What Is Missing In The Following Nuclear Equation

Alright, so you wanna talk science? Specifically, nuclear equations? Sounds kinda heavy, right? Like something you’d see on a chalkboard in a dusty university lecture hall. But trust me, it’s actually way cooler than it sounds. Think of it like a cosmic detective story. We’ve got clues, and we gotta figure out what’s missing.
So, what’s the deal with nuclear equations? Basically, they’re like recipes for atoms. They show us how one atom can change into another, or how it can break apart. It’s all about the tiny, tiny stuff that makes up everything. And when we talk about nuclear stuff, we’re talking about the very heart of the atom: the nucleus. That’s where the protons and neutrons hang out.
Now, here’s where the fun really starts. Imagine you’re looking at a nuclear equation, and something just… isn’t adding up. It’s like a magic trick gone slightly wrong. One side of the equation has a certain amount of "stuff," and the other side… well, it’s a bit short. Something has vanished into thin air, or perhaps, into pure energy!
The Case of the Missing Particle
Let’s say we’ve got something like this:
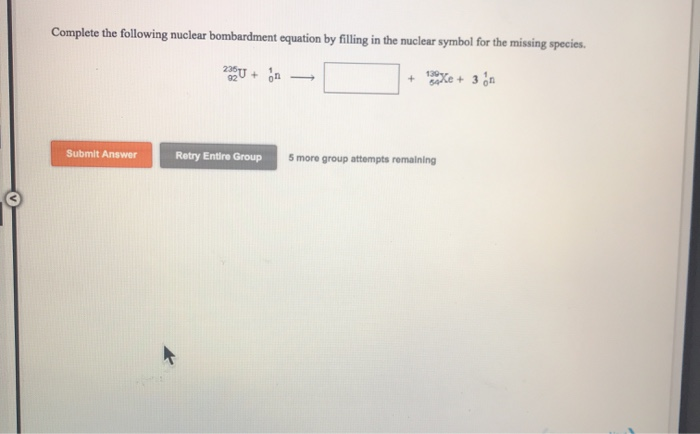
Uranium-238 → Thorium-234 + ?
See that question mark? That’s our mystery guest. Uranium-238 is a big, happy atom. It’s famous for being radioactive. That means it’s a little unstable and likes to shed some of its inner bits to become more chill. In this case, it’s shedding stuff to become Thorium-234. But wait a minute!
We need to make sure everything balances out. It’s like a cosmic balancing act. Two main things gotta stay equal: the mass number and the atomic number.
The mass number? That’s the total count of protons and neutrons. Think of it as the atom’s "weight." The atomic number? That’s just the number of protons. It tells you which element you’re dealing with. Every element has its own special atomic number. Helium always has 2 protons. Gold always has 79. It’s their ID card.
So, let's look at our equation again. Uranium-238. That 238 is its mass number. Its atomic number? That’s 92. So we have: (92 protons, 238 total particles).
On the other side, we have Thorium-234. Its mass number is 234. Its atomic number is 90. So we have: (90 protons, 234 total particles).

The Numbers Game
Let’s check the mass number first. Before the reaction, we had 238 particles. After, we have 234. That means we’re short 238 - 234 = 4 particles. So, our missing particle has a mass number of 4.
Now, let’s check the atomic number. Before, we had 92 protons. After, we have 90. That means we’re short 92 - 90 = 2 protons. So, our missing particle has an atomic number of 2.
So, we’re looking for something with a mass number of 4 and an atomic number of 2. What could that be?
Enter the Alpha Particle!
BAM! We found it. The culprit is an alpha particle. What is an alpha particle? It’s basically a helium nucleus. Yep, a tiny bit of helium! It has 2 protons and 2 neutrons. That makes its mass number 4 and its atomic number 2. How’s that for a twist?
So, our complete equation looks like this:
Uranium-238 → Thorium-234 + Alpha Particle (Helium-4)
Or, in the cool science shorthand:

²³⁸₉₂U → ²³⁴₉₀Th + ⁴₂He
See? Everything balances out perfectly. It’s like a cosmic puzzle solved! It’s pretty neat how nature keeps things in check, isn't it? It’s not just random chaos; there are rules, and these equations help us see them.
More Mysteries to Uncover
But wait, there’s more! Alpha particles aren’t the only things that can go missing. Sometimes, atoms decide to ditch an electron. That’s called a beta particle. Beta decay is super common. It happens when a neutron in the nucleus transforms into a proton and an electron, and that electron is ejected.
Here’s a peek at beta decay:
Carbon-14 → Nitrogen-14 + ?
Carbon-14 is famous, right? It's used for radiocarbon dating. It’s all about figuring out how old ancient stuff is. Pretty cool, huh?
Carbon-14 has a mass number of 14 and an atomic number of 6. So, (6 protons, 14 total particles).
Nitrogen-14 has a mass number of 14 and an atomic number of 7. So, (7 protons, 14 total particles).
Let’s check the numbers. Mass number: Before, 14. After, 14. That’s a perfect match! No missing mass here. But the atomic number is different. Before, 6. After, 7. We’ve gained a proton. That’s weird, right? How does that happen?
The Beta Particle Strikes!
This is where the beta particle comes in. Remember, it's an electron. Electrons have almost no mass, so they don't affect the mass number. But they have a negative charge, which is like having a "-1" for the atomic number.
So, in our equation, we started with 6 protons. We ended up with 7 protons. That means something must have been emitted that increased the proton count by 1. And that’s exactly what a beta particle does! It’s like a proton suddenly decided to sprout an electron and kick it out.
The full equation is:
Carbon-14 → Nitrogen-14 + Beta Particle + Antineutrino
Okay, okay, I added a little something extra there: an antineutrino. Don’t worry too much about that little guy. He’s super elusive and doesn’t affect our mass or atomic number calculations. He’s just there to keep the energy balanced. Think of him as the invisible friend at the party.
In shorthand:
¹⁴₆C → ¹⁴₇N + ⁰₋₁e + ν̄
See how the 0 for the mass number means it barely adds anything? And the -1 for the atomic number means it effectively makes the "proton count" go up by one when you consider charges.
Why Is This Stuff Fun?
So, why is this whole "what’s missing" thing so fascinating? Because it shows us that the universe has a built-in sense of order. Even at the tiniest scales, things have to balance. It’s like playing with building blocks; you can’t just make pieces appear out of nowhere.
And it’s not just about balancing numbers. These missing pieces are real particles with their own properties. Alpha particles are essentially miniature helium atoms. Beta particles are speedy electrons. These are the tiny building blocks of everything we see, from stars to your coffee mug!
Plus, it’s a glimpse into the weirdness of quantum mechanics. Things can transform, energy can be released, and particles can be incredibly elusive. It’s like peeking behind the curtain of reality. And who doesn't love a good mystery?
So next time you hear about nuclear equations, don't just think of complicated math. Think of cosmic puzzles, detective work, and the incredible, invisible world that makes up our universe. It's a fun game of "spot the missing piece," and the universe is the ultimate playground!
