What Is The Correct Formula For Iron Iii Sulfide

Ever found yourself staring at a mysterious little powder or a shiny mineral and wondering, "What is this stuff, chemically speaking?" It's a question that pops up more often than you'd think, especially when you start delving into the fascinating world of chemistry. Today, we're going to tackle one such mystery: Iron(III) Sulfide. Now, don't let the name scare you off! We're going to break it down in a super chill way, no intimidating equations or complicated jargon. Think of this as a casual chat over coffee, exploring a bit of chemical curiosity.
So, what's the deal with Iron(III) Sulfide? You might be picturing a big, burly iron atom teaming up with a bunch of sulfur atoms. And you'd be partly right! But the "III" in there is super important. It's like a secret code that tells us how many buddies the iron atom has brought to the party, and what kind of party it is. This is where we start to uncover the correct formula, and trust me, it's pretty neat.
Let's Talk About the "III"
In chemistry, we often use numbers in parentheses after the name of an element, like Iron(III). This isn't just for show! This little Roman numeral is a superhero, telling us about the charge of that particular iron atom. Iron is a bit of a chameleon; it can exist in different forms, or "oxidation states," depending on who it's hanging out with. It's like how you might act differently with your best friends versus with your grandparents. Iron can be Iron(II) (with a +2 charge) or Iron(III) (with a +3 charge).
When we say "Iron(III) Sulfide," that Roman numeral "III" tells us that the iron atom involved has a positive charge of +3. This is a crucial piece of information, like knowing the main character's personality before diving into a book. It sets the stage for how the iron will interact with other elements.
And What About Sulfur?
Now, let's bring in the sulfur. Sulfur is pretty common in the chemical world, often found in volcanic gases or even in that eggy smell you sometimes get (don't worry, not all sulfur smells like that!). Sulfur typically likes to gain electrons, and when it does, it forms an ion with a negative charge of -2. Think of it as sulfur being a bit of a grabber, always looking to pick up a couple of electrons to feel complete.
So, we have our Iron(III) with its +3 charge, and Sulfur with its -2 charge. How do these two become a stable compound? This is where the balancing act of chemistry comes in. Nature, and chemistry, loves balance. It wants everything to be neutral, like a perfectly weighted scale. No overall positive or negative charge hanging around.
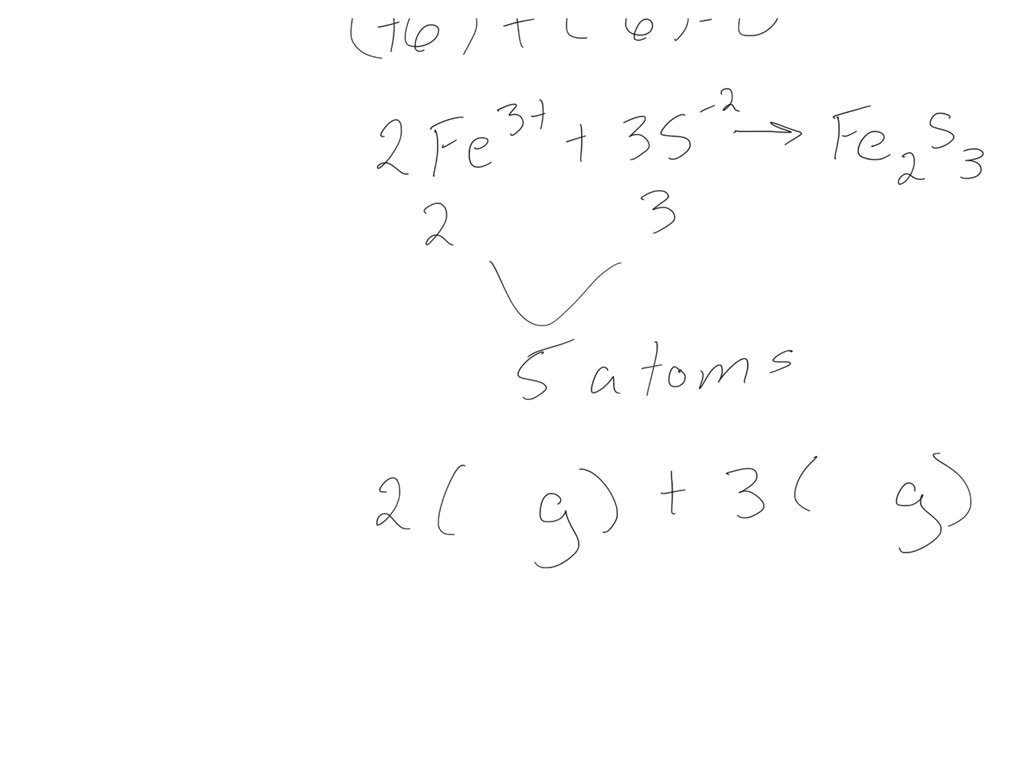
The Balancing Act: Finding the Right Formula
This is where the magic happens, and we find the correct formula for Iron(III) Sulfide. We need to figure out how many iron atoms (with their +3 charge) and how many sulfur atoms (with their -2 charge) we need to make the charges cancel out and create a neutral compound. It’s like trying to fit puzzle pieces together until the picture is complete and harmonious.
