What Is The Definition Of A Lewis Base Brainly
Hey there, science curious folks! Ever stumbled upon a term like "Lewis base" and wondered what in the world it means? It sounds a bit like something out of a sci-fi movie, right? Well, buckle up, because we're about to demystify this cool chemical concept in a way that's totally chill and, dare I say, even a little bit fun.
So, what exactly is a Lewis base? Forget those stuffy textbook definitions for a second. Think of it like this: in the grand orchestra of chemistry, there are always players looking to share. And a Lewis base? It's basically the ultimate generous sharer when it comes to electrons.
The Electron Exchange Game
You see, atoms and molecules are constantly interacting, and a lot of that interaction boils down to how they handle their electrons. Electrons are like the tiny, energetic currency of the chemical world. They're what hold things together, what make reactions happen, and generally what make the universe… well, work.
Now, not everyone is equally keen on holding onto their electrons. Some atoms or molecules are a bit like that friend who always has extra snacks to offer. They've got a little something extra, a surplus of electrons that they're perfectly happy to lend out.
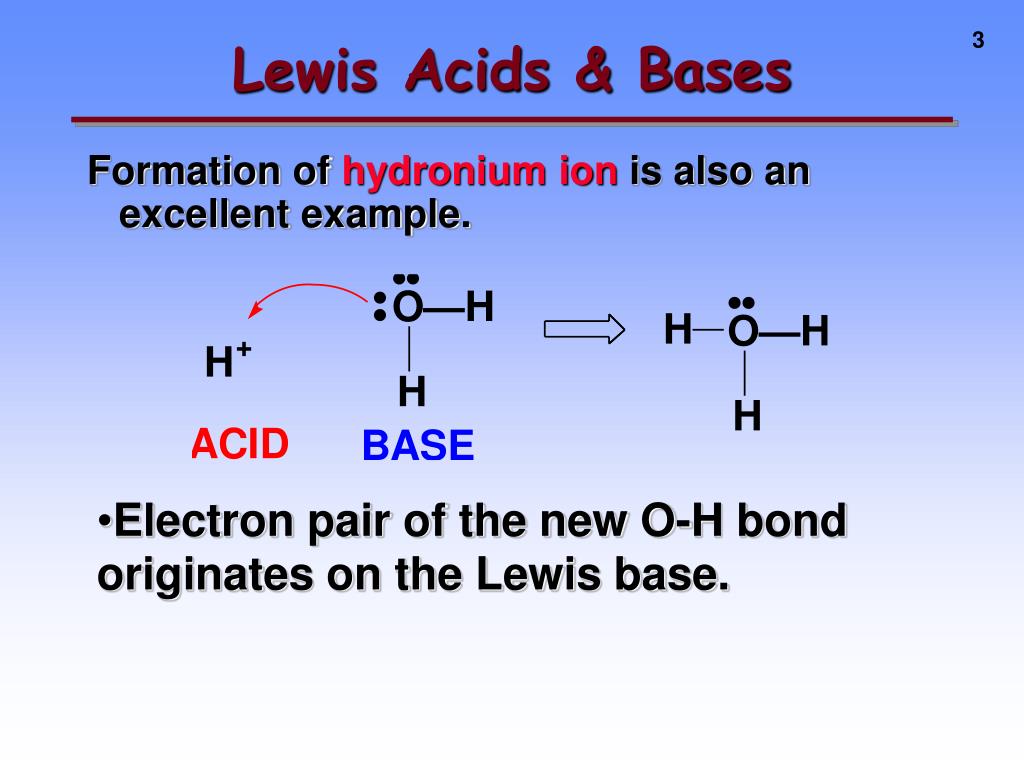
That, my friends, is the essence of a Lewis base. It's a substance that has a pair of electrons it can donate to another chemical species. Think of it as having a little electronic purse that it can open up and say, "Here, you need some? Take 'em!"
It's All About the Pairs!
It's not just about having any electrons, though. It's specifically about having a lone pair of electrons. Imagine a cozy little couple, two electrons that aren't busy bonding with anything else. These are the electrons a Lewis base is ready to share. They're not tied down; they're free agents, looking for a good cause!

