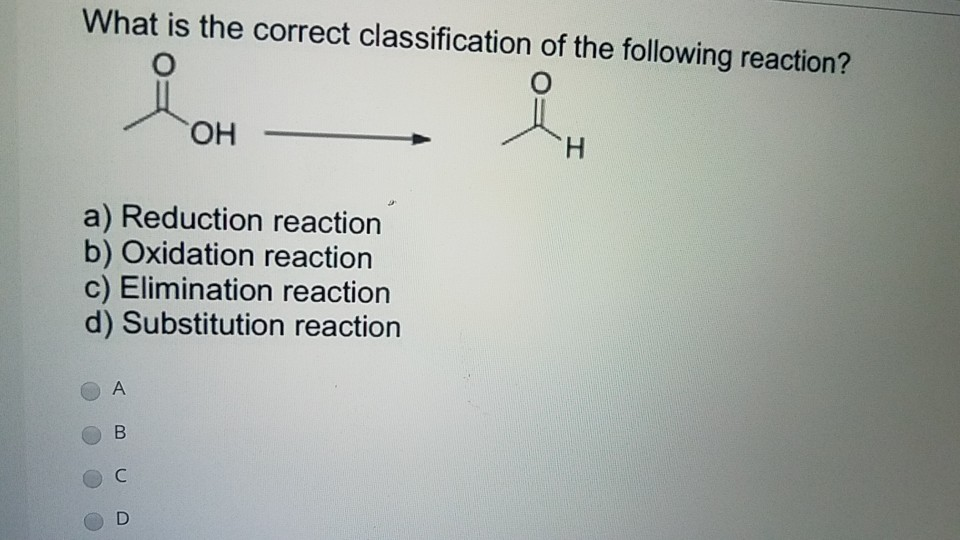
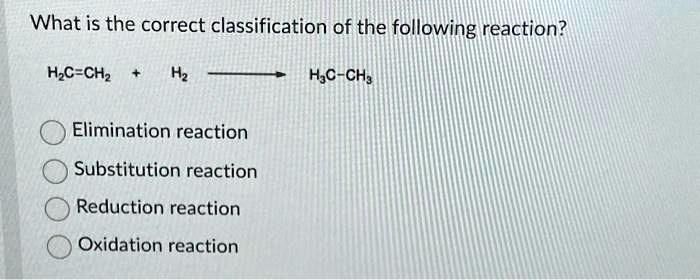
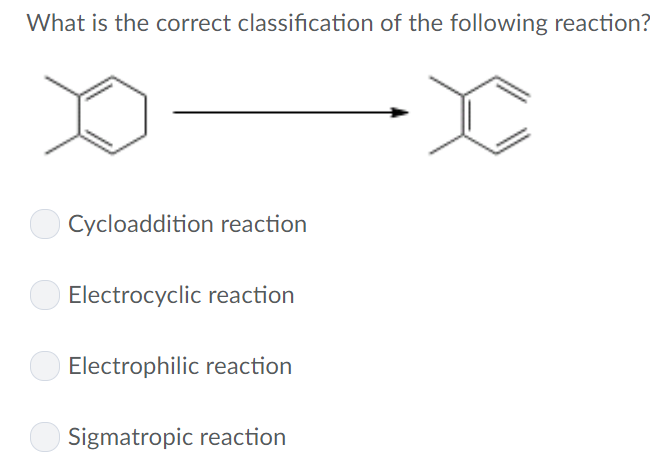
What Is The Correct Classification Of The Following Reaction
Ever looked at a recipe and wondered why certain ingredients react the way they do? Or maybe you've seen a science demonstration and been utterly captivated by the transformations happening before your eyes? That's the magic of chemistry, and at its heart lies the fascinating world of chemical reactions. Figuring out what kind of reaction is happening is like being a detective, piecing together clues to understand how matter changes. It's not just for scientists in lab coats; it's a fundamental concept that helps us understand everything from baking a cake to the complex processes that keep our planet alive. So, let's dive into the exciting realm of chemical classifications and become reaction-identifying wizards!
The Thrill of the Classification Game
Why is classifying chemical reactions so cool? Think of it as putting things into neat little boxes, but instead of socks or tools, we're organizing the fundamental ways molecules interact and rearrange themselves. It's a way to make sense of the seemingly chaotic world of chemical change. When you can classify a reaction, you unlock a deeper understanding of its behavior, predict its products, and even control its outcomes. This isn't just about memorizing terms; it's about developing a framework for thinking about how the universe works at its most fundamental level. Plus, imagine the satisfaction of looking at a chemical equation and instantly knowing its type – it’s like cracking a secret code!