If we have one Iron(III) ion (+3) and one sulfide ion (-2), we have a net charge of +1 (+3 + -2 = +1). That's not balanced! If we try two Iron(III) ions (+3 + +3 = +6) and one sulfide ion (-2), we still have a positive charge of +4. Not quite there.
What if we try this: two Iron(III) ions and three sulfide ions?

Let's do the math (the fun, simple kind!):
- Two Iron(III) ions: 2 * (+3) = +6
- Three sulfide ions: 3 * (-2) = -6
And what do we get when we add those up? +6 + -6 = 0! Bingo! We've reached neutrality. The charges have perfectly balanced each other out. This is the essence of ionic bonding, where oppositely charged ions come together to form a stable compound.
The Reveal: Fe₂S₃
So, after our little chemical balancing act, we’ve arrived at the correct formula for Iron(III) Sulfide: Fe₂S₃. This means that for every two iron atoms that have a +3 charge, there are three sulfur atoms that have a -2 charge. It’s a beautiful little dance of electrons and charges!
You might see this compound represented in different ways depending on the context, but Fe₂S₃ is the standard empirical formula. It tells us the simplest whole-number ratio of atoms in the compound. It’s like the most efficient recipe for creating this particular chemical entity.
Why Is This Cool?
Beyond just knowing the formula, understanding how we get there is the really interesting part! It shows us the underlying order and logic in the universe. Even the tiniest building blocks of matter follow these rules of attraction and balance. It's like discovering the hidden rules of a game that everything plays by.
Iron(III) Sulfide itself isn't as famous as, say, rust (which is another iron compound, but a much more complicated mixture!). However, iron sulfides do occur naturally. For example, pyrrhotite is a mineral that's essentially an iron sulfide, though its exact formula can vary a bit. It's often found in meteorites, which is pretty mind-blowing when you think about it – this stuff has traveled through space!
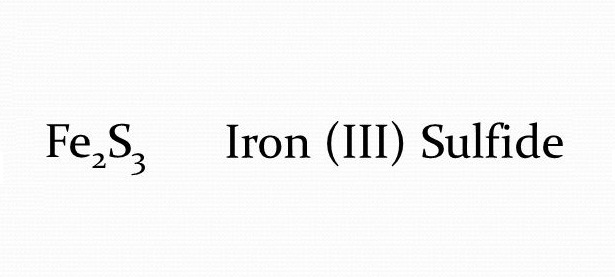
Think of it this way: when you see Fe₂S₃, it’s not just a jumble of letters and numbers. It’s a tiny blueprint for a specific arrangement of atoms that has unique properties. These properties dictate how it will behave in the world, whether it will be magnetic, what color it will be, and how it might react with other substances.
A World of Possibilities
The world of chemistry is full of these fascinating formulas and the compounds they represent. Each one is a little story, a testament to the way different elements interact and combine. Understanding the formula for something like Iron(III) Sulfide gives you a glimpse into that intricate dance. It’s like learning a new language, the language of atoms and molecules. And with every new formula you decipher, you unlock a little more of the universe’s secrets.
So, the next time you encounter a chemical name or a formula, don't shy away from it. Lean in with curiosity! Ask those simple questions. Because hidden within those seemingly complex labels are elegant solutions and the fundamental building blocks of everything around us. And that, in itself, is pretty darn cool, wouldn't you say?