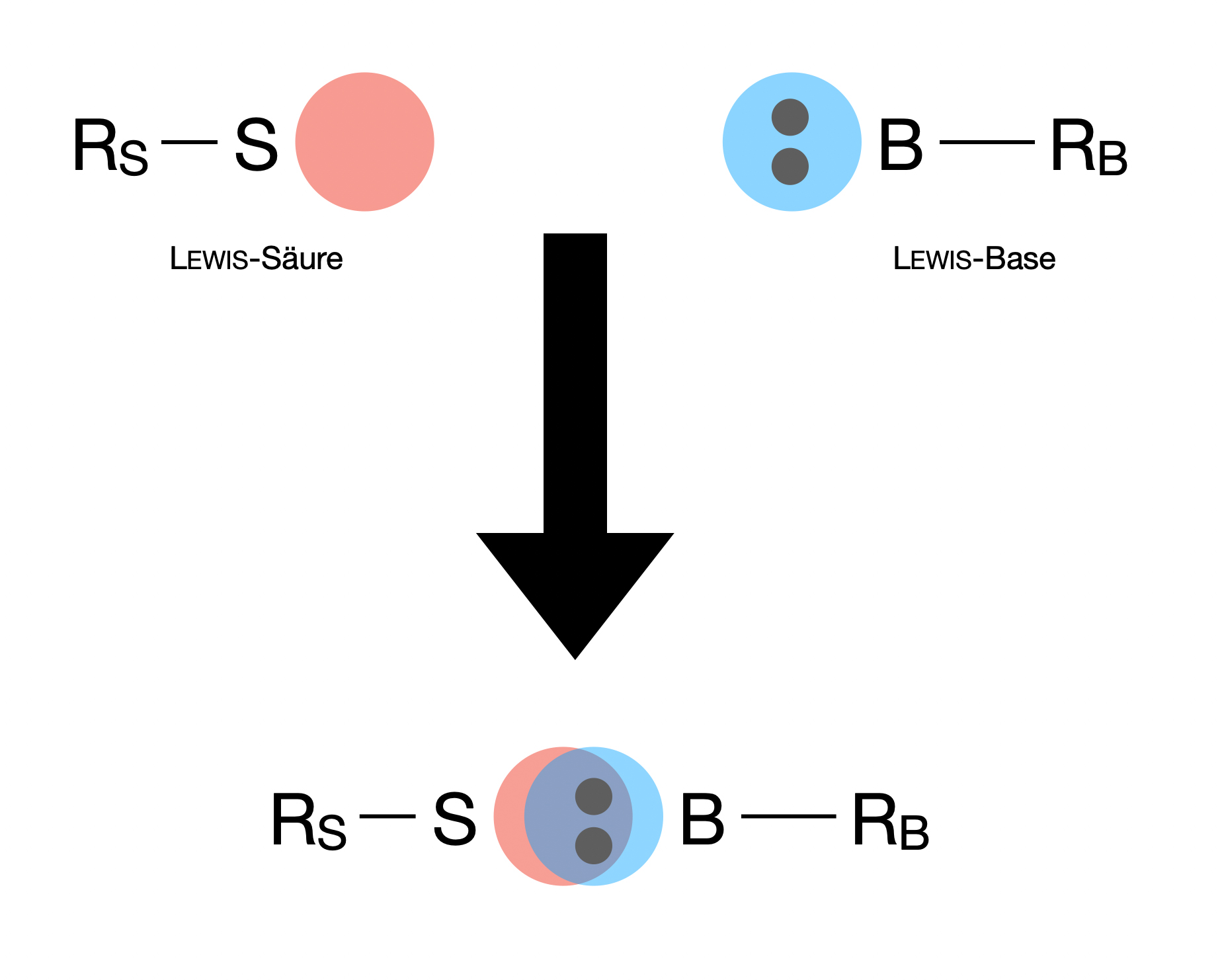
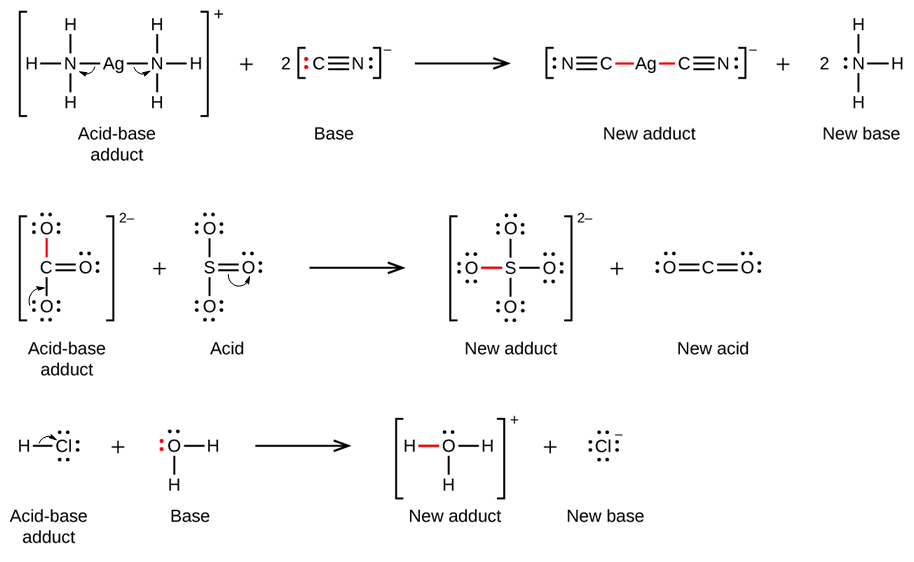
So, why is this electron-sharing thing so important? Because it's the foundation of a whole category of chemical reactions. When a Lewis base finds a willing partner to accept its donated electron pair, a special kind of bond is formed. This isn't your typical covalent bond where electrons are shared equally between two atoms. This is a coordinate covalent bond, also known as a dative bond. It's a one-way street for electrons, with the Lewis base being the generous donor.
