What Is The Electron Pair Geometry For N In Nf3
Hey there, fellow science enthusiast! So, you're curious about the electron pair geometry for nitrogen in NF3, huh? Awesome! Let's grab a virtual coffee (or tea, no judgment!) and dive into this, shall we?
It's not as scary as it sounds, promise! Think of it like this: we're basically trying to figure out how all those little electron buddies are arranging themselves around the nitrogen atom in our NF3 molecule. It’s all about giving everyone their own personal space, you know?
Nitrogen, bless its heart, is usually the center of attention in NF3. It’s got its own set of electrons, and then it’s making nice with three fluorine atoms. So, what’s the deal with all these electrons? Are they all huddled together, or are they spread out?
This is where VSEPR theory comes in, which, by the way, sounds way cooler than it is. It’s basically just a fancy way of saying that electrons, being the natural-born introverts they are (or maybe just really wanting their own space!), will push each other away as much as possible. It’s like trying to fit too many people on a tiny couch – everyone ends up squished! Not ideal, right?
So, for our nitrogen friend in NF3, we need to count up all the things that have electrons around it. This includes not just the electrons it's sharing with the fluorines (those are the bonds, duh!), but also any lone pairs of electrons. Lone pairs are like the nitrogen's personal stash, not involved in bonding, just hanging out. And trust me, they take up space!
Let’s break it down. Nitrogen, from its cozy spot on the periodic table, has 5 valence electrons. That’s the outer shell, the ones involved in all the action. And each of those three fluorine atoms chips in one electron to make a bond. So, that’s 3 electrons from fluorine, adding to nitrogen's original 5. But wait, we're only counting what's around the nitrogen. So, three bonds means three electron pairs that are being shared. Easy peasy so far, right?
But here’s the twist, the plot thickening! Nitrogen started with 5 valence electrons. It’s using 3 of them to make those bonds with fluorine. What happened to the other 2? Aha! They form a lone pair. Yep, nitrogen is keeping a little secret stash for itself. How dramatic!

So, if we’re counting everything around the nitrogen, we have our three bonding pairs (one for each N-F bond) and our one lone pair. Add ‘em up, and what do we get? Four groups of electrons! Four electron domains, if you want to be all scientific about it.
Now, here's the fun part. When you have four groups of electrons all trying to get away from each other, what’s the best arrangement? Imagine you're trying to get four people to stand as far apart as possible in a small room. They're not going to stand in a line, are they? That would be way too crowded on the ends. Nope, they're going to spread out in a way that maximizes their distance.
The magic number for four electron domains is tetrahedral. Ta-da! It’s like a little pyramid, or a tripod if you prefer. Each of those four electron groups wants to be as far away from the others as possible. Think of it like a perfectly balanced game of musical chairs, but with electrons.
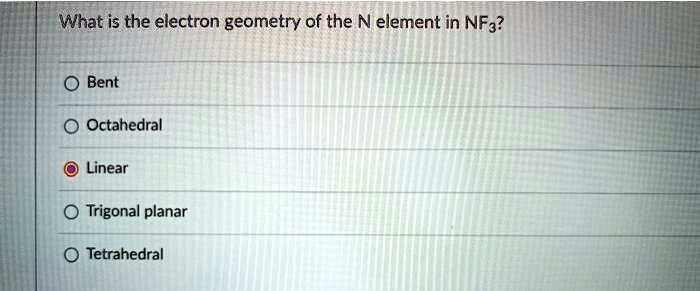
So, the electron pair geometry for nitrogen in NF3 is tetrahedral. This tells us how those electron pairs – both the bonding pairs and the lone pair – are arranged in 3D space around the central nitrogen atom. They're all pointing towards the corners of a imaginary tetrahedron.
Now, a tiny little caveat, just to keep things interesting. While the electron pair geometry is tetrahedral, the molecular geometry is a little different. This is where we only look at the positions of the atoms, not the lone pairs. Since one of those corners of our tetrahedron is occupied by a lone pair (which we can't actually see), the molecule itself looks a bit different. It’s like if you had a tripod with one leg missing – it’s still based on that tripod shape, but the overall form is different.

The molecular geometry of NF3 is actually trigonal pyramidal. Think of a little pyramid with a flat base. That's the shape of the atoms. The nitrogen is at the apex, and the three fluorines are at the base. It’s not a perfect pyramid because of the lone pair’s influence, which is a whole other conversation about bond angles, but that’s for another coffee break!
But the question was about the electron pair geometry, so let’s stick to that! It’s the foundation, the blueprint, if you will, for the molecular shape. And that blueprint is definitely tetrahedral because of those four electron domains.
It’s so important to distinguish between electron pair geometry and molecular geometry, you know? It's like the difference between the blueprint of a house and the actual house itself. The blueprint dictates how things are laid out, but the final structure can look a bit different depending on what's inside (or, in our case, what’s a lone pair!).
So, to recap, nitrogen in NF3 has 3 bonding pairs and 1 lone pair. That gives us a total of 4 electron domains. And when you have 4 electron domains, the universe conspires to arrange them in a tetrahedral fashion to minimize repulsion. It’s the most spread-out, happiest arrangement for those electron buddies.
Isn’t it neat how these simple rules can predict something as complex as molecular shapes? It’s like a little bit of chemical magic, all thanks to electrons wanting their personal space. Who knew electrons could be so particular?
Think about it: that lone pair on nitrogen is quite the character. It's not participating in the N-F bonds, but it’s still a significant player in determining the overall geometry. It’s like that one friend at the party who doesn’t say much but still influences the vibe of the whole room. That lone pair is doing just that!
And the repulsion from that lone pair is actually pretty strong. Lone pairs are like the teenagers of the electron world – they’ve got a lot of energy and tend to push around the younger, more easily influenced bonding pairs. This is why the bond angles in NF3 are a bit squished compared to a perfect tetrahedron.
In a truly tetrahedral arrangement with no lone pairs (like methane, CH4), the bond angles would be around 109.5 degrees. But in NF3, because of the lone pair’s assertive nature, those N-F bond angles are a little smaller. It's a subtle but important detail!
So, while the electron domains are arranged tetrahedrally, the atoms are arranged in a trigonal pyramidal shape. It’s a bit of a trick question if you’re not careful, but we’re here to get it right, right?
Let’s do a quick mental check. Nitrogen has 5 valence electrons. It forms 3 single bonds with fluorine. That uses 3 electrons. We have 2 electrons left, which form one lone pair. So, 3 bonding pairs + 1 lone pair = 4 electron domains. Four electron domains around a central atom will always adopt a tetrahedral electron pair geometry. Boom! Nailed it.

It’s this understanding of electron pair geometry that then leads us to predicting molecular geometry, polarity, reactivity, and all sorts of other cool stuff about molecules. It’s like building blocks for understanding chemistry.
So, next time you see NF3, you can confidently say, "Ah yes, the electron pair geometry around that nitrogen is definitely tetrahedral!" And then you can impress your friends, or at least feel smugly knowledgeable over your coffee. 😉
It’s all about those electron clouds, really. They’re like little fuzzy balloons, and they don’t want to be too close to each other. The tetrahedron shape is the best way to give them maximum personal space. It’s just good manners in the molecular world.
And remember, the key is to count all the electron domains. Don't just count the atoms. Count the bonds, and count the lone pairs. That’s your ticket to figuring out the electron pair geometry. It’s like solving a little puzzle!
So, the electron pair geometry for N in NF3 is tetrahedral. Simple, elegant, and scientifically sound. Hope this little chat over coffee (or whatever you’re sipping!) helped clear things up. Keep those science questions coming!