Unlocking the Benefits of Understanding Reactions
The benefits of understanding chemical reaction classifications extend far beyond the classroom. In everyday life, this knowledge can make you a savvier consumer and a more informed individual. For example, understanding combustion reactions helps us appreciate the energy harnessed in fuels, while knowing about neutralization reactions can explain why antacids work so well. In a more professional context, these classifications are absolutely crucial. Engineers use them to design efficient industrial processes, from manufacturing plastics to producing life-saving medicines. Environmental scientists rely on them to understand pollution and develop remediation strategies. Even aspiring chefs can benefit; understanding acid-base reactions can help them create the perfect tangy sauce or leavened bread.
The Case of the Mysterious Transformation
Now, let's get to the heart of the matter. Imagine you're presented with a chemical scenario and asked: "What is the correct classification of the following reaction?" This is where the detective work truly begins. We're not just looking at what goes in and what comes out; we're analyzing the pattern of change. Is something being added? Is something being taken away? Is there a transfer of electrons? Or is it a simple swap of partners? Each of these patterns falls into a specific category, and each category tells us something unique about the forces at play.
The ability to classify reactions empowers us to predict, control, and innovate with chemical processes.
What is the correct classification of the following reaction? Select
Let's explore some of the most common and foundational types of reactions you'll encounter. It's like learning the alphabet before you can write a novel – these basic building blocks are essential.
Combination (Synthesis) Reactions: Building Blocks Unite!
Think of a combination reaction, also known as a synthesis reaction, as two or more simpler substances joining forces to create a single, more complex substance. It's like LEGO bricks clicking together to form a larger structure. The general form is often represented as: A + B → AB. A classic example is when hydrogen gas (H₂) and oxygen gas (O₂) combine to form water (H₂O). It’s a fundamental process in the universe, creating new compounds from existing elements.
Decomposition Reactions: Breaking Things Down
On the flip side, we have decomposition reactions. Here, a single compound breaks down into two or more simpler substances. Imagine a complex machine being dismantled into its individual parts. The general form is usually: AB → A + B. A common example is the decomposition of hydrogen peroxide (H₂O₂) into water (H₂O) and oxygen gas (O₂), which you might have seen as a foamy "elephant toothpaste" experiment. These reactions are vital for recycling elements and breaking down complex molecules.
Single Replacement (Displacement) Reactions: A Game of Musical Chairs
In a single replacement reaction, also called a single displacement reaction, one element takes the place of another element in a compound. It's like a new player stepping onto the field and kicking out an existing one. The general form is: A + BC → AC + B. For instance, if you place a zinc metal strip into a solution of copper sulfate, the zinc will replace the copper in the compound, forming zinc sulfate and solid copper. These reactions are important in metallurgy and electrochemistry.
Double Replacement (Displacement) Reactions: A Swap of Partners
Taking it a step further, double replacement reactions, or double displacement reactions, involve the ions of two compounds swapping places to form two new compounds. It's like two couples at a dance switching partners. The general form looks like this: AB + CD → AD + CB. A common example is when silver nitrate (AgNO₃) reacts with sodium chloride (NaCl) to form silver chloride (AgCl) – a white precipitate – and sodium nitrate (NaNO₃). These reactions are often used to form precipitates or to neutralize acids.
Combustion Reactions: The Fire Starters
Combustion reactions are perhaps the most visually dramatic. They involve a substance reacting rapidly with oxygen, usually producing heat and light. Think of burning wood or natural gas. The most common type of combustion involves hydrocarbons (compounds of hydrogen and carbon), where the products are carbon dioxide (CO₂) and water (H₂O), along with the release of energy. The general form for a hydrocarbon combustion is: Hydrocarbon + O₂ → CO₂ + H₂O + Energy. These reactions are the backbone of our energy production.
Acid-Base Reactions (Neutralization): Finding Balance
Acid-base reactions, often called neutralization reactions, occur when an acid and a base react to form a salt and water. It's about bringing opposing forces into balance. The general form is: Acid + Base → Salt + Water. For example, hydrochloric acid (HCl) reacting with sodium hydroxide (NaOH) produces sodium chloride (NaCl) – common table salt – and water (H₂O). Understanding these reactions is key in everything from digestion to industrial chemical processes.
By recognizing these fundamental patterns, you can begin to decode the incredible chemical transformations happening all around us, making you a true master of the reaction classification game!

