What Is Being Oxidized And Reduced In Photosynthesis
Hey there, amazing humans! Ever stopped to think about those green superheroes, plants, and how they pull off their daily magic? You know, the whole "turning sunshine into food" thing? It’s pretty wild, right? Well, today, we’re going to peek behind the curtain of one of their most impressive tricks: photosynthesis. And guess what? It’s all about a little dance called oxidation and reduction. Sounds fancy, I know, but trust me, it’s way cooler and more relevant to your life than you might think!
So, what’s this whole "oxidation and reduction" hullabaloo about? Think of it like a cosmic game of "tag" happening at the atomic level, specifically with electrons. Electrons are tiny, energetic particles that are basically the MVPs of chemistry. They love to jump from one atom or molecule to another. And when they do, things get interesting!
In photosynthesis, our leafy pals are essentially trying to capture the energy from sunlight. They do this by performing a series of chemical reactions, and at the heart of it all are these electron transfers. Let's break down the players:
The Two Main Acts: Oxidation and Reduction
Imagine you have two friends, Alex and Ben. Alex has an extra cookie and is happy to share. Ben doesn’t have a cookie and is a bit peckish. When Alex gives Ben a cookie, Alex has lost a cookie, and Ben has gained one, right? It’s kind of like that with electrons!
Oxidation is when a molecule or atom loses electrons. Think of it as "giving away the goods." The molecule becomes a bit less "electron-rich."
Reduction is the flip side. It’s when a molecule or atom gains electrons. This is like "scoring the prize!" The molecule becomes more "electron-rich."
And here’s the super important, totally mind-blowing part: oxidation and reduction always happen together. You can’t have one without the other! It's like a handshake – one person offers their hand, and the other accepts. They are a dynamic duo, a chemical bromance, if you will! This is why chemists often call the whole process redox reactions.
Who's Getting Oxidized in Photosynthesis?
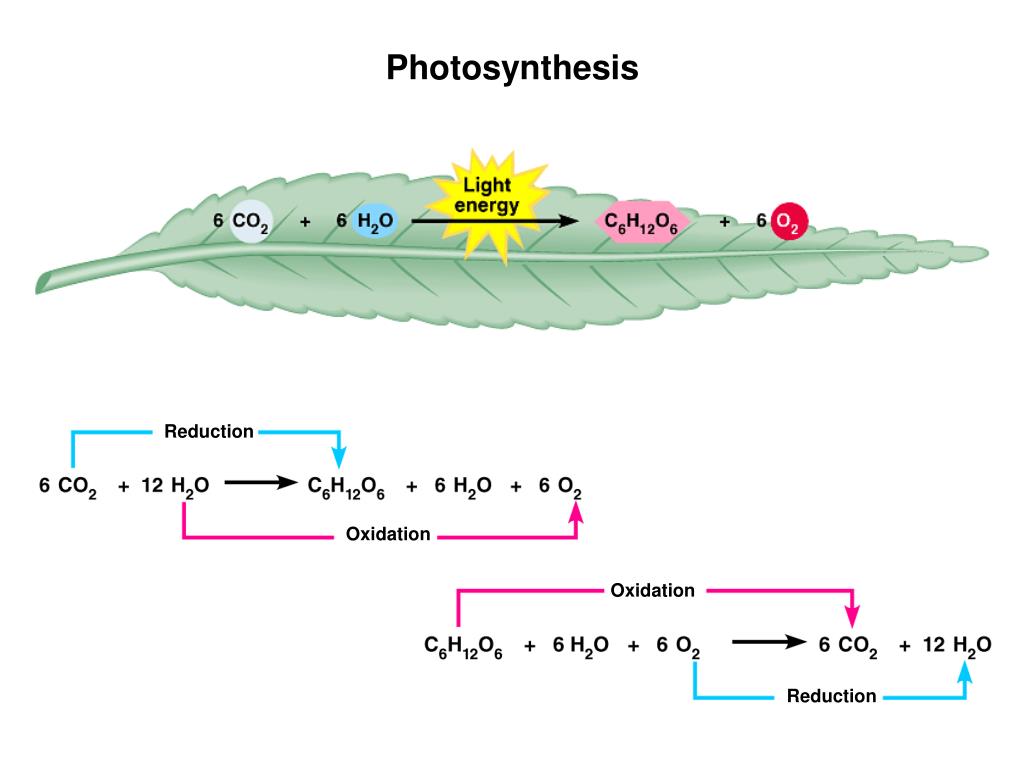
Now, let's get back to our green friends. What are they sacrificing in this electron-tag game? Well, it all starts with water. Yep, that H₂O you drink and that falls as rain? Plants are doing amazing things with it!
