What Is The Electron Configuration Of The Iodide Ion

Ever thought about the tiny building blocks of everything around us? We're talking about atoms, the microscopic superstars that make up your favorite comfy chair, the salty snack you love, and even the air you breathe. Now, within these atoms are even smaller characters called electrons, zipping around like hyperactive toddlers at a birthday party. Today, we're going to peek into the world of one particular electron posse: the one belonging to the iodide ion.
Imagine an atom is like a little apartment building. The nucleus is the landlord in the center, and the electrons are the tenants living on different floors, or energy levels. These floors have different numbers of rooms, and the electrons fill them up in a specific order. It's like a quirky housing policy – some floors are cozy studios, while others are sprawling penthouses.
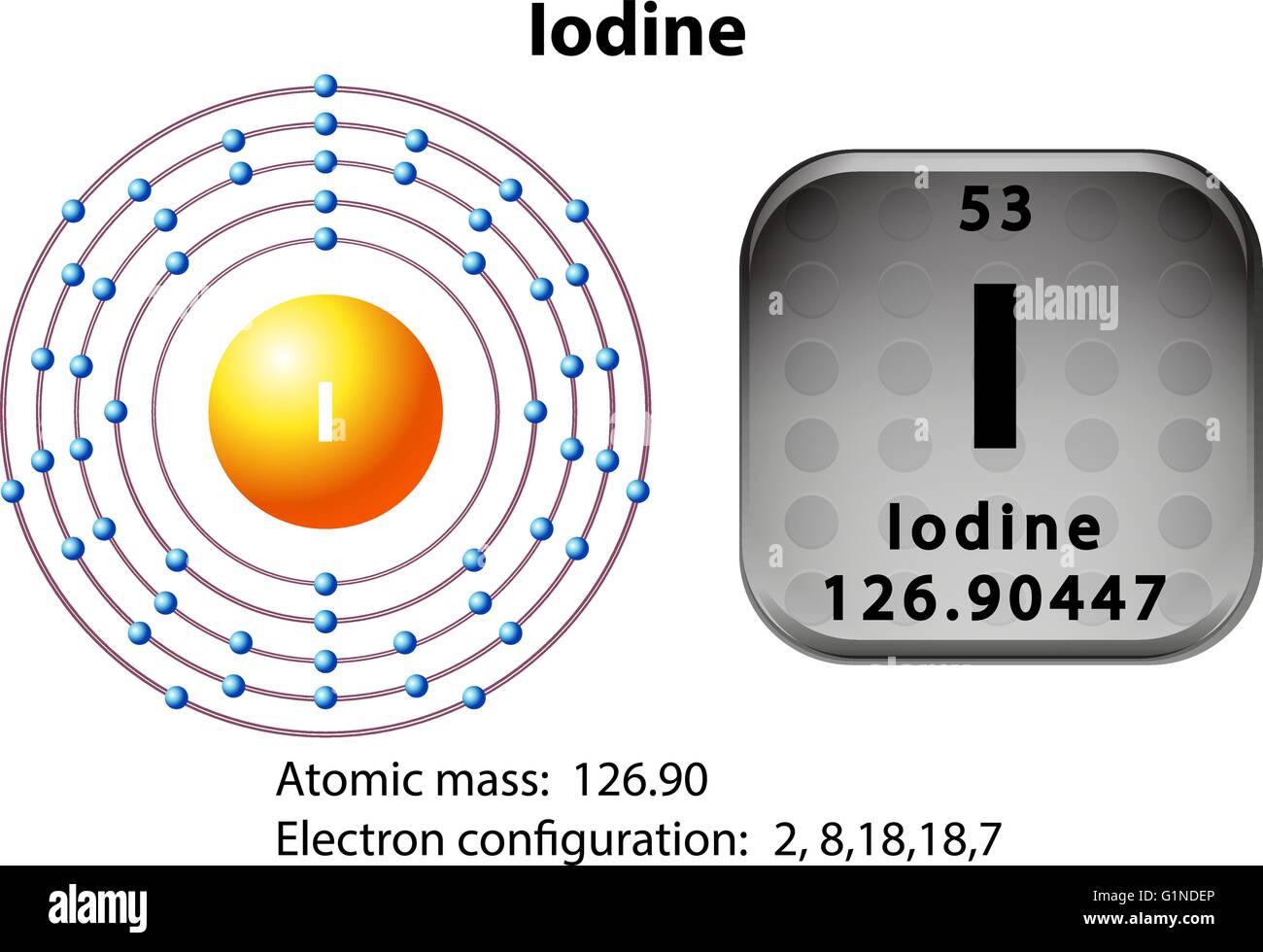
Now, our star of the show, the iodide ion, is like a tenant who’s just gotten a promotion and moved into a slightly bigger, comfier apartment. Normally, a regular iodine atom has 53 electrons, a good number of residents in its atomic apartment. But the iodide ion is special. It's like iodine that's welcomed one extra electron into its home. Think of it as a generous host who’s always got room for one more friend. So, now, instead of 53, it has a grand total of 54 electrons.
