What Is The Ground State Electron Configuration Of Arsenic
Ever wondered what makes atoms tick? It’s a question that might sound a bit academic, but understanding something like the ground state electron configuration of arsenic can actually be quite fascinating and surprisingly relevant. Think of it like peeking behind the curtain of the universe, discovering how the tiny building blocks of everything behave when they’re at their most stable, their most comfortable. It’s a fundamental concept in chemistry that explains why elements are the way they are and how they interact.
So, what exactly is the ground state electron configuration? Imagine an atom as a tiny solar system. The nucleus is the sun, and electrons are the planets orbiting around it. But these orbits aren't random! Electrons exist in specific energy levels and sublevels, like distinct rings or even specific paths within those rings. The ground state simply means all the electrons are in their lowest possible energy positions. It's the atom’s default setting, its most relaxed and stable arrangement. The electron configuration is the map of where each electron hangs out.
Why bother with this kind of atomic mapping? For starters, it's the bedrock of understanding chemical behavior. Knowing arsenic’s ground state electron configuration tells us how it will form bonds with other elements, what kind of compounds it will create, and its overall reactivity. This knowledge is crucial in countless fields. In education, it's a cornerstone of learning chemistry, helping students grasp the periodic table's organization and predict element properties. Think of science classes – this is the kind of detail that unlocks deeper understanding.
Beyond the classroom, these principles have real-world applications. Materials science relies heavily on understanding electron configurations to design new alloys, semiconductors, and even pharmaceuticals. For example, understanding how arsenic's electrons are arranged helps in developing specific treatments or understanding its toxicity. In the realm of technology, elements like arsenic have historically played a role in semiconductors, and knowing their electronic structure is key to their function. Even understanding pollution and environmental remediation can involve knowing how elements like arsenic interact with their surroundings.
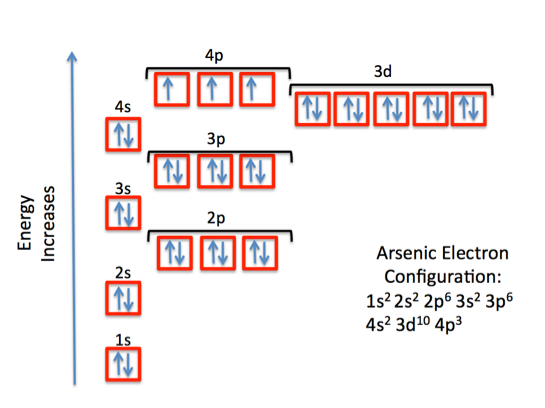
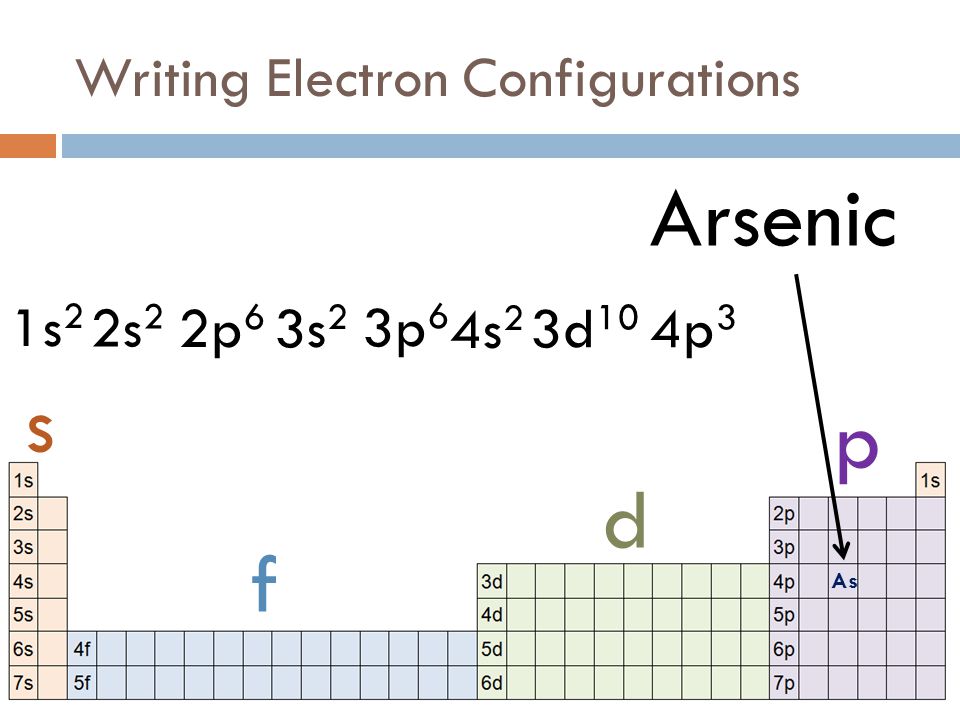
Curious to explore this yourself? You don't need a lab coat! A great way to start is by looking at a periodic table. Notice how elements are arranged? It’s not arbitrary; it’s a direct reflection of their electron configurations. You can find charts online that show the electron configurations for various elements. For arsenic (As), which is element number 33, its ground state configuration is generally written as [Ar] 4s² 3d¹⁰ 4p³. Don't let the shorthand scare you! [Ar] represents the configuration of Argon, a noble gas, and the rest tells us how the remaining electrons fill specific energy levels and sublevels. It's like a shorthand code for atomic structure.
A simple tip to visualize this is to think about filling shelves in a library. Each shelf has a certain capacity, and books (electrons) fill them up in a specific order, starting from the lowest shelves (lowest energy levels). You can even search for interactive electron configuration diagrams online to see this visually. It’s a journey into the intricate, elegant rules that govern the very fabric of our existence, starting with the seemingly simple question: what’s arsenic doing when it’s just being arsenic?