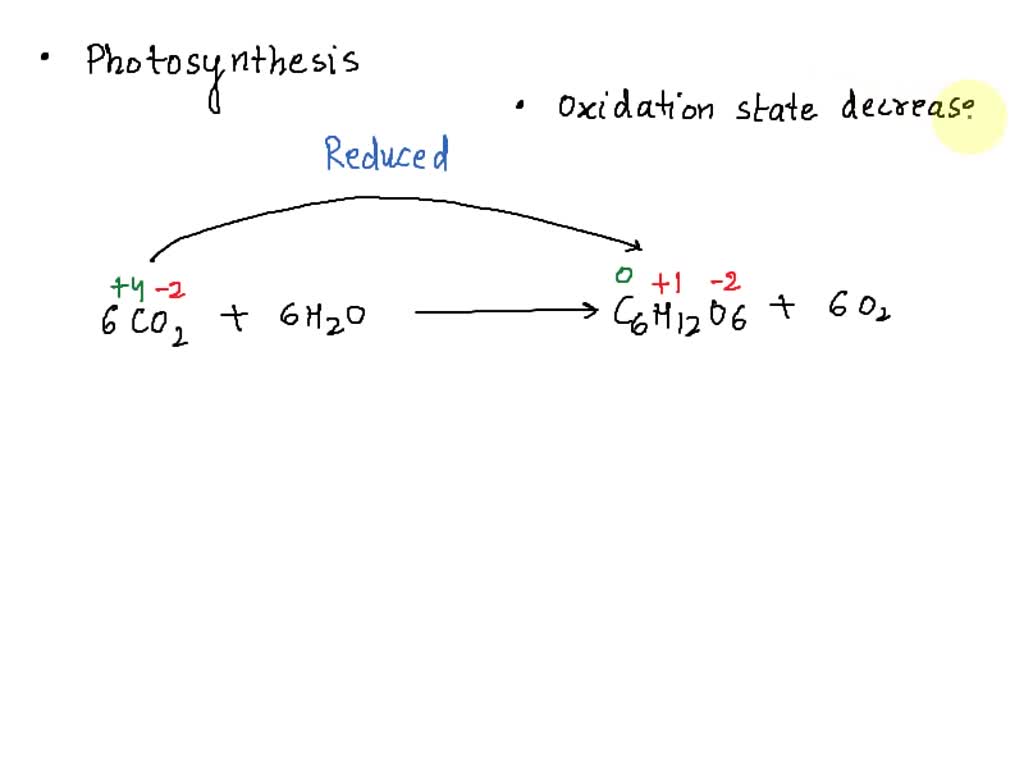
During the first stage of photosynthesis, which happens in the chloroplasts (those little green powerhouses inside plant cells), water molecules get split apart. And when water (H₂O) is split, it loses electrons. So, in this crucial step, water is being oxidized.
This process is called photolysis, which literally means "splitting by light." Sunlight’s energy is the spark that ignites this reaction. As water loses its electrons, it also releases oxygen (O₂) – hello, the air we breathe! – and protons (H⁺ ions).
Isn't that incredible? Our very breath, the life-giving oxygen, is a byproduct of plants being oxidized! It’s like they’re saying, "Here, have some of my leftovers, and thanks for the sunshine!"

And Who's Getting Reduced?
So, if water is losing electrons, who’s happily picking them up? Who’s getting reduced? This is where the plant's "food-making" really kicks into gear. Those electrons, energized by sunlight and "donated" by water, are passed along a series of special molecules. Think of it like a tiny, incredibly efficient conveyor belt powered by light!
The ultimate recipient of these electrons (after a few exciting stops along the way) is a molecule called NADP⁺. This is a bit of a mouthful, so let’s just call it "our electron taxi."
NADP⁺ is waiting around, looking a bit electron-deprived. When it picks up electrons (along with some protons), it becomes NADPH. So, NADP⁺ is being reduced into NADPH. It has gained electrons and is now an energized, electron-carrying molecule, ready for the next stage of photosynthesis.
This NADPH is like a fully charged battery, packed with the energy and electrons that the plant needs to build sugars from carbon dioxide.

The Grand Finale: Making Sugar!
Now, you might be asking, "Okay, so water gets oxidized, and NADP⁺ gets reduced. But where does the sugar come from?" Great question! This is where the second stage of photosynthesis, known as the Calvin cycle, comes in.
In the Calvin cycle, the plant takes carbon dioxide (CO₂) from the air. Using the energy and electrons that were captured and carried by NADPH (thanks to our redox reactions!), the plant rearranges the atoms to create glucose – that’s a type of sugar!
