What Is The Difference Between Osmosis And Diffusion Brainly

Okay, so you've probably heard of osmosis and diffusion. Maybe you’re a student trying to ace a test. Or maybe you’re just plain curious about how stuff moves around. Either way, let's break it down. And trust me, it’s way more interesting than it sounds. Think of it like this: life is all about movement. And these two words are basically the secret language of that movement.
So, what’s the big deal? Why do we even care about how molecules decide to mosey on over to a new spot? Well, it’s happening all the time. In your body. In plants. Even in your kitchen!
Let’s start with diffusion. Imagine you’ve got a room. And you spray some really yummy air freshener in one corner. What happens? Boom! Pretty soon, that scent is everywhere, right? Even in the opposite corner where you sprayed it. That’s diffusion in action. Simple. Elegant. A little bit magical.
Diffusion is all about things moving from where there are lots of them to where there are few of them. Think of it as a crowd thinning out. When there are too many people in one place, some will naturally spread out. Molecules are the same way. They’re just chilling, bumping into each other, and eventually, they spread out until they’re evenly distributed. No biggie.
It’s like when you drop a tiny bit of food coloring into a glass of water. At first, it’s all concentrated. But give it a minute, and you’ll see those pretty colors start to swirl and spread. Eventually, the whole glass is the same color. That’s pure diffusion. Those little color molecules are saying, “Hey, too crowded here! Let’s go explore!”
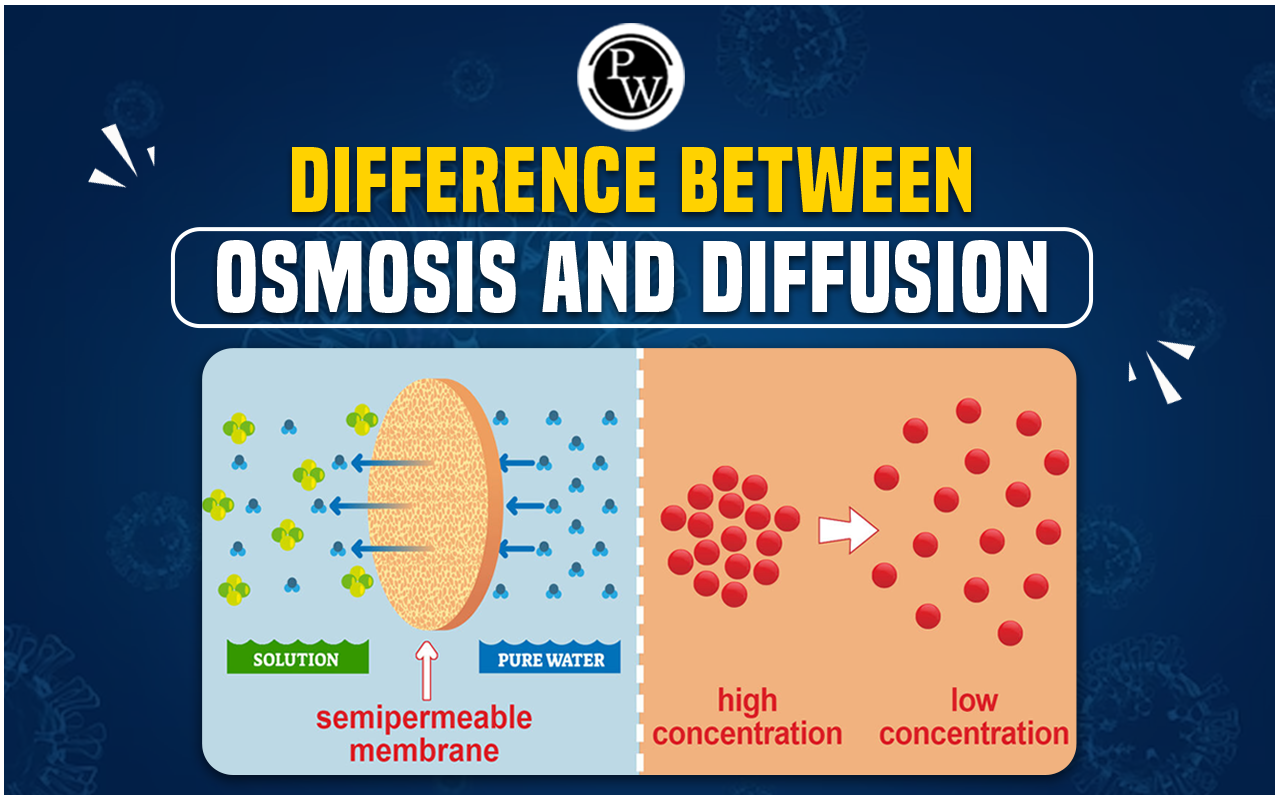
Now, osmosis. This one’s a bit more… particular. Osmosis is a special case of diffusion. It’s diffusion, but with a twist. And that twist involves water. And a semi-permeable membrane. Whoa, fancy words, right? Don’t sweat it.

