What Is The Difference Between Density And Dispersion

Hey there, curious minds! Ever find yourself wondering about the world around you, the stuff that makes up everything from your morning coffee to the stars in the sky? Today, we're diving into a couple of super fundamental ideas that explain how things are put together: density and dispersion. Now, these might sound a little science-y, but trust me, they're actually pretty darn cool and surprisingly easy to get your head around. Think of it as peeking behind the curtain of how things are arranged and packed in our universe!
So, what’s the big deal? Well, understanding density and dispersion helps us make sense of why a tiny pebble sinks like a stone while a giant log floats, or why a well-organized bookshelf looks so different from a chaotic pile of books. It’s all about how much "stuff" is packed into a certain space and how that "stuff" is spread out. Ready to get a little nerdy, but in a totally chill way?
Density: How Much Stuff Is Crammed In?
Let’s start with density. Imagine you have two boxes, right? Both boxes are the exact same size – let's say, a cubic foot. Now, in one box, you meticulously pack in as many tiny, super-heavy lead balls as you possibly can. In the other box, you toss in a bunch of fluffy feathers. Which box do you think is going to feel heavier?
Of course, it's the box with the lead balls! Even though both boxes are the same size, the lead balls pack a much bigger punch when it comes to mass. This is the essence of density. Density is basically a measure of how much mass is squeezed into a given volume.
Think of it like this: If you're at a crowded concert, and everyone is really tightly packed together, that crowd is dense. If you’re in a huge park with only a few people scattered around, that’s not very dense at all. The amount of "people" (mass) in the "space" (volume) is much lower in the park.
So, in scientific terms, we often say density is mass per unit volume. That’s why lead is so dense – a small piece of lead has a lot of mass. Air, on the other hand, is not very dense; a big volume of air has relatively little mass.

Why is this interesting? Well, density is what explains so many everyday phenomena! It's why ships made of steel can float. You might be thinking, "Wait a minute, steel is way denser than water!" And you're right, solid steel is denser than water. But a ship isn't just a solid block of steel. It's designed with a hollow hull, filled mostly with air. The overall density of the ship, including all that air, becomes less than the density of water. So, it floats! Pretty clever, huh?
It's also why you can have a helium balloon float up into the sky. Helium is less dense than the surrounding air, so it’s like a super-light bubble rising through a denser medium. Conversely, a rock sinks because it's much denser than water. It’s all about that mass-to-volume ratio!
Density: The "Heaviness" of Stuff in a Space
Let's solidify this with another fun comparison. Imagine you’re baking. You have a cup of flour and a cup of sugar. They take up the same amount of space, right? But if you weigh them, they’ll have different masses. Flour is generally less dense than sugar, meaning sugar packs more "stuff" into that same cup.
So, when we talk about density, we're essentially asking: "How tightly packed is this material?" Is it like a can of sardines (very dense), or is it like a bag of popcorn (less dense, with lots of air pockets)? It’s a fundamental property of materials that tells us a lot about their behavior.

Dispersion: How Spread Out Is It?
Now, let's shift gears to dispersion. While density is about how much stuff is in a space, dispersion is about how that stuff is arranged or spread out within that space. Think of it as the pattern or distribution of particles.
Imagine you're looking at a field of flowers. In one field, the flowers might be planted in perfect, straight rows, equally spaced apart. This is a very ordered or non-dispersed arrangement. In another field, the flowers might have sprung up randomly, clustered together in some spots and leaving other areas bare. This is a more dispersed or random arrangement.
In physics and chemistry, dispersion often refers to how particles (like atoms or molecules) are spread out in a gas, liquid, or solid, or how waves are spread out in a medium. It’s about the spacing and arrangement of these components.

