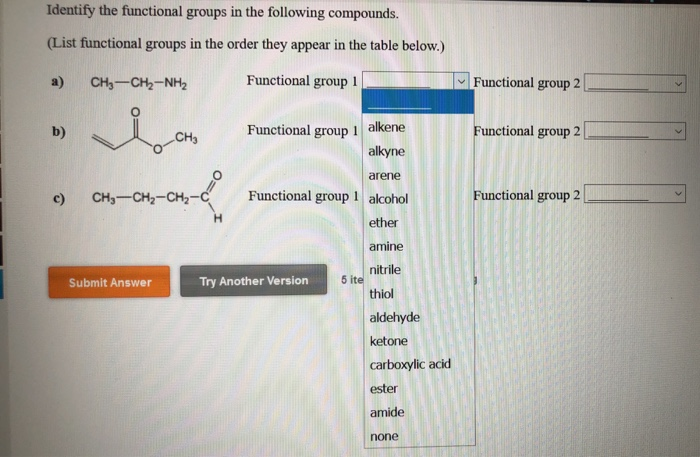
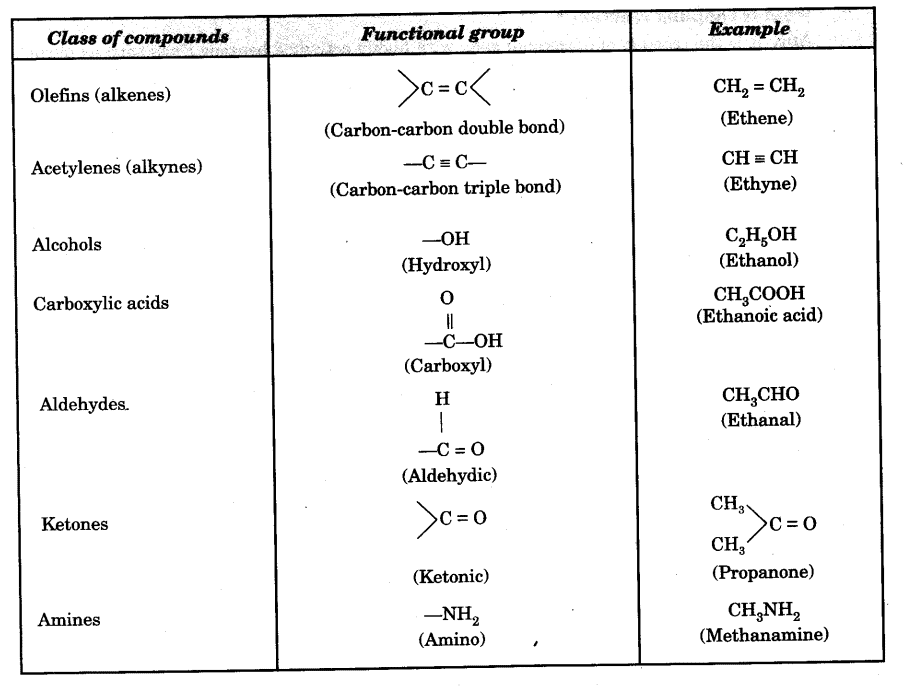
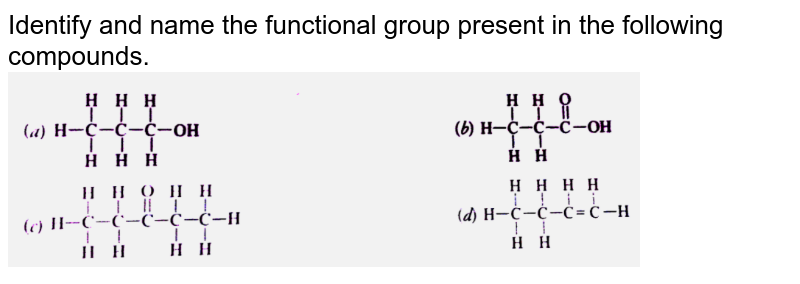
What Is The Functional Group In The Following Compound

Let's dive into the wacky world of molecules! You know, those tiny building blocks that make up everything around us. Sometimes, these molecules have special little bits attached to them. Think of it like a car having a cool spoiler or a comfy seat – these bits give the molecule its personality and a whole bunch of superpowers.
These special bits are what scientists call functional groups. And honestly, they're the rockstars of the chemistry world. They're not just random attachments; they're the parts that actually do things. They decide how a molecule will behave, what it can react with, and what kind of cool tricks it can pull off.
Imagine you have a plain old toy car. It rolls, sure. But what if you add a little jet engine to it? Suddenly, it's not just a toy car anymore, right? It's a super-fast, zoomy thing! That jet engine is kind of like a functional group. It gives the car a whole new set of abilities.
So, when we look at a particular compound, one of the most exciting things to figure out is: what's its functional group? It's like finding the secret ingredient that makes this particular molecule so special. It's the key to unlocking its mysteries and understanding its unique talents.
Let's say we're looking at a molecule. It might seem a bit like a jumble of atoms at first. But then, we spot it – a specific arrangement of atoms that pops out. That's our functional group, and it's ready to steal the show!
These groups are like the LEGO bricks of chemistry. You can have a basic brick, but then you can add wheels, or a little flag, or a tiny minifigure. Each addition changes the whole structure and what it can do. Functional groups are those amazing additions!