Think of that semi-permeable membrane like a bouncer at a very exclusive club. It’s got a guest list. And it only lets certain people in. In the case of osmosis, the membrane is picky about what passes through. It’s mostly letting water molecules do their thing. But it’s blocking bigger things, like salt or sugar molecules.
So, osmosis is the movement of water across this special membrane. And where does it go? Just like with diffusion, it goes from an area where there’s more water to an area where there’s less water. But here’s the kicker: it’s often about the concentration of solutes (like salt or sugar) on either side of the membrane. Where there are more solutes (meaning less water), the water will move towards that side.
Confused? Let’s use an analogy. Imagine you have two rooms separated by a wall with tiny doors. These doors are only big enough for tiny bouncy balls (water molecules). But there are also bigger, heavier balls (solute molecules) that can’t get through the doors. Now, in Room A, you have a lot of tiny bouncy balls and only a few heavy balls. In Room B, you have fewer tiny bouncy balls but a whole bunch of heavy balls. Where will the bouncy balls naturally want to go?
They’ll want to go to Room B! Why? Because there’s more space for them there, relative to the heavy stuff. The water is trying to dilute the side with the higher concentration of solutes. It’s trying to balance things out. It’s like water saying, “Whoa, that side’s too packed with… stuff! I’ll go hang out over there and even things out a bit.”

So, the main difference? Diffusion is the general movement of particles from high to low concentration. It can be anything – scent molecules, food coloring, even tiny dust motes dancing in a sunbeam. Osmosis is specifically the movement of water across a semi-permeable membrane, driven by differences in solute concentration.
Here’s a fun thought: Think about your own body. Your cells are basically tiny bags filled with stuff, surrounded by watery fluids. And they have these amazing semi-permeable membranes. Osmosis is happening inside you constantly! It’s how your cells get the water they need and how they get rid of waste. Without osmosis, you’d basically shrivel up or swell up like a balloon. Not ideal.
And what about plants? Plants are masters of osmosis. They soak up water from the soil through their roots using osmosis. This is what makes plants turgid – that firm, upright feeling. When a plant doesn’t get enough water, the cells lose water through osmosis, and the plant wilts. Sad plants. We don’t want sad plants.

Let’s get quirky. Ever put a raisin in water? It plumps up, right? That’s osmosis! The raisin has a higher concentration of sugars inside than the water outside. So, water moves into the raisin. It’s like the raisin is having a little drink. And the opposite? If you put a very salty pretzel on a damp cloth, the cloth might feel a bit drier where the pretzel is. That’s osmosis too! Water is being pulled out of the damp cloth into the salty pretzel to try and dilute those salt crystals.
Think about pickles. Cucumbers are pretty watery. When you brine them, the salty brine pulls water out of the cucumber cells through osmosis, making them shrink and become that firm, delicious pickle texture. It’s a culinary superpower powered by science!
So, to recap, because repetition is key, especially when talking about cool science stuff: * Diffusion: Stuff moving from where there’s a lot to where there’s a little. No special membrane needed. Like a party spreading out. * Osmosis: Specifically water moving across a selective barrier (semi-permeable membrane) from an area of higher water concentration (lower solute concentration) to an area of lower water concentration (higher solute concentration). Think of it as water trying to dilute things.
The key here is that osmosis is a type of diffusion, but it’s a very specific kind. You wouldn’t say diffusion is osmosis because diffusion can involve any particles, and osmosis must involve water and a membrane. It’s like saying all squares are rectangles, but not all rectangles are squares. All osmosis is diffusion, but not all diffusion is osmosis.
Why is this fun to talk about? Because it explains so much about the world around us! From why your skin wrinkles after a long bath (water entering your skin cells) to how your kidneys filter your blood, osmosis and diffusion are the unsung heroes. They’re the invisible forces that keep everything running.
It’s like a silent ballet happening at a microscopic level. Tiny molecules waltzing and diffusing, water molecules elegantly crossing barriers. It’s a constant dance of balance and movement. And understanding it just makes you appreciate the complexity and beauty of nature even more.
So, next time you see a plant perk up after watering, or smell your favorite candle fill a room, you’ll know. It’s not magic. It’s just diffusion and osmosis doing their amazing thing. And that’s pretty darn cool, don’t you think?
It’s a reminder that even the simplest processes are incredibly important. And that the universe is full of fascinating mechanisms, working tirelessly behind the scenes. So go forth, and ponder the marvelous world of molecular movement! It’s a journey worth taking.