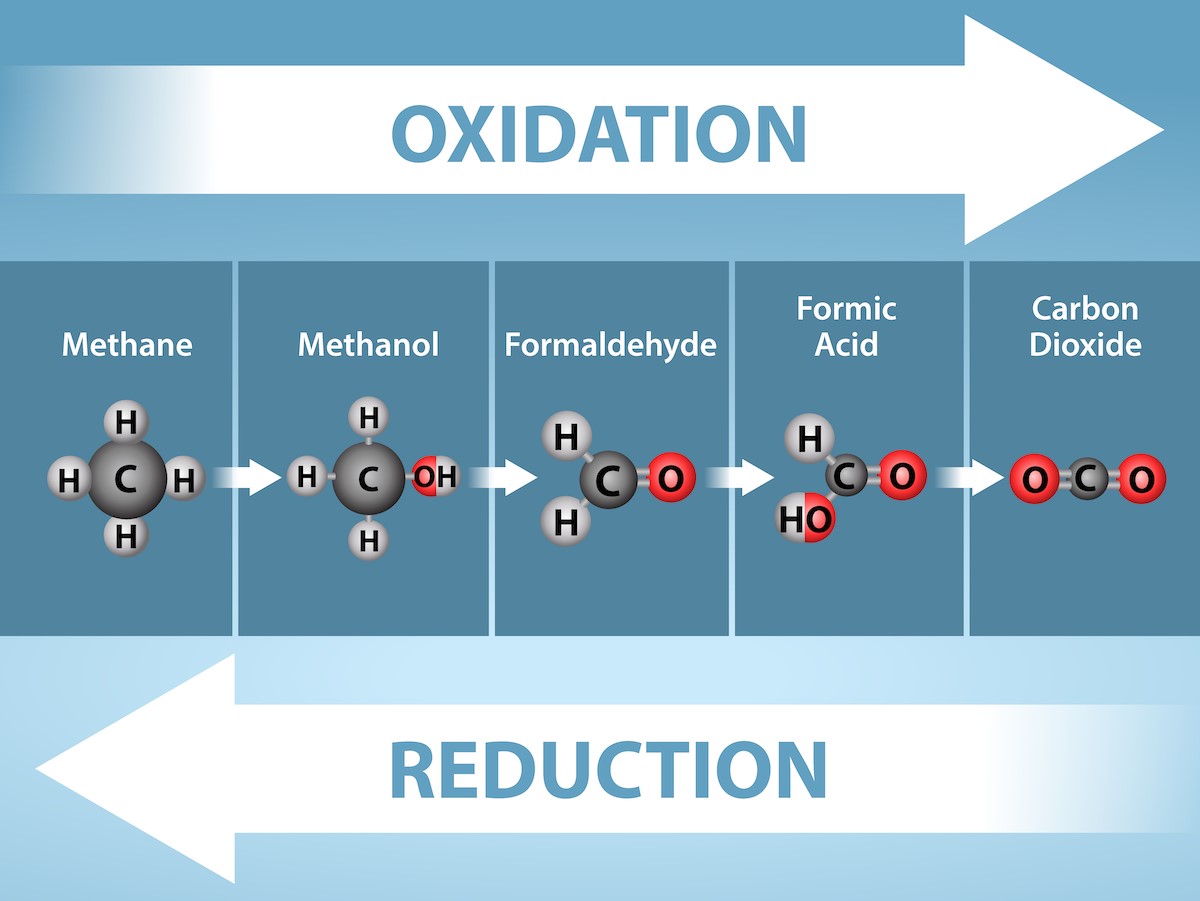
So, while CO₂ isn’t directly gaining electrons in the same way as NADP⁺, the entire process of sugar synthesis, fueled by the reduced NADPH, involves a series of redox reactions where carbon compounds are ultimately being reduced. Think of it as the plant taking simple building blocks (CO₂) and, with the help of those energetic electrons, assembling them into something more complex and energy-rich – sugar!
In essence, the carbon in CO₂ is going from a more oxidized state to a more reduced state as it’s incorporated into sugars. It's a fascinating transformation!
Why This Makes Life More Fun
Okay, so we’ve talked about water, electrons, and sugars. How does this make life more fun? Think about it! Every time you see a lush green leaf, or take a deep breath of fresh air, you’re witnessing the incredible power of these redox reactions in action.
Plants are literally turning sunlight, water, and air into the food that sustains them and, ultimately, us! It’s a beautiful, intricate dance of molecules, a testament to the ingenuity of nature. It means that the energy in that apple you’re eating? It all started with sunlight and a plant performing some seriously cool oxidation and reduction!
Understanding this can add a whole new layer of appreciation to the world around you. It’s like knowing the secret handshake of the universe. You can look at a tree and think, "Wow, that majestic being is performing complex chemistry right now to keep us all alive!" It’s a little bit science, a whole lot of magic, and definitely makes for some fascinating dinner conversation.
So, next time you’re out in nature, take a moment to thank those photosynthetic marvels. They’re not just making oxygen and sugar; they're engaged in a fundamental chemical ballet that powers our planet. And isn’t it inspiring to know that such profound and life-sustaining processes are happening all around us, all the time, driven by the simple, yet extraordinary, exchange of electrons?
Keep exploring, keep wondering, and keep appreciating the amazing chemistry that makes our world thrive!
