What Is Happening To A Substance Undergoing Thermal Expansion

So, you've probably noticed this phenomenon before, even if you didn't have a fancy scientific name for it. Think about a really hot summer day. You're walking barefoot on the pavement, and BAM! It’s like the ground decided to do a Hulk-out and get bigger. Or maybe you've tried to open a stubborn jar lid, and it feels like it's fused on. Ever wondered why? Well, my friends, you've been witnessing the magic, or sometimes the mild annoyance, of thermal expansion.
Basically, when you heat something up, it gets a little… stretched out. It’s like your favorite pair of jeans after a particularly enthusiastic laundry session in hot water. They start out snug, but after a good soak and some heat, they might feel a tad looser, right? Substances are kind of the same way, just on a molecular level.
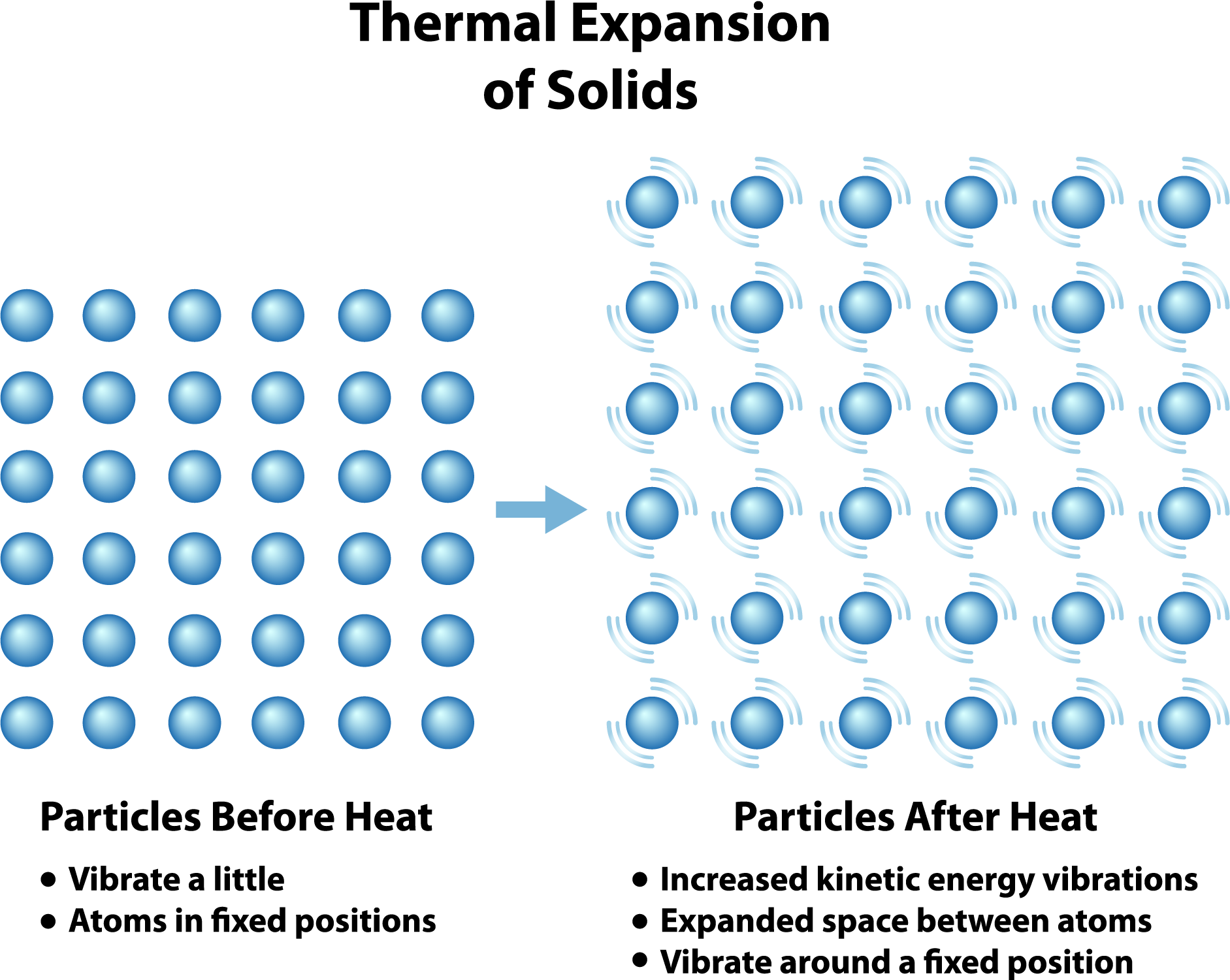
Imagine all the tiny little particles (atoms and molecules, the building blocks of everything) that make up your spoon, your car tire, or even that loaf of bread you’re about to toast. When things are cool, these particles are pretty chill. They’re just hanging out, doing their thing, vibrating gently. Think of them as a group of teenagers at a sleepover, all huddled together, whispering and fidgeting a bit, but not really moving around much.
