What Is The Electron Configuration Of A Neutral Magnesium Atom

So, you’ve probably heard of atoms, right? Those tiny little LEGO bricks that make up absolutely everything. From your morning coffee to that slightly-too-loud car that just drove by, it’s all atoms. And within these atoms, there are even tinier bits called electrons. Think of electrons as the hyperactive toddlers of the atomic world, always bouncing around, but in very specific, organized ways. Today, we’re going to chat about the electron configuration of a neutral magnesium atom. Sounds fancy, right? But don't worry, it's less like a complicated tax form and more like figuring out where to put your socks when you do laundry.
First off, let's get to know our star for today: Magnesium. If atoms were people, magnesium would be that friendly, reliable neighbor who’s always willing to lend a cup of sugar or help you jump-start your car. It’s an element with a bit of a reputation for being a team player in chemical reactions. You know, the kind that’s happy to share its electrons if it means making a new friend (or, in chemistry terms, forming a bond).
Now, what does "neutral" mean in this context? Well, in the atomic world, being neutral is like having an equal number of pros and cons, or an even number of cookies and kids to share them with. It means the atom has the same number of positive protons in its nucleus as it has negative electrons buzzing around the outside. No net charge, just chilling. It’s not trying to steal anyone’s electrons, and no one’s really trying to steal theirs. It’s just… balanced. Like a perfectly calibrated scale, or a well-behaved toddler who’s just had a nap.
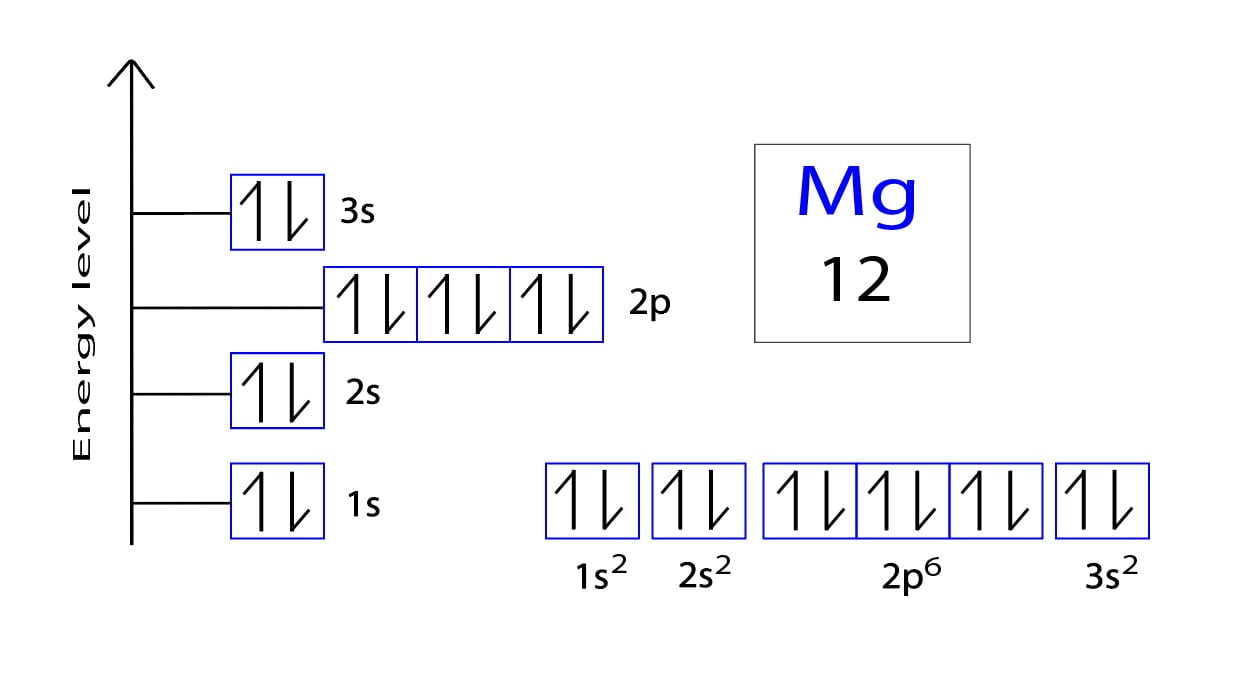
So, how many electrons are we talking about for a neutral magnesium atom? This is where we bring in the trusty atomic number. Every element has its own unique atomic number, like a social security number for atoms. For magnesium, that number is 12. And since our magnesium atom is neutral, it means it has 12 protons and, you guessed it, 12 electrons. These 12 electrons are the main characters in our story, and they’re about to go on an adventure!
These electrons don't just float around randomly, like confetti at a parade. Oh no. They have a very specific way of arranging themselves, almost like they’re queuing up for the best seats at a concert. This arrangement is called the electron configuration. It tells us which "energy levels" or "shells" the electrons occupy, and how many electrons are in each of those shells.
Think of the atom’s nucleus as the central stage. The electrons orbit this stage in distinct layers, or energy levels. The closer an electron is to the nucleus, the less energy it has. The farther away, the more energy it has. It’s like being closer to the heater on a chilly day – warmer, more comfortable, less energy needed to stay that way. The outermost shells are like the balcony seats, a bit farther from the action, and usually occupied by the more energetic, potentially rebellious electrons.