So, how do these 54 electrons arrange themselves? Well, it’s all about filling up those floors and rooms. The first floor, the one closest to the landlord, is small and can only hold 2 electrons. These are like the ground-floor residents, always right there. Then we move to the second floor, which has more room, accommodating 8 electrons. These are the folks on the second floor, a bit more spread out.
The third floor is even bigger, holding 18 electrons. These are the families on the third floor, with plenty of space to play. Then comes the fourth floor, another generous level that can fit 18 electrons. By this point, we've housed 2 + 8 + 18 + 18 = 46 electrons. We’re getting close!
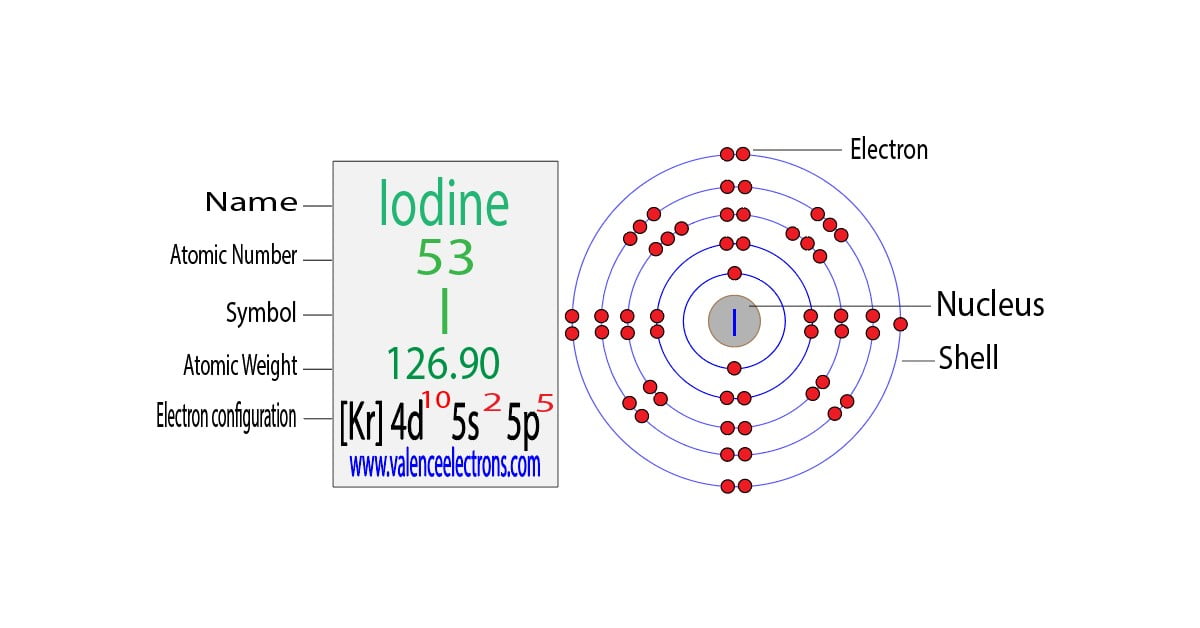
Now, here’s where it gets a little interesting. The fifth floor is where our extra electron, the one that made iodine into iodide, decides to settle. This floor can hold a whopping 32 electrons in total, but it’s a bit picky. It likes to fill up its rooms in a specific pattern. So, our remaining electrons – the 47th, 48th, 49th, 50th, 51st, 52nd, 53rd, and finally, our special guest, the 54th – find their spots. They fill up some of the available rooms, making the fifth floor quite cozy. If you were to draw it out, it would look like a meticulously organized dance card, with each electron finding its pre-assigned partner and position.
The official way scientists describe this electron arrangement is called electron configuration. For the iodide ion, it looks something like this: 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s2 4d10 5p6. Now, that might look like a secret code from a spy movie, but it's just a shorthand for where all those electrons are hanging out. Each number and letter combo tells you which floor and which type of room an electron is in. It’s like a detailed address for every single electron in our friendly iodide ion.
What’s truly heartwarming about this is how these electrons, despite being so tiny, have a fundamental drive to be in stable, comfortable positions. The iodide ion, by gaining that extra electron, achieves a particularly stable electron configuration, much like a perfectly balanced game of Tetris. It’s this quest for stability that drives atoms to bond and form the world we see and experience. So, next time you’re enjoying a perfectly seasoned meal, remember the humble iodide ion and its beautifully organized electron neighborhood, all working together to make things just right!
The iodide ion is like a little apartment building that decided to be extra welcoming, inviting one more electron to join the party. And that extra electron makes all the difference in how the whole place is arranged!
It's fascinating to think about how these invisible particles are constantly interacting and organizing themselves. The electron configuration of the iodide ion is a testament to the order and elegance found even at the most fundamental level of existence. It’s a story of balance, of finding the right fit, and of a surprisingly stable home for its many electron residents.