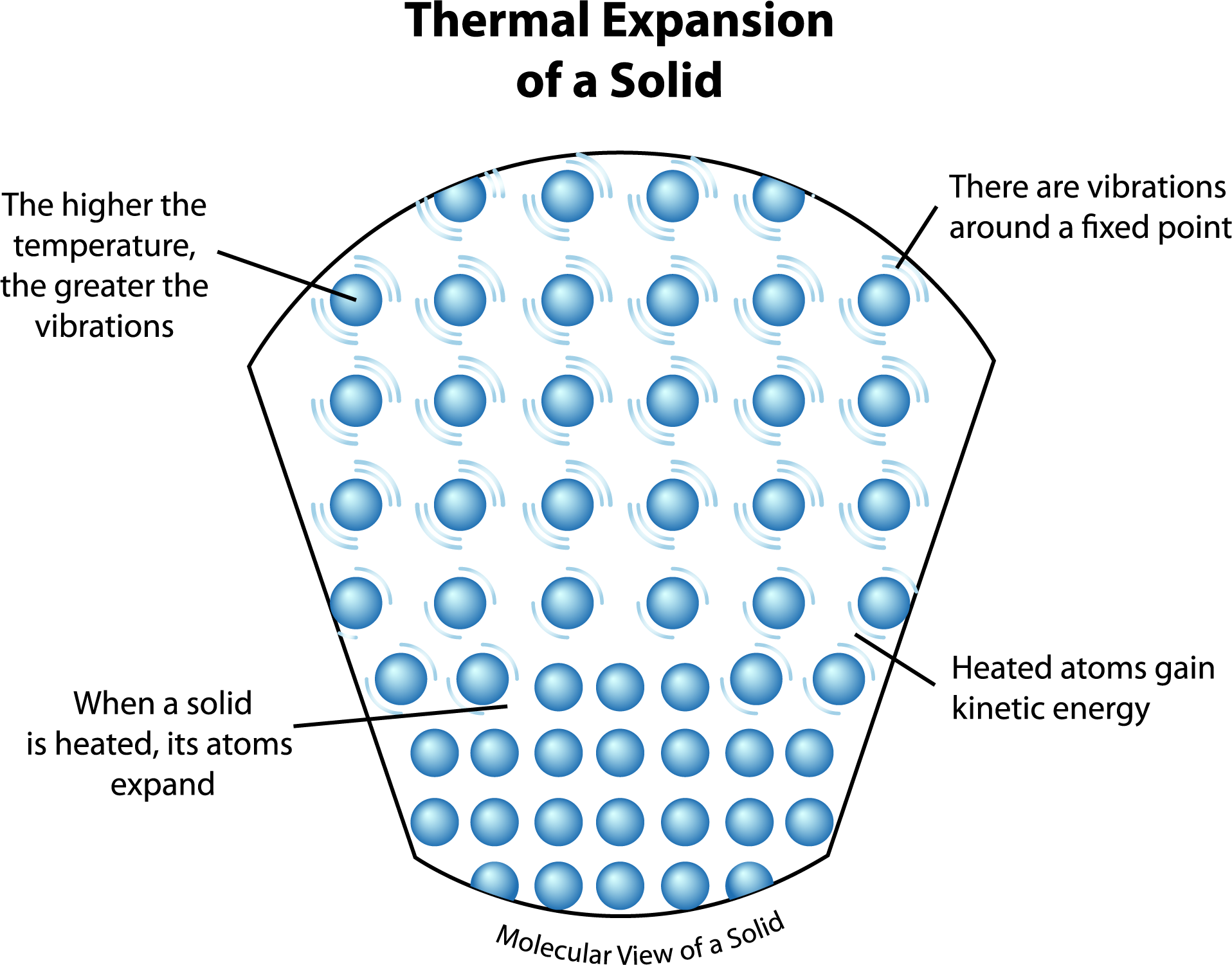
Now, crank up the heat! This is where things get exciting, or at least more exciting. Those particles start to get energized. It's like someone just blasted their favorite pump-up playlist in the middle of that sleepover. They start bouncing around, vibrating more vigorously, and generally getting a lot more enthusiastic. They need more elbow room, so to speak.
Because these little guys are now jittering and jiggling with more gusto, they push away from their neighbors. It’s like they’re saying, "Whoa there, buddy, give me some space!" This pushing and shoving, this collective desire for more personal space, causes the entire substance to expand. It’s not just one particle getting bigger; it’s the whole darn thing taking up more room.
