What Ionic Compound Is Formed From Cesium And Nitrogen
Hey there! So, you're curious about what happens when you smoosh together cesium and nitrogen, huh? Like, what magical molecule do they cook up? It's a question that might pop into your head while you're staring at your periodic table, maybe after a particularly strong cup of coffee, right?
Well, let's get cozy, grab our virtual mugs, and chat about it. This is going to be way more fun than memorizing electron shells, I promise. We're talking about the real-deal chemistry here, the kind that makes things… well, make sense! Or at least, as much sense as chemistry can sometimes make, anyway.
So, we've got cesium (Cs), and we've got nitrogen (N). Two totally different characters on the chemical stage. Cesium, that big ol' alkali metal, all shiny and reactive. And nitrogen, the gas that makes up most of the air we breathe, pretty chill usually. What do they form? Drumroll, please… cesium nitride!
Yeah, I know, not the most exciting name in the world. It doesn't exactly roll off the tongue like "chocolate chip cookie," does it? But behind that slightly clunky name is some pretty neat science. Think of it as the chemical equivalent of a really solid, dependable partnership. No flashy fireworks, maybe, but definitely a strong bond.
Cesium: The Generous Giant
Let's talk about cesium for a sec. This guy is in Group 1 of the periodic table, the alkali metals. And you know what that means? It means it's practically begging to give away an electron. Like, it has one lonely little electron in its outermost shell, just chilling, waiting for a purpose. It's so eager to lose it, it's almost embarrassing!
Cesium is the biggest and heaviest of the stable alkali metals. It’s super reactive. Seriously, you can't just leave it lying around. It reacts with water so violently, it’s a bit of a showstopper. Think poof! Not something you want to mess with without proper safety gear, obviously. So, it’s a bit of a drama queen, but in a very useful way for chemists.
Because it's so keen to shed that electron, cesium loves to bond with things that want electrons. Things that are, you know, a bit electron-hungry. It's the ultimate electron donor. It’s like the generous friend who’s always paying for the round of drinks. Always willing to share!
And when it loses that electron, it becomes a positively charged ion, a cesium ion. We write that as Cs+. Simple enough, right? That little plus sign is its way of saying, "Yep, I gave away an electron!"
Nitrogen: The Demanding Diva
Now, let's turn our attention to nitrogen. This is the element that’s everywhere! It's 78% of the air you're breathing right now. Pretty important stuff, considering. But nitrogen, in its elemental form (N2), is pretty stable. It has a triple bond, which is like, the ultimate chemical handshake. Super strong and not easily broken.
However, when we're talking about forming compounds, nitrogen can be a bit… demanding. It likes to gain electrons. It's got five electrons in its outer shell, and it’s really only happy with eight. So, it’s always looking for three more to complete its set. It’s like someone who’s always looking for that last missing piece of a puzzle.
To get those three extra electrons, nitrogen often forms negatively charged ions. When it does, we call it a nitride ion. And this little nitride ion is not messing around. It’s got a charge of -3. That’s right, three electrons it’s snatched up. It's the chemical equivalent of a vacuum cleaner, sucking up all the electron goodness it can find.
So, we have our cesium, ready to give away a positive charge (Cs+), and our nitrogen, ready to snatch up negative charges (N3-). See where this is going?
The Big Chemical Hug
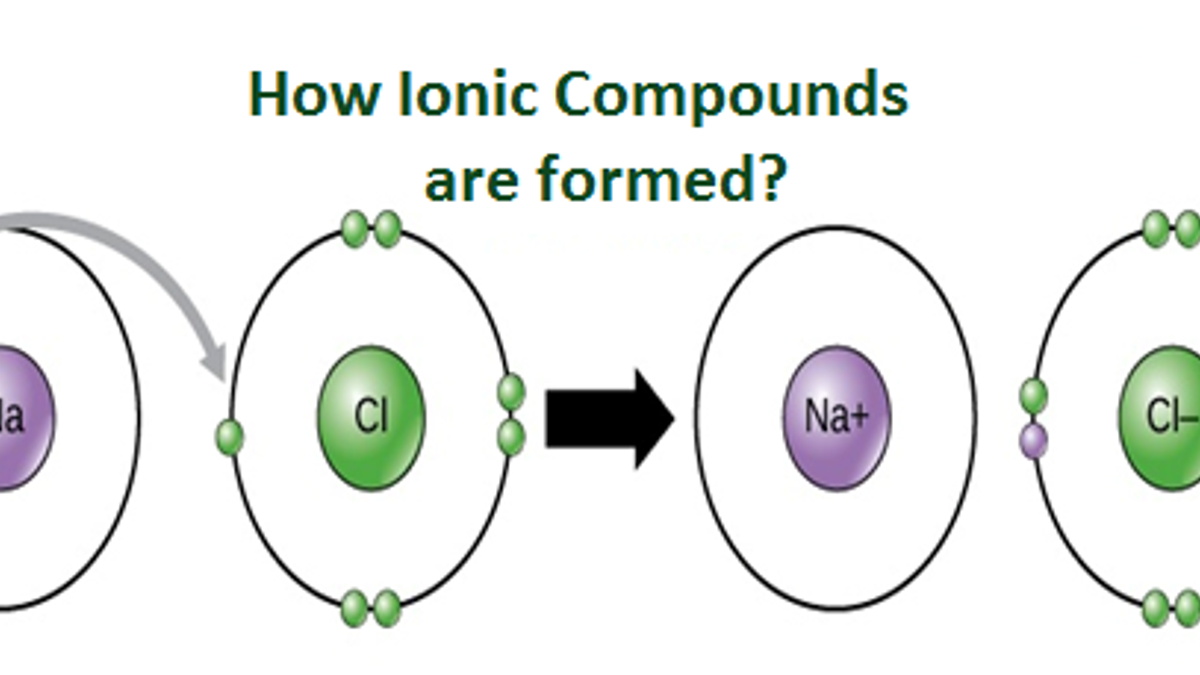
This is where the magic, or rather, the electrostatics, happens! We have our Cs+ ion, positively charged and ready to mingle, and our N3- ion, negatively charged and looking for some positive attention. What do they do? They get attracted to each other, of course!

