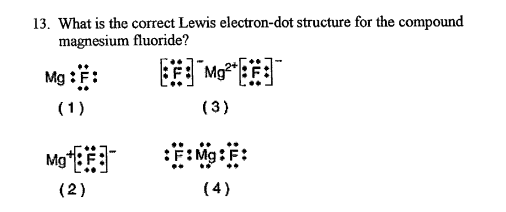
What Is The Correct Lewis Electron-dot Diagram For Magnesium Fluoride

Get ready for a dazzling dive into the wonderfully wacky world of chemical bonding! Today, we’re going to unravel the mystery behind magnesium fluoride and its super cool Lewis electron-dot diagram. Think of it like a tiny, atomic matchmaking service, where elements find their perfect partners to achieve ultimate stability. And oh boy, does magnesium fluoride make a fantastic couple!
Imagine atoms as tiny people, each with a little collection of electrons zipping around their center. These electrons are like their prized possessions, and they really want to have just the right number to feel complete and happy. It’s a bit like having a favorite toy collection – you don’t want too few, and you definitely don’t want too many!
Our star of the show today is magnesium, a metal that’s lighter than air (okay, not really, but it feels that way when you see how it behaves!) and loves to share. Magnesium, bless its metallic heart, has two extra electrons that it’s just itching to give away. It’s like that friend who always has an extra cookie to offer – generous to a fault!
And then we have fluorine, a halogen that’s a bit of a drama queen. Fluorine absolutely adores electrons. It’s constantly looking to snatch up any electron it can find to fill its outer shell. It’s the ultimate electron hoarder, but in the best possible way for chemical stability!
So, what happens when these two energetic personalities meet? It’s a match made in ionic heaven! Magnesium, with its two spare electrons, spots fluorine, who is practically begging for electrons. It’s like a perfectly orchestrated dance of give and take.
The Grand Electron Giveaway!
Magnesium, that generous soul, decides to donate one of its precious electrons to one fluorine atom. Poof! That electron is now happily nestled with the first fluorine. But wait, magnesium has another electron it wants to get rid of!

And guess who’s still looking for more electron friends? Yep, another fluorine atom! So, magnesium performs its electron-giving magic again, bestowing its second spare electron upon the second fluorine atom. It’s a double dose of electron generosity!
Now, here’s where the magic of Lewis diagrams comes in. We represent these electron exchanges using simple dots. Each dot is a little electron, and we draw them around the element's symbol. It's like drawing little smiley faces on happy electrons!
The Magnesium Moment
For magnesium, which started with two electrons in its outermost shell, it’s now feeling a little… empty on the outside, in a good way! It’s like it shed its outer coat to reveal a perfectly complete inner layer. We represent this by showing magnesium with a positive charge, usually written as Mg2+. This charge signifies that it has lost two negatively charged electrons, making it positively charged overall.
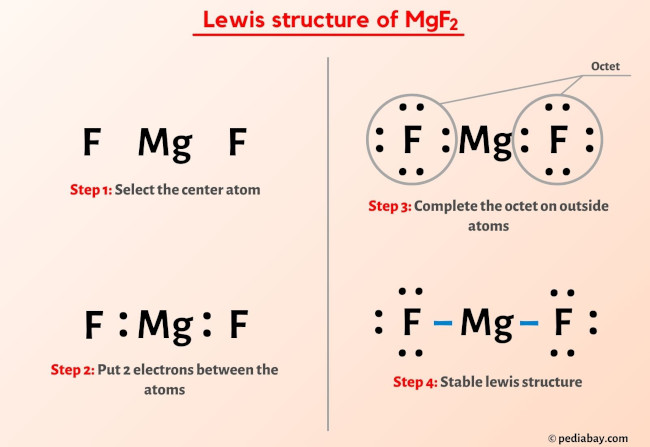
There are no dots around the magnesium symbol in its final ionic form in the Lewis diagram because all its valence electrons have been transferred. It's achieved that blissful state of having a full inner shell, like finally finding the perfect hiding spot for all your treasures!
The Fluorine Fiesta!
Now, each fluorine atom, which started with seven electrons in its outer shell, is absolutely thrilled. One fluorine atom has gained one electron, bringing its total to eight. The other fluorine atom has also gained one electron, bringing its total to eight. They’ve both achieved the coveted octet rule – eight electrons in their outer shell, which is like winning the lottery in the atomic world!
We show this by drawing six dots (representing the original seven valence electrons plus the one gained electron) around each fluorine symbol, arranged in pairs. And because they’ve gained a negatively charged electron, each fluorine atom now carries a negative charge, written as F-. So, we have two of these very happy, negatively charged fluorine atoms!
Putting It All Together: The Iconic Trio!
So, the complete Lewis electron-dot diagram for magnesium fluoride shows one magnesium ion (Mg2+) and two fluoride ions (F-). The magnesium ion is represented without any dots, just its symbol and the positive charge. Each fluoride ion is surrounded by eight dots (four pairs), representing its full outer shell, and its negative charge.
Imagine it like a proud parent (magnesium) who has helped two of their children (fluorine atoms) find their perfect matching sets of toys. The parent is happy and stable, and the children are also incredibly content with their full toy collections. It's a beautiful symphony of ionic attraction, where the positive magnesium is strongly attracted to the negatively charged fluorines, holding them all together like superglue!
It’s this powerful attraction between the positively charged magnesium and the negatively charged fluorines that makes magnesium fluoride such a stable compound. They've achieved their electron dreams, and they're sticking together forever (or at least until some other chemical reaction comes along!). It’s a fantastic illustration of how atoms strive for stability by exchanging electrons.

So, the next time you encounter magnesium fluoride, remember the epic electron journey! Remember the generous magnesium and the electron-adoring fluorines. Their Lewis diagram is a tiny, yet powerful, testament to the incredible and often humorous quest for electron happiness that drives the entire universe of chemistry. Isn’t science just the coolest?
It’s a story of giving, receiving, and ultimately, achieving a state of perfect electron harmony. The diagram isn’t just a drawing; it’s a snapshot of atoms finding their happy place. And that, my friends, is a cause for celebration in the world of chemistry!
