What Is The Correct Structure For 1 4 Dimethylbenzene
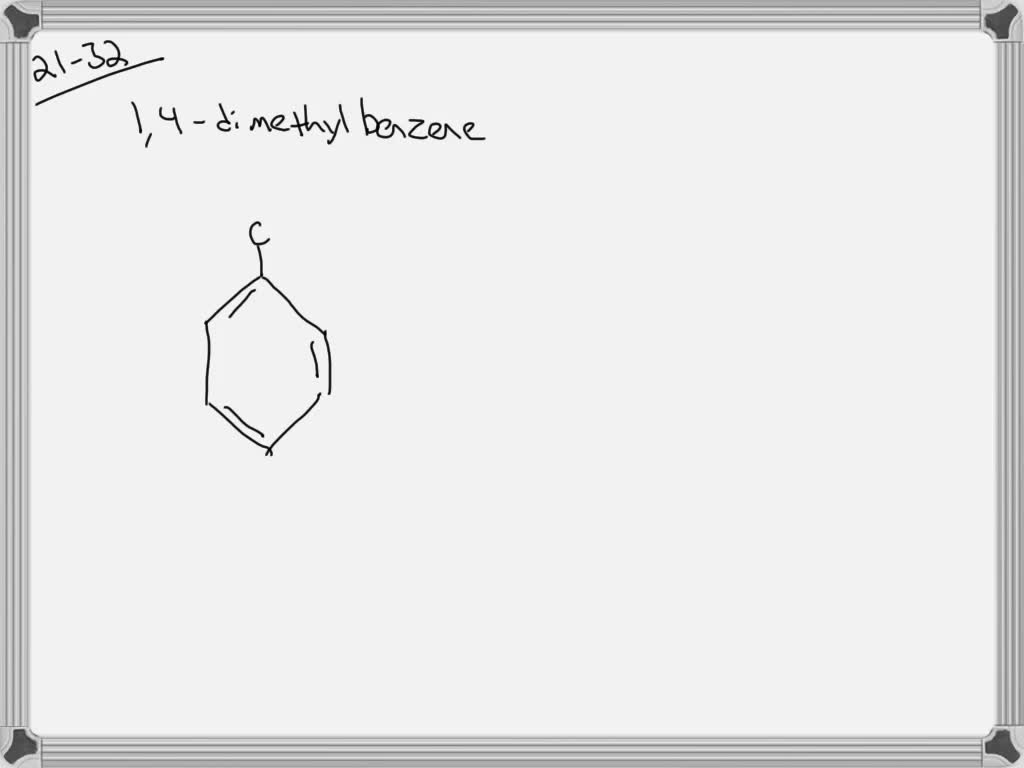
Hey there, science curious folks! Ever stumbled upon a chemical name that sounds a bit like a secret code? Today, we're going to crack one of those codes: 1,4-dimethylbenzene. Sounds fancy, right? But really, it's just a molecule chilling out in the world, and understanding its structure is actually pretty neat. Think of it like figuring out the layout of a really cool LEGO set – once you know where the bricks go, it all makes sense!
So, what exactly is 1,4-dimethylbenzene? Well, before we get to the "1,4" and the "dimethylbenzene" part, let's break down the "benzene" itself. Imagine a ring, like a tiny, perfect hexagon. But instead of sides made of solid material, this hexagon is made of six carbon atoms. And these carbon atoms are holding hands with six hydrogen atoms, sort of like a perfectly symmetrical dance party.
This benzene ring is a fundamental building block in a whole lot of organic chemistry. It's super stable, like a well-built structure that can withstand a lot. Think of it as the fundamental shape of a comfy armchair – everything else gets built around it.
The Benzene Ring: A Stable Foundation
This six-carbon ring, with its alternating single and double bonds (though it's a bit more complicated than that, with electrons kind of buzzing around everywhere), gives benzene its unique personality. It's not your average carbon chain. This ring structure is what makes it so stable and allows it to be the basis for so many other interesting molecules.
Now, let's talk about the "dimethyl" part. What does that add to our hexagonal party? "Di" means two, and "methyl" refers to a methyl group. A methyl group is basically a carbon atom with three hydrogen atoms attached – CH3. It's like a little, tiny branch that can attach to our main benzene ring.
