What Is The Electron Configuration For Chlorine
Hey there, science enthusiasts and curious minds! Ever found yourself staring at a periodic table and thinking, "Okay, but what's really going on in there?" We're going to dive into a little bit of atomic jazz today, and trust me, it's way more fun than you might think. We're talking about the electron configuration of chlorine. Sounds intimidating? Nah! Let's make it as breezy as a spring afternoon.
So, what even is electron configuration? Think of an atom like a tiny, bustling apartment building. The nucleus is the lobby, and the electrons are the residents. But these residents aren't just hanging out anywhere! They have specific floors and rooms they like to occupy. Electron configuration is just a fancy way of describing where these little electron tenants are living in their atomic abode.
And why chlorine, you ask? Well, chlorine is a pretty common element, found in everything from your kitchen salt (sodium chloride, yum!) to swimming pools. It’s like the reliable friend of the periodic table – always there, doing its thing. Understanding its electron configuration is like getting a backstage pass to see how it operates. And who doesn't love a good backstage pass?
Let's Meet Chlorine!
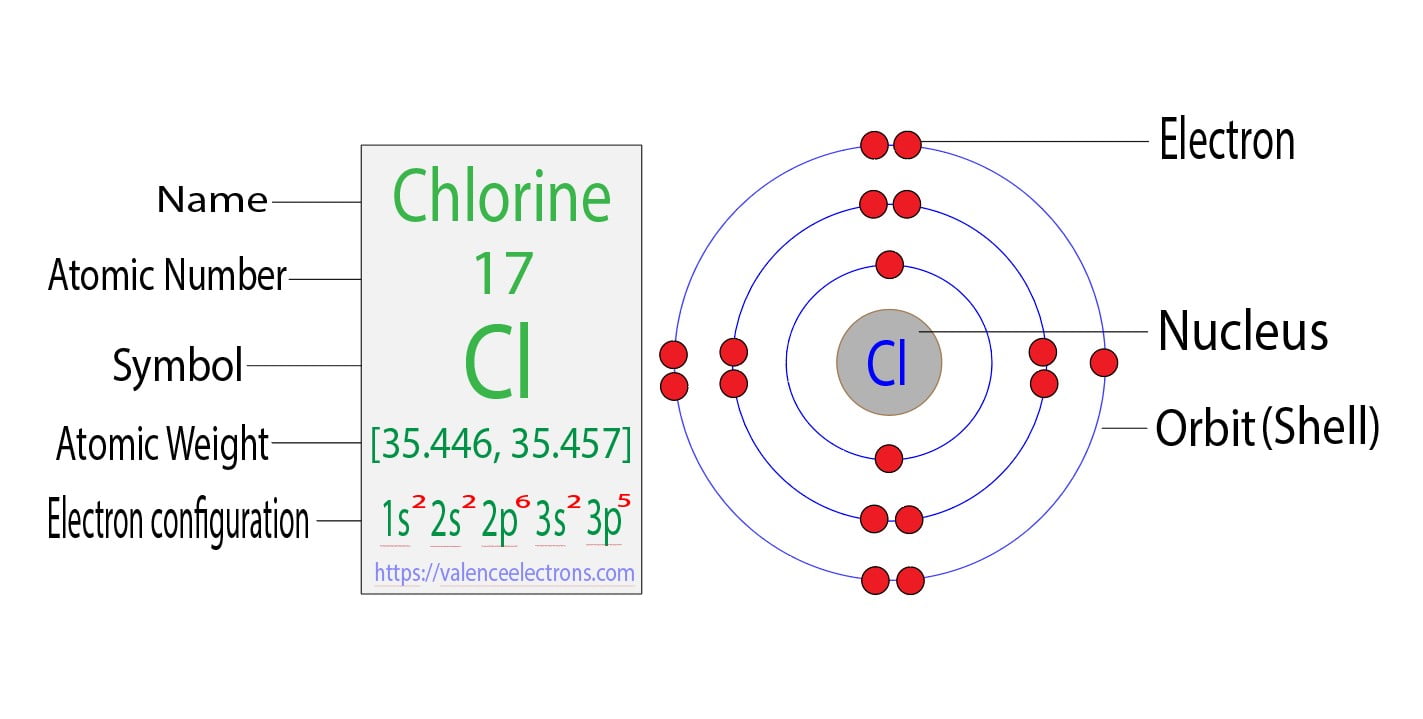
First off, let's give our star player a proper introduction. Chlorine, with the symbol Cl, is element number 17 on the periodic table. This "17" is a big deal! It means a neutral chlorine atom has 17 protons in its nucleus and, crucially for our chat, 17 electrons buzzing around. These are the residents we need to house.
Now, these electrons don't just pick any old spot. They follow some pretty strict rules, like a picky landlord enforcing lease agreements. They fill up "energy levels" and "sublevels," which are like floors and different types of apartments on those floors. Imagine it like this: the first floor is super close to the lobby (the nucleus), so it's the lowest energy and gets filled first. The higher the floor, the more energy the electrons need to get there, and the farther they are from the center.
The Shell Game: Energy Levels
We've got our 17 electrons, and they need homes. The first energy level, often called the 'n=1' shell, can hold a maximum of 2 electrons. Think of it as a cozy studio apartment – only room for two!
So, the first 2 electrons for our chlorine atom? They'll happily settle into this n=1 shell. We denote this as 1s². The '1' is for the first energy level, 's' tells us it's a specific type of orbital (think of it as a shape of room), and the '²' means there are two electrons in that 's' orbital. Easy peasy, right?
After filling up the n=1 shell, we've got 15 electrons left. Where do they go? To the next floor, of course! The second energy level, the 'n=2' shell, is a bit bigger. It can hold up to 8 electrons. This level has different types of rooms: 's' rooms and 'p' rooms. The 's' room can hold 2 electrons, and the three 'p' rooms can hold 6 electrons (2 in each). So, the n=2 shell can totally fit 8 residents!
Our 17 electrons are eager to move in. So, the next 2 electrons will go into the 's' orbital of the n=2 shell, making it 2s². Then, we have 6 more electrons that can fit into the 'p' orbitals of the n=2 shell, making it 2p⁶. So far, we've housed 2 + 2 + 6 = 10 electrons. We're getting there!
The Outer Reaches: The Valence Shell
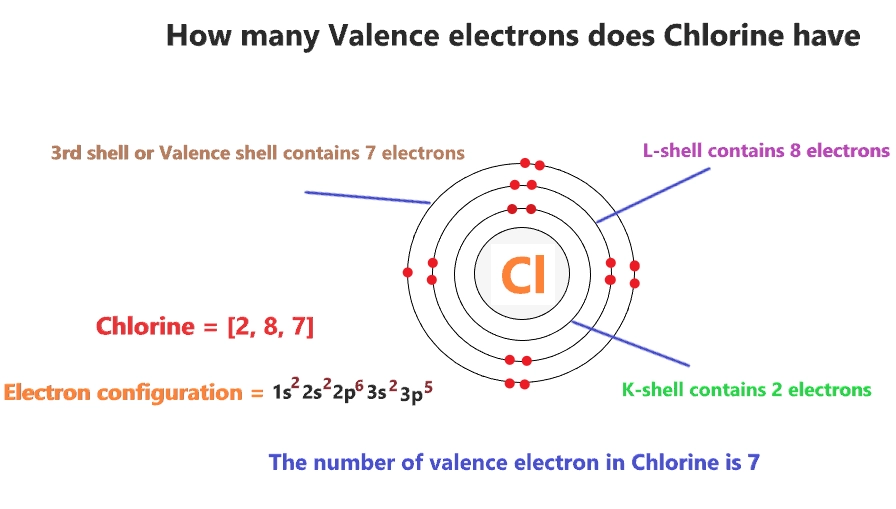
We've used up our first two energy levels. That's 10 electrons accounted for. We still have 17 - 10 = 7 electrons left to place. These remaining electrons are going to our third energy level, the 'n=3' shell. This is where things get really interesting, as this is often the outermost shell, also known as the valence shell.