It's like a cosmic dance, a chemical matchmaking service. Opposite charges attract, right? It’s the fundamental rule of the universe, or at least, chemistry. So, the positive cesium ion and the negative nitride ion lock onto each other with a strong electrostatic force. This is what we call an ionic bond. It’s not a covalent sharing of electrons, like in some relationships. This is more like a complete transfer, a give-and-take that results in a stable connection.
But here’s the quirky part, the bit that makes you go, "Huh?" We have a cesium ion with a +1 charge and a nitride ion with a -3 charge. If we just stuck them together one-to-one, like CsN, we’d have a +1 and a -3, which doesn't add up to a neutral compound. It would be like trying to balance a checkbook with way more money coming out than going in. It just wouldn’t work!
So, nature, being clever as always, figures out a way to make it work. To balance out that -3 charge from the nitride ion, we need three cesium ions, each with a +1 charge. That's three positive charges to perfectly cancel out the three negative charges. Get it?
So, the formula for cesium nitride isn't just CsN. Oh no. It's Cs3N. Three cesium atoms for every one nitrogen atom. It's like a little family unit, a team that's perfectly balanced and happy.
So, What IS Cesium Nitride?
Okay, so we've figured out the recipe: three Cs+ ions and one N3- ion. What does this Cs3N actually look like? What are its properties? Well, it’s a solid, for starters. Most ionic compounds are solids at room temperature. They form these crystalline structures, like tiny, perfectly arranged building blocks.

Think of it like a microscopic crystal lattice. The cesium ions and nitride ions are packed together in a very specific, repeating pattern. It's organized, it's neat, and it's held together by those powerful electrostatic forces. It's not going to fall apart easily.
Cesium nitride is typically described as a dark red or even black solid. Not exactly a cheerful colour, is it? Maybe it’s reflecting its intensity! It’s also a pretty strong oxidizing agent, which means it likes to steal electrons from other things. See, nitrogen is still a bit greedy, even in this compound!
And, just like a lot of alkali metal nitrides, it’s quite reactive. Remember how we said cesium is a drama queen? Well, when it's bonded with nitrogen, it’s still got some of that spirited energy. It can react with water, for instance, but in a slightly different way than pure cesium. It can decompose when exposed to air and moisture.
It’s definitely not something you’d find in your kitchen cupboard, making toast. This is more of a specialized lab chemical, used in specific research or industrial applications. You know, the kind of stuff that scientists get excited about, and the rest of us nod and go, "Fascinating!"
Why Does This Even Matter?
You might be thinking, "Okay, but why should I care about Cs3N?" Good question! It’s not like we're going to be using it to build bridges anytime soon. But understanding how elements like cesium and nitrogen combine is fundamental to chemistry. It helps us understand:

- How chemical bonds form: This is the bedrock of all chemistry. Why do atoms stick together? What forces are at play?
- The properties of matter: The way atoms bond determines everything about a substance – its colour, its reactivity, its state of matter, and so on.
- Developing new materials: While cesium nitride itself might be niche, the principles we learn from it can be applied to designing new materials with specific properties. Think advanced batteries, catalysts, or even new types of electronics. Who knows what the future holds?
Plus, it’s just plain cool to understand how these tiny, invisible particles interact to create the world around us. It's like having a secret superpower – the ability to peek behind the curtain of reality!
A Quick Recap, Just for Fun!
So, to sum it all up, when you put cesium (the electron-giving giant) and nitrogen (the electron-seeking diva) together, they form cesium nitride, which has the formula Cs3N. This is because you need three positively charged cesium ions (Cs+) to balance out the charge of one negatively charged nitride ion (N3-).
It’s a dark, crystalline solid, held together by strong ionic bonds. It's reactive and has its own unique set of chemical behaviours. It might not be a household name, but it’s a perfect example of how elements combine based on their electron configurations and the fundamental laws of attraction.
So next time you see cesium or nitrogen on the periodic table, you can wink and say, "Ah, I know what you guys get up to!" You've cracked the code. You’ve joined the ranks of the chemically enlightened. How cool is that?
And that, my friend, is the story of cesium nitride. A little bit of chemistry, a dash of explanation, and hopefully, a lot of fun. Now, who's up for another imaginary coffee?