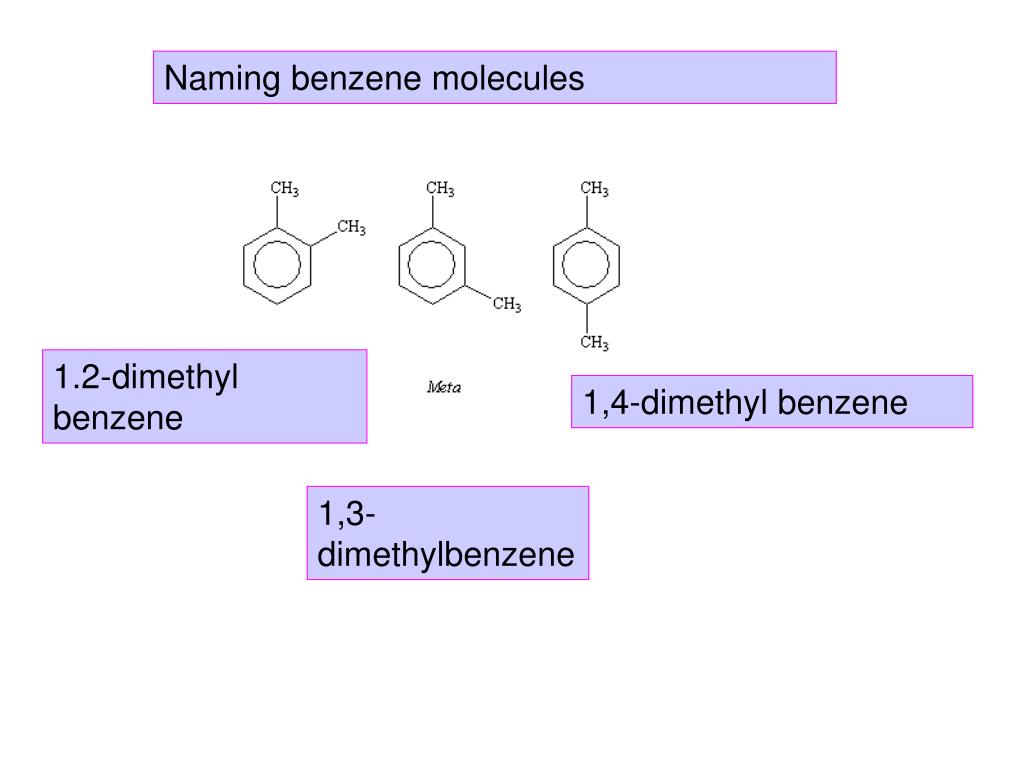
So, we've got our benzene ring, and we're going to stick two of these methyl groups onto it. Easy peasy, right? But where we stick them makes all the difference. And this is where the "1,4" comes in. Think of the carbon atoms in the benzene ring as being numbered 1 through 6, going around the ring.
The Crucial "1,4": Location, Location, Location!
The numbers "1,4" tell us which carbon atoms on the benzene ring these two methyl groups have decided to call home. So, if we number the carbons in our benzene ring, say, starting at the top and going clockwise, the "1,4" means one methyl group is attached to carbon number 1, and the other methyl group is attached to carbon number 4. It's like having two friends meeting up at specific spots on a circular race track.
This specific arrangement – one methyl on carbon 1 and another on carbon 4 – is what defines 1,4-dimethylbenzene. It's also commonly known by a much cooler name: paraxylene. Yep, sounds a bit more rockstar, doesn't it? "Para" is a prefix that chemists use to describe this "opposite sides" arrangement on a benzene ring. So, para-xylene is the friendly nickname.
Imagine our benzene ring as a clock face. If you put a methyl group at the 12 o'clock position, in 1,4-dimethylbenzene, the other methyl group would be directly opposite at the 6 o'clock position. They are on opposite sides of the ring. This symmetry is a big part of why it behaves the way it does.
Why This Structure Matters
So, why should we care about where these methyl groups are hanging out? Well, the position of these substituents (that's the fancy word for groups attached to the main molecule) significantly affects the molecule's properties. It's like how the arrangement of furniture in a room can change how you feel when you're in it. The same pieces of furniture, but a different layout, makes a whole different vibe.
If the methyl groups were attached to adjacent carbons (like 1,2-dimethylbenzene, or ortho-xylene), or separated by one carbon (like 1,3-dimethylbenzene, or meta-xylene), the molecule would have slightly different physical and chemical characteristics. Think about it: a molecule is a 3D thing, and even small changes in the positioning of its parts can affect how it interacts with other molecules, how it melts, how it boils, and how it reacts.
For 1,4-dimethylbenzene (paraxylene), this specific arrangement makes it particularly useful. It's a key ingredient in the production of something called terephthalic acid. And that stuff? It's a super important precursor for making PET plastic. You know, the stuff your water bottles and lots of other everyday plastic items are made from. So, the simple act of placing two methyl groups on opposite sides of a benzene ring has a HUGE impact on the materials we use every day!
A Family of Xylenes
It's also interesting to note that 1,4-dimethylbenzene isn't the only game in town when it comes to dimethylbenzenes. As we mentioned, there are its siblings: ortho-xylene (1,2-dimethylbenzene) and meta-xylene (1,3-dimethylbenzene). They all share the same basic ingredients – a benzene ring and two methyl groups – but their different arrangements lead to different personalities and uses.
It’s a bit like a family of three siblings. They look similar, share the same parents (the benzene ring and the methyl groups), but they each have their own quirks and talents. One might be super athletic, another a brilliant artist, and the third a fantastic storyteller. Similarly, ortho-, meta-, and paraxylene, despite their close chemical relationship, have distinct properties and applications.
The Structure Visualized
To really get it, it helps to visualize. Imagine our benzene ring as a flat, hexagonal plate. Now, take two little ball-and-stick models of a methyl group. In 1,4-dimethylbenzene, you'd place one methyl group sticking straight up from the top carbon, and the other sticking straight up from the bottom carbon. They are perfectly aligned across the center of the plate.
This symmetry in 1,4-dimethylbenzene is really quite elegant. It's like a perfectly balanced scale. This structural feature is what makes it so valuable in industrial processes. It's efficient, predictable, and allows for targeted chemical reactions.
So, the next time you hear "1,4-dimethylbenzene" or "paraxylene," don't let it intimidate you. It's just a molecule with a specific, symmetrical structure that makes it incredibly useful. It's a testament to how tiny, fundamental building blocks can lead to the vast array of materials that shape our world. Pretty cool, huh? It’s a reminder that even seemingly complex names often point to elegant and understandable structures when you break them down.