These energy levels are named by numbers: 1, 2, 3, and so on, starting from the one closest to the nucleus. And within each energy level, there are sub-levels, kind of like different sections in the concert hall. We have the 's' sub-level, the 'p' sub-level, the 'd' sub-level, and even the 'f' sub-level, though for magnesium, we won't get that far. Each of these sub-levels has a specific shape and can hold a certain number of electrons.
The 's' sub-level is like a nice, round, cozy studio apartment. It can only hold a maximum of 2 electrons. The 'p' sub-level is more like a three-bedroom house, able to accommodate up to 6 electrons. The 'd' sub-level is a mansion, holding up to 10 electrons, and the 'f' sub-level? That’s a whole resort, with space for 14!
Now, the electrons fill these levels and sub-levels in a specific order, starting with the lowest energy. It's like a “first come, first served” policy, but with strict rules. They fill up the 1s sub-level first, then the 2s, then the 2p, and so on. This principle is called the Aufbau principle, which sounds like a German word for "building up" – because that’s exactly what we’re doing, building up the electron configuration!

So, let’s get down to business with our 12 electrons for the neutral magnesium atom. We start with the very first energy level, which only has an 's' sub-level. This 's' sub-level can hold 2 electrons. So, our first two electrons happily hop into the 1s sub-level. We write this as 1s². The '1' is the energy level, the 's' is the sub-level, and the '²' tells us there are 2 electrons there. Easy peasy!
We've used up 2 electrons. We have 12 total, so we still have 10 left to place. We move on to the next energy level, which is level 2. Level 2 has both an 's' sub-level and a 'p' sub-level. First, we fill the 's' sub-level. The 2s sub-level can hold 2 electrons. So, the next two electrons go into the 2s, giving us 2s². Now we’ve used 2 + 2 = 4 electrons.
We’re still short 8 electrons (12 - 4 = 8). We move on to the next sub-level in the second energy level: the 2p sub-level. Remember, the 'p' sub-level can hold up to 6 electrons. So, we fill that up with 6 electrons. This part of our configuration becomes 2p⁶. Now we’ve used 4 + 6 = 10 electrons.

We’ve got 2 electrons left to place (12 - 10 = 2). We’ve finished up the second energy level. Time to move to the third energy level! The third energy level starts with an 's' sub-level. The 3s sub-level can hold 2 electrons. And guess what? We have exactly 2 electrons left! Perfect! So, these last two electrons snuggle into the 3s sub-level, making it 3s².
And there you have it! We've successfully placed all 12 electrons. The complete electron configuration for a neutral magnesium atom is: 1s²2s²2p⁶3s².
Let’s break it down one last time, like we’re reviewing the seating chart for our atomic concert: * 1s²: The first row, front and center, a cozy spot for 2 electrons. * 2s²: The next row, a slightly bigger seat, but still just 2 electrons. * 2p⁶: The next section over, a larger area, filled with 6 electrons. * 3s²: The farthest row, a prime spot for the last 2 electrons, ready for action.
Why is this even important, you might ask? Well, it’s like knowing your own strengths and weaknesses. The electrons in the outermost shell, the ones in the 3s² for magnesium, are called the valence electrons. These are the ones that are most likely to get involved in chemical reactions. They're the ones that get passed around, shared, or even given away when magnesium decides to bond with another element. Think of them as the extroverts of the atom, always ready to mingle.
Magnesium, with its 2 valence electrons in the 3s sub-level, is quite happy to let those two go to form stable bonds. It's like having two extra coins in your pocket you don't really need – you’re more than happy to give them away to buy a snack or contribute to a group gift. This is why magnesium readily forms ionic compounds, like when it combines with oxygen to create magnesium oxide, a key component in things like antacids and even some ceramics. It's basically saying, "Here, take these two, and let's make something cool together!"
Understanding electron configuration is like having a secret decoder ring for how elements will behave. It helps chemists predict what kind of reactions an element will participate in, what kind of bonds it will form, and what properties it will have. It’s the fundamental blueprint for the entire universe of chemistry. So, the next time you see magnesium, whether it's in a fireworks display (it adds that brilliant white spark!) or in your multivitamin, you can think, "Ah, yes, that’s a 1s²2s²2p⁶3s² kind of element."
It’s a beautiful, organized chaos, really. These tiny electrons, following these incredibly precise rules, are responsible for the diversity and complexity of everything we see and interact with. From the metallic sheen of a fork to the glow of a lightbulb, it all comes down to how these little guys are arranged. So, next time you’re sorting your laundry, just remember you’re not the only one dealing with an organized system. The atoms are doing it too, and magnesium is doing it with a very specific, very predictable, 1s²2s²2p⁶3s² kind of flair.