The n=3 shell is even bigger! It has 's', 'p', and even 'd' type orbitals. The 's' orbital can hold 2 electrons, and the 'p' orbitals can hold 6 electrons. So, the n=3 shell could hold a lot more, but we only have 7 electrons left to distribute.
Following the rules, the next 2 electrons will fill the 's' orbital of the third energy level. That's 3s². Now we have 7 - 2 = 5 electrons remaining.
These last 5 electrons will then go into the 'p' orbitals of the third energy level. So, we get 3p⁵. Remember, 'p' orbitals can hold up to 6, so we're filling it up but not quite to its maximum capacity. Think of it as having a few empty spots in the last apartment – this is super important for how chlorine likes to interact!
Putting It All Together!
So, let's recap the grand tour of chlorine's atomic apartment building. We have:

- Energy Level 1: 1s² (2 electrons)
- Energy Level 2: 2s² 2p⁶ (8 electrons)
- Energy Level 3: 3s² 3p⁵ (7 electrons)
Add them all up: 2 + 8 + 7 = 17 electrons. Boom! We've successfully arranged all of chlorine's electrons.
The full electron configuration for chlorine is therefore 1s²2s²2p⁶3s²3p⁵. Isn't that neat? It’s like a little code that tells us exactly how this atom is built from the inside out.
Why Does This Matter? (Spoiler: It Makes Life Sparkle!)
You might be thinking, "Okay, but why should I care about where electrons hang out?" Well, my friend, this is where the magic happens! The arrangement of electrons, especially those in the outermost valence shell (that's our 3s²3p⁵ group for chlorine), dictates how an atom behaves. It's like knowing someone's social habits – it tells you a lot about how they’ll interact with others.
Chlorine, with its 7 valence electrons, is just one electron short of having a full outer shell. A full outer shell is like the ultimate atomic comfort zone, super stable and happy. Because it's so close to this ideal state, chlorine is very eager to gain that one extra electron. This eagerness is what makes it so reactive!

This is why chlorine loves to bond with other elements. It's like it's always looking for that one perfect dance partner to complete its set. When it bonds with sodium (remember our salt friend, NaCl?), it grabs an electron from sodium, forming an ionic bond. This is a fundamental concept in chemistry, leading to the formation of all sorts of compounds that are essential for life as we know it.
From the bleach that cleans your bathroom to the salt that seasons your food, to the very processes that keep your body functioning, chlorine's electron configuration plays a vital role. It's this seemingly small detail about electron placement that enables the incredible chemical reactions that shape our world. Pretty powerful for something so tiny, right?
Embrace Your Inner Explorer!
So, the next time you see chlorine, or any element on that periodic table, remember its electron configuration. It's not just a string of numbers and letters; it's a story of how atoms organize themselves, how they seek stability, and how they interact to create the universe around us. It’s a tiny peek into the grand, intricate dance of matter.
Learning about electron configurations is like unlocking a secret language. It empowers you to understand the fundamental building blocks of everything. It’s a gateway to appreciating the complexity and beauty of the natural world. And who knows? Maybe understanding this will spark your curiosity to learn even more. The universe is full of amazing secrets waiting to be discovered, and your journey of learning has just begun. Keep asking questions, keep exploring, and never stop being amazed!