Meet the Other Half of the Story: The Lewis Acid
You can't have a donor without a receiver, right? And that's where the Lewis acid comes in. If a Lewis base is the sharer, a Lewis acid is the one who's a bit peckish for electrons. It's a substance that can accept a pair of electrons. Think of it as the person with an empty plate, eagerly waiting for that delicious donated electron pair.
So, when a Lewis base and a Lewis acid meet, it's a match made in chemical heaven! The base offers its lone pair, and the acid gratefully accepts it, forming that special coordinate covalent bond. It's a beautiful dance of electron redistribution.
Examples to Make it Click
Let's sprinkle in some real-world (or at least lab-world) examples to make this concept really stick. You've probably heard of ammonia, right? That stuff with the… distinct smell?
Ammonia (NH3) is a classic Lewis base. Why? Because the nitrogen atom in ammonia has a lone pair of electrons just itching to be shared. It's like Nitrogen's saying, "Hey, I've got this extra pair of electrons chilling here, anyone need 'em?"
Now, what could ammonia want to react with? It might bump into something like a boron trifluoride molecule (BF3). Boron in BF3 is a bit electron-deficient, meaning it's looking for some electron love. So, the ammonia molecule, with its generous electron pair, can donate those electrons to the boron in BF3, forming a stable compound. Voila! A Lewis base and a Lewis acid teaming up.
Another common example of a Lewis base is water (H2O). The oxygen atom in water also has those handy lone pairs. You see water doing this kind of electron-sharing dance all the time in chemical reactions, especially when it's acting as a solvent.
Why Should We Care? It's Pretty Cool!
Okay, so we've got these electron-sharing buddies. Why is it so interesting? Well, understanding Lewis acids and bases is fundamental to so many areas of chemistry.
It helps us predict how molecules will react with each other. It's like having a cheat sheet for chemical interactions. If you know which molecule is likely to be a base (electron donor) and which is likely to be an acid (electron acceptor), you can make pretty good guesses about what kind of reaction will occur.
This concept is at play in everything from how our bodies digest food to how batteries generate electricity. It's involved in the catalysts that speed up industrial processes and in the way drugs interact with our cells.
Think of it Like This…
Imagine you're at a party. Some people are holding onto their snacks really tightly (like molecules that don't readily donate electrons). Others are super chill and are happy to pass around a bowl of chips (like Lewis bases). And then there are those who look a little hungry and would appreciate a bite (like Lewis acids).
When the hungry person (Lewis acid) sees the person with the chips (Lewis base), they might politely ask for some, and a connection is made. It's a simple exchange that leads to something new.

Or think about magnets. You have a north pole and a south pole. They're attracted to each other, right? A Lewis acid and base interaction is a bit like that, but instead of magnetic attraction, it's electron attraction. The electron-rich base is drawn to the electron-poor acid.
Beyond the Basics: It Gets Even Cooler
The beauty of the Lewis definition is that it's broader than older definitions of acids and bases. For example, the traditional Brønsted-Lowry definition focuses on the transfer of protons (H+ ions). But the Lewis definition expands our view to include many more substances that don't necessarily involve protons but still exhibit acidic or basic behavior.
This broader perspective allows chemists to understand a wider range of reactions and chemical phenomena. It's like upgrading from a black-and-white TV to a high-definition, surround-sound experience – you see and understand so much more!
So, the next time you hear the term "Lewis base," don't get intimidated. Just remember that it's simply a chemical species that's feeling generous with its electrons, ready to share a lone pair and make some cool new chemical connections. It’s a fundamental concept that underpins a massive amount of chemical activity, and understanding it opens up a whole new world of chemical understanding. Pretty neat, huh?
