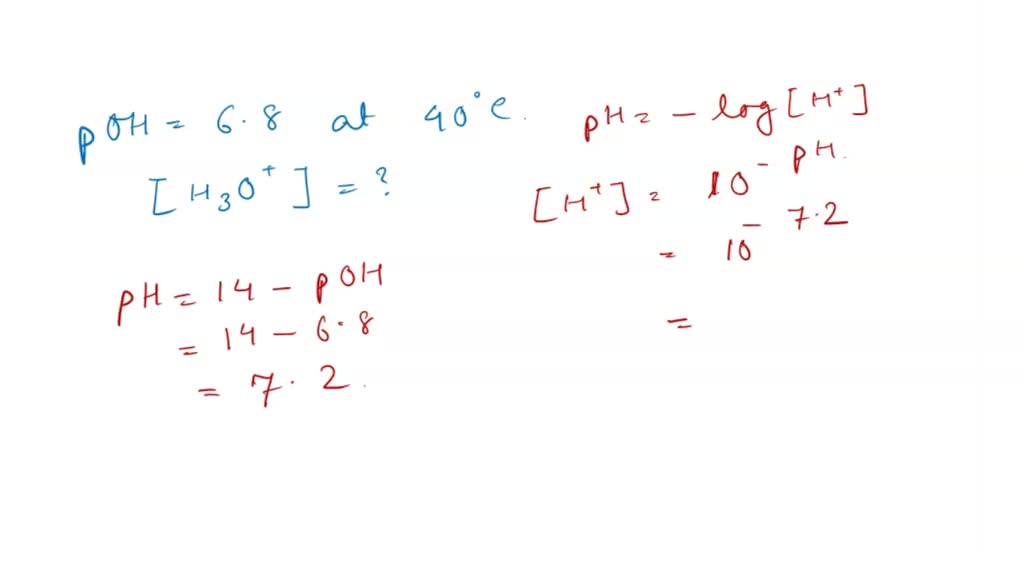
The fact that the H₃O⁺ concentration is so low and stable at your body temperature is like having a perfectly calm, reliable environment for your cells to thrive in. It’s the quiet hum of a well-oiled machine.
So, next time you take a sip of water, or even just feel the warmth of your own body, remember this tiny, fascinating secret. You're filled with a universe of water molecules having a gentle, regulated party, with just the right touch of adventurous H₃O⁺.
This low and steady presence of hydronium ions is like the gentle background music that allows the main performance – your life – to play out perfectly.
It's a testament to the elegant simplicity of nature, even at the microscopic level. That cool, refreshing glass of water you enjoy? It's not just H₂O; it’s a tiny chemical ballet, perfectly choreographed for your well-being.
The amazing thing is how nature has designed our bodies to work with this incredibly dilute solution. We are, in essence, water-based beings, and the chemistry of that water is finely tuned to keep us going.
Think about how much we take water for granted. We drink it, we bathe in it, we cook with it. It’s so common, it’s almost invisible.
But within that seemingly simple liquid, there’s this constant, silent hum of activity, a delicate balance maintained by the presence of these H₃O⁺ ions.
It's a beautiful reminder that even the most ordinary things can hold extraordinary stories. Your body's water is a constant source of wonder, a testament to the power of tiny molecules working in harmony.
So, raise a glass (or just a sip!) to your internal water, to its incredible properties, and to the tiny, adventurous H₃O⁺ ions that are always there, keeping things just right at your perfect body temperature. Cheers to you, and cheers to your amazing internal world!