Think about water. It's pretty simple, right? Just two hydrogen atoms and one oxygen atom. It's incredibly important, of course, but it doesn't have a flashy functional group that makes it do a million different things in super exciting ways all on its own.
But then, you start adding different functional groups to other molecules, and boom! You get all sorts of amazing substances. It's like taking a plain canvas and adding different colors, textures, and shapes to create a masterpiece. The functional group is the artist's brush that adds all the flair.
So, what exactly makes a functional group so entertaining? It's their sheer versatility! They're the workhorses, the problem-solvers, the characters in the molecular drama. Without them, molecules would be a lot more… boring. And who wants boring when you can have spectacular?
Let's talk about some of the fan favorites. You've got groups that make things sweet, like the ones found in sugars. Then there are groups that make things oily, like in fats. And some groups are super reactive, ready to jump into action and form new bonds. It’s like a molecular superhero league!
One of the most common and super useful functional groups is the hydroxyl group. It’s just an oxygen atom bonded to a hydrogen atom (-OH). Sounds simple, right? But this little guy is everywhere!
When you see a hydroxyl group attached to a molecule, it often makes that molecule soluble in water. Think about alcohol – that's ethanol, and it has a hydroxyl group. That's why it mixes with water! It's like a friendly handshake between the molecule and water.
Another crowd-pleaser is the carbonyl group. This one has a carbon atom double-bonded to an oxygen atom (C=O). It’s like a tiny, powerful magnet within the molecule.
The carbonyl group is super important because it can be found in two major types of compounds: aldehydes and ketones. They’re like cousins in the molecular family, and they both owe their cool properties to that carbonyl group.
Aldehydes have the carbonyl group at the end of a carbon chain. Ketones have it somewhere in the middle. This little positional difference can lead to different personalities and reactions, which is just fascinating! It’s like how twins can be so similar but still have their own unique quirks.
And then there's the carboxyl group. This one is a bit of a celebrity. It’s a carbonyl group attached to a hydroxyl group (-COOH). It's acidic, which means it can donate a proton.
Carboxyl groups are the stars of carboxylic acids. Think of vinegar – that's acetic acid, and it’s all thanks to that carboxyl group! It gives food that tangy, zesty flavor. It’s the secret behind many delicious tastes.
So, when you encounter a new compound, the first question on your mind should be: "What's its functional group?" It’s like getting the VIP pass to understanding that molecule. It tells you its name, its game, and its potential for awesomeness.
These groups aren't just for scientists in lab coats, either. They're behind so many things we use and experience every day. From the flavors in our food to the medicines that heal us, functional groups are the unsung heroes.
Let's imagine a compound. It's got a chain of carbon atoms, like a little molecular backbone. And then, sticking out from that chain, we see a special arrangement. Maybe it’s a sulfur atom with two hydrogen atoms attached (-SH). This is called a thiol group.
Thiols are known for their… distinctive aromas. Yes, some can smell quite… pungent. But they are incredibly important in biological processes, and they can also be used in industry. They might have a strong smell, but they’re mighty in function!
Or, what if we see a nitrogen atom bonded to hydrogen atoms? This is an amine group. Amines are basic and are fundamental building blocks of proteins. They’re like the essential connectors that hold our bodies together.
The type and position of these groups can lead to compounds with wildly different properties. A molecule with a hydroxyl group might be a refreshing drink component, while a similar molecule with a different functional group could be a powerful cleaning solvent. It’s the subtle differences that create the dramatic effects!
It’s this complexity and variety that makes chemistry so endlessly fascinating. Every time you identify a functional group, you’re not just labeling a part of a molecule; you’re understanding its purpose, its behavior, and its potential. It's like deciphering a secret code that reveals the molecule's true identity.
So, the next time you hear about a chemical compound, don't be intimidated. Just remember the concept of functional groups. Look for those distinctive arrangements of atoms. They are the keys to unlocking the molecule’s story. They’re the reason why some molecules are sweet, some are sour, some are sticky, and some are downright explosive!

It’s a bit like being a detective. You’re presented with a clue (the compound), and your job is to find the main suspect (the functional group) that’s causing all the action. And once you find it, you can start to understand the whole story.
The beauty of functional groups lies in their predictability, too. Once you learn about the properties of a specific functional group, you can often predict the behavior of any molecule that contains it. It’s a system of building blocks that chemists have figured out.
So, if you’re ever curious about what makes a particular substance do what it does, try to find its functional group. It’s where the real magic happens, the core of its identity, and the source of its unique character. It’s what makes chemistry so alive and exciting!
So, what is the functional group in a given compound? It's the special cluster of atoms that dictates its chemical personality and makes it do its thing! It's the heart and soul of the molecule.
It's the part that allows it to participate in reactions, to bond with other molecules, and to exhibit its particular properties. It’s the reason why sugar is sweet and salt is… well, salty!
This journey into understanding functional groups is just the beginning. It opens up a whole universe of chemical wonders, from the complex molecules in your own body to the vast substances that make up our world. Every new functional group you discover is like finding a new superpower!
And who knows, by understanding these simple groups, you might just unlock a new appreciation for the incredible science that surrounds us every day. It’s a tiny peek into a gigantic and endlessly fascinating world.