Think about it like a crowded elevator. When it's just a few people in there, it's cozy. But imagine everyone suddenly gets really hyper and starts doing little jigs and trying to high-five each other. What happens? The elevator starts to feel way smaller, and you’d desperately want to expand the walls if you could. Thermal expansion is the opposite – the substance expands to accommodate the energized particles.
This is why bridges have those weird little gaps at the ends. They look a bit like they’re having an identity crisis, those gaps. But they’re not. They’re there for a very good reason. On a scorching hot day, the metal and concrete of the bridge will get longer, or expand. If those gaps weren’t there, the bridge would have nowhere to go! It would be like trying to shove a giant beach ball into a small suitcase – something’s gotta give, and usually, it’s not pretty. The bridge might buckle or crack, which would be a much bigger problem than a slightly awkward gap.
It’s the same principle with railway tracks. Ever seen those little zigzags in the middle of the tracks? They’re not a design choice by a very artistic railway engineer. They’re there to allow the metal to expand without warping. Imagine a long, straight metal rod on a super hot day. It wants to get longer. If it's constrained, it'll start to bend and buckle. Those zigzags give it the freedom to wiggle and jiggle its way into a slightly longer form without causing trouble.
Now, let’s talk about liquids. Water is a classic example. You might notice that water in a pot seems to take up a bit more space when it’s boiling than when it’s cold. It’s not your imagination. The water molecules are having a party, and they need more room to dance. This is why you should never fill a glass container with boiling water and put it straight into the freezer, or vice-versa. The sudden temperature change can cause the water to expand or contract so rapidly that it can shatter the glass. It’s like asking someone to go from a hot yoga class to an ice bath in zero seconds. Not a good look for anyone involved.
And then there are gases. Gases are the ultimate party animals when it comes to thermal expansion. If you heat up a gas, those particles go absolutely bonkers! They bounce off the walls of their container like a toddler who’s just discovered a hidden stash of candy. They spread out, filling every available nook and cranny. This is why a balloon filled with hot air floats higher than a balloon filled with cold air. The hot air is less dense because the gas particles have spread out, making the balloon lighter overall.
Think about a hot air balloon. It’s basically a giant bag of air that someone’s decided to heat up. The burner is like a massive hairdryer, energizing the air inside. This energized air takes up more space, making the balloon less dense than the surrounding cooler air, and voila! Up, up, and away you go. It’s a beautiful, majestic demonstration of thermal expansion in action, often enjoyed with a picnic basket and a sense of wonder.
What about when things get cold? Well, that’s the opposite of thermal expansion. It’s called thermal contraction. When you cool something down, those particles start to calm down. They get less energetic, vibrate less, and start to huddle closer together. It’s like those teenagers at the sleepover realizing it’s 3 AM and time to wind down. They stop bouncing and start snuggling up for warmth.

So, that jar lid that was impossible to open when it was cold? Try running it under some hot water. The metal lid expands slightly, and suddenly, it’s like magic – it twists right off! It’s the ultimate kitchen hack, powered by science. You’re not a superhero; you’re just a smart individual who understands how heat affects matter.
This contraction thing can also cause problems. In very cold climates, pipes can freeze and burst. Why? Because as the water inside freezes, it actually expands! This is one of the few exceptions to the general rule, and it’s a rather inconvenient one. The ice crystals form in a way that takes up more space than the liquid water. So, the water expands, pushes against the pipe walls, and if the pressure gets too much, the pipe splits. It’s like trying to cram an oversized souvenir into your luggage on the way home from vacation. Something’s going to break.
You see this contraction in action in various places. Think about a thermometer. The mercury or alcohol inside rises when it's hot (expanding) and falls when it's cold (contracting). It’s a little liquid barometer of your day’s temperature, quietly doing its job based on these fundamental principles.

Even the Earth’s crust experiences thermal expansion and contraction, albeit on a geological timescale. Imagine the planet’s surface as a giant, slow-moving puzzle. Different temperatures cause these massive plates to shift and groan, contributing to earthquakes and volcanic activity over millions of years. So, that little tremor you felt? Could be the Earth’s way of stretching after a long day.
It’s not just solids, liquids, and gases that expand. Even plasma, the fourth state of matter (think lightning bolts and the sun), expands when heated. Though, trying to get a plasma sample to demonstrate thermal expansion in your kitchen might be a tad more challenging than heating up a metal rod.
So, next time you see a gap in a bridge, a slightly loose lid, or a balloon floating majestically in the sky, give a little nod to thermal expansion. It’s the unseen force that explains so much of our everyday world, from the practical engineering marvels around us to the simple joys of a perfectly fitting jar lid. It’s the universe’s way of reminding us that everything, even seemingly solid objects, are just collections of energetic particles, always ready to stretch out and make some room when things heat up. It’s a subtle dance of expansion and contraction, a fundamental rhythm of the cosmos, happening all around us, all the time. And it’s pretty darn cool, if you ask me.