Let's use another analogy. Think about the crowd at that concert again. If everyone is packed shoulder-to-shoulder, that's high density, and the dispersion is very uniform (everyone is right next to someone else). Now, imagine that same number of people at the park. They might be spread out over a huge area, some in small groups, some alone. The density of the crowd is much lower, and the dispersion is much more varied – it’s scattered.
Another cool example is in light! When white light passes through a prism, it splits into a rainbow of colors. This happens because different colors of light (which are essentially waves of different wavelengths) travel at slightly different speeds through the glass. This spreading out of light by wavelength is called dispersion. It’s what allows us to see a beautiful spectrum!
Dispersion: The "Scatteredness" of Stuff
So, we can have materials that are dense but have a uniform dispersion (like a solid block of metal where atoms are tightly packed in a regular lattice). Or, we could have something that's not very dense but has a very scattered dispersion (like a nebula in space, which is mostly empty space with gas and dust spread thinly across vast distances).
Consider a classroom. If all the students are sitting in their assigned desks in neat rows, that's a very ordered or low dispersion arrangement in terms of seating. If they're all milling around, talking in small groups, or playing in different corners of the room, that's a much more dispersed arrangement. The overall number of students in the room (density of students) might be the same, but how they are spread out is completely different.
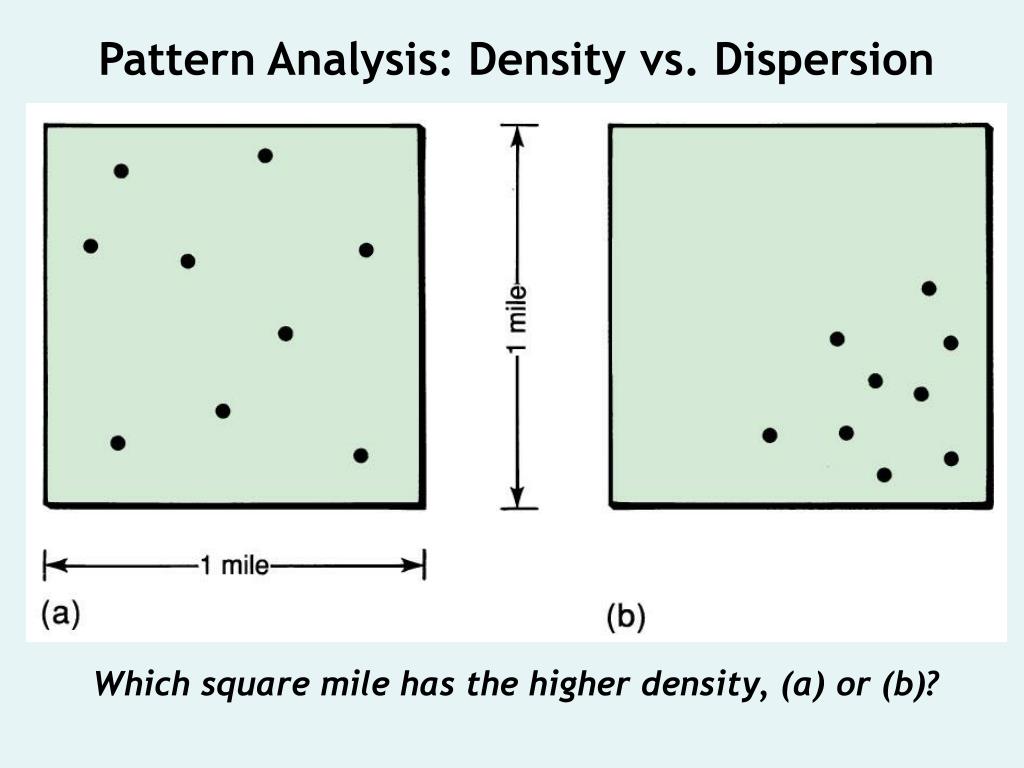
Dispersion also plays a role in how substances mix. If you put a drop of ink into water, the ink molecules will gradually spread out and mix with the water molecules. This process of spreading and mixing is a form of dispersion.
Putting It All Together: The Dynamic Duo
So, to recap: density is about how much stuff is packed into a space, and dispersion is about how that stuff is arranged or spread out within that space. They’re like two sides of the same coin, describing different aspects of how matter is organized.
You can have high density with low dispersion (a tightly packed, ordered structure) or low density with high dispersion (a spread-out, scattered arrangement). And there are all sorts of combinations in between!
Why is this stuff cool? Because it helps us understand the fundamental building blocks of our universe and how they interact. From the microscopic world of atoms to the macroscopic world of galaxies, density and dispersion are at play, shaping everything we see and experience. It's a simple concept, but it unlocks a deeper appreciation for the intricate order (and sometimes, beautiful chaos) of nature. So next time you see something float, sink, or spread out, you'll have a little more insight into the fascinating principles behind it!
