What Is The Correct Formula For Barium Chlorate
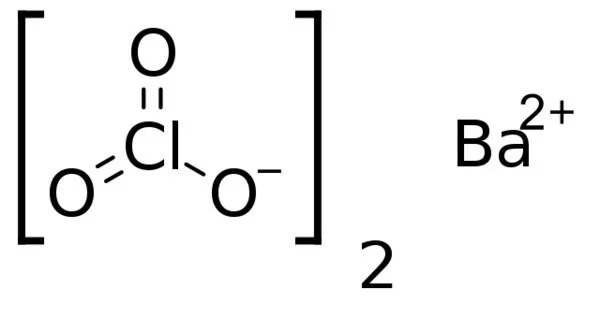
Hey there, curious minds and science enthusiasts! Ever found yourself wondering about the building blocks of the world around us, or perhaps even dabbling in a little bit of home chemistry? There's a certain magic to understanding how different elements come together to create new substances, and today, we're diving into one such compound that might just spark your interest: Barium Chlorate.
Now, you might be thinking, "Barium Chlorate? What's that got to do with me?" Well, while you're probably not mixing up a batch in your kitchen sink (and please, don't!), understanding its formula and properties is key to appreciating its role in various industries and even in some surprisingly common applications. Think about it – from the vibrant colors of fireworks to the precise workings of certain medical tests, chemistry is the silent architect of much of our modern life. Barium chlorate, in its own unique way, contributes to this fascinating world.
So, what's the big secret? What's the correct formula for Barium Chlorate? Drumroll please… it’s Ba(ClO₃)₂. Let's break that down a little. You've got Barium, represented by the symbol Ba. Then you have the chlorate ion, which is a polyatomic ion made up of one chlorine atom (Cl) and three oxygen atoms (O). This chlorate ion carries a charge of -1, and Barium, being an alkaline earth metal, typically forms a +2 ion. To balance these charges and create a neutral compound, you need two chlorate ions for every one barium ion. Hence, the (ClO₃)₂. The parentheses are important because they tell us that the subscript '2' applies to both the chlorine and the oxygen atoms within the chlorate ion. Pretty neat, right?
The benefits and purpose of understanding this formula are more practical than you might initially think. For chemists and material scientists, it's the fundamental blueprint for creating and working with this compound. Barium chlorate is a powerful oxidizing agent, meaning it readily gives up oxygen to other substances. This property makes it incredibly useful in a variety of applications. For instance, it's a crucial ingredient in the manufacture of pyrotechnics, contributing to those brilliant flashes and colors you see in fireworks displays. Imagine the visual spectacle – that’s chemistry at play!

Beyond the dazzling lights, barium chlorate also finds its way into other areas. It can be used in certain types of analytical chemistry and in the production of other barium compounds. While less common for everyday consumers, these specialized uses highlight the compound's versatility. Think of it as a specialized tool in the vast toolbox of chemistry that enables innovation and progress in different fields.
Now, how can you "enjoy" learning about barium chlorate more effectively? Well, the best way is through curiosity and responsible learning. If you're fascinated by this topic, consider exploring educational resources. Look for reputable science websites, educational videos, or even introductory chemistry books. Understanding the context – why certain elements bond, the principles of oxidation, and the safety precautions involved in handling chemicals – will deepen your appreciation. And remember, while learning about these compounds is engaging, any practical exploration should always be conducted under the supervision of qualified professionals in a controlled environment. So, keep that inquisitive spirit alive, and happy learning!
