What Happens When Nitrogen Fills Its Valence Shell

Hey there, chill vibes only! Ever found yourself staring up at the sky, that vast, seemingly infinite blue expanse, and wondered what’s really going on up there? Beyond the fluffy clouds and the occasional drone delivery (kidding… mostly), there’s a whole lot of science happening. And today, we’re diving into the wonderfully stable world of nitrogen, specifically what goes down when this ubiquitous element decides it’s time to chill out and fill its valence shell.
Think of the valence shell as nitrogen’s happy place. It’s the outermost layer of electrons, and like us, atoms just want to feel complete, stable, and, well, not too reactive. For nitrogen, this happy place means having a full outer shell, which translates to eight electrons. It’s the ultimate glow-up for an atom, turning a potentially eager-to-bond element into something rather… chill.
Nitrogen, you see, is the MVP of our atmosphere. About 78% of the air you’re breathing right now is nitrogen gas (N₂). That’s a pretty big deal! It’s like the background music to our lives, always present, rarely demanding attention, yet absolutely essential.
The Quest for Equilibrium: Nitrogen’s Atomic Journey
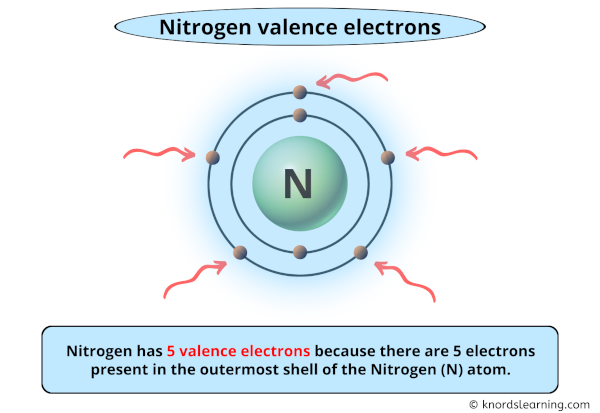
So, how does nitrogen get to its coveted eight-electron valence shell? Well, nitrogen has seven electrons to begin with. Its electron configuration is a bit like having a few friends over but still feeling like the party’s not quite complete. It’s got 5 electrons in its outer shell, and it’s just dying for 3 more to hit that magic number of 8.
Instead of trying to snatch electrons from other atoms (which, let’s be honest, can get a bit dramatic), nitrogen is a master of cooperation. It finds another nitrogen atom, and they decide to share. And not just a casual “hey, let me borrow that” kind of sharing. We’re talking about a triple bond situation. Imagine a friendship so strong, you’re literally sharing three pairs of electrons. That’s N₂ for you!
This triple bond is incredibly strong, like a perfectly brewed cup of coffee on a Monday morning – it’s solid, dependable, and takes a lot to break. This strength is why nitrogen gas is so stable and unreactive under normal conditions. It’s already achieved its ultimate state of zen.

Why This Matters: From Atmosphere to Your DNA
You might be thinking, "Okay, cool, atoms are sharing. What’s in it for me?" More than you realize! This stability is what keeps our atmosphere from exploding into a frenzy of chemical reactions. If nitrogen were constantly trying to bond with everything, life as we know it wouldn't exist. Imagine if the air itself was constantly combusting or reacting!
But here's the twist: while N₂ is super stable, nitrogen in other forms is incredibly vital. Think about it: DNA, proteins, enzymes – the very building blocks of life – all rely on nitrogen. It’s like the shy, introverted friend who, when coaxed out of their shell (pun intended!), becomes the life of the party in different contexts.
The process of converting atmospheric nitrogen (N₂) into usable forms is called the nitrogen cycle. This is where the real magic happens, and it involves a fascinating cast of characters.
The Nitrogen Cycle: Nature's Grand Chemical Symphony
The nitrogen cycle is essentially nature’s way of making sure that while N₂ chills in the atmosphere, its elemental cousin gets to do all the important work in living organisms. It’s a global recycling program that’s been running for eons.
First up, we have nitrogen fixation. This is the crucial step of breaking that super-strong triple bond in N₂ and converting it into ammonia (NH₃) or other nitrogen compounds. This can happen in a few ways:
- Biological Fixation: This is the rockstar of nitrogen fixation. Certain bacteria, especially those living in the roots of legumes (like peas, beans, and peanuts – your favorite snacks!), are nitrogen-fixing wizards. They have special enzymes that can break the N₂ triple bond. It’s like having a tiny, molecular locksmith in your soil!
- Atmospheric Fixation: Lightning strikes are nature’s high-energy nitrogen fixers. The intense heat and energy from lightning can break apart N₂ molecules and allow them to react with oxygen to form nitrogen oxides. These then dissolve in rainwater, bringing nitrogen down to Earth in a usable form. So, next time you see a thunderstorm, remember it’s a nitrogen-making event!
- Industrial Fixation: Humans have also gotten in on the act. The Haber-Bosch process, developed in the early 20th century, is a chemical marvel that allows us to produce ammonia from nitrogen and hydrogen gas under high pressure and temperature. This is how we make most of our fertilizers, which are essential for feeding the world's growing population. It’s a bit like the industrial-scale equivalent of those soil bacteria, but way more… industrial.
Once nitrogen is in a fixed form like ammonia, it can be further processed by other microbes. Nitrification is the next step, where ammonia is converted into nitrites and then nitrates. Nitrates (NO₃⁻) are the preferred form of nitrogen for most plants. They absorb these from the soil through their roots, using them to build proteins and nucleic acids.
This nitrogen then travels up the food chain. When animals eat plants, they incorporate that nitrogen into their own bodies. When plants and animals die, or when animals excrete waste, decomposition kicks in. Microbes break down organic matter, returning nitrogen compounds back into the soil.
Finally, there's denitrification. This is where some bacteria convert nitrates back into nitrogen gas (N₂), which then returns to the atmosphere, completing the cycle. It’s a neat way for nitrogen to say "see ya later!" to the soil and head back up for some atmospheric R&R.

Fun Facts and Cultural Connections
Did you know that the word "nitrogen" comes from the Greek words "nitron" (niter, or potassium nitrate) and "genes" (forming)? So, literally, "niter-forming." Quite descriptive, isn’t it?
Nitrogen is also the key ingredient in laughing gas (nitrous oxide, N₂O), which, while it can induce merriment, is also a potent greenhouse gas. So, a little goes a long way, and sometimes, too much can be a bit of a buzzkill for the planet.
In the culinary world, liquid nitrogen is used for its rapid freezing capabilities, creating dramatic smoky effects in desserts and cocktails. Think of those Instagrammable ice creams that seem to be billowing smoke – that’s our friend nitrogen, adding a touch of theatrical flair to your foodie adventures. It’s the original fog machine!
Culturally, nitrogen’s prevalence often goes unnoticed, much like the silent hum of a refrigerator. It's the unsung hero of our planet's life support system. It’s the quiet confidence of the universe, always there, providing stability without fuss.

Practical Tips for a More Nitrogen-Aware Life (Sort Of!)
While you can’t directly influence atmospheric nitrogen’s valence shell (sadly, no atom-manipulation apps yet), you can appreciate its role and support the nitrogen cycle in your own way.
- Go for the legumes! Eating beans, lentils, and peas not only gives you protein but also supports the farmers who grow these nitrogen-fixing champions. It’s a delicious way to engage with the nitrogen cycle.
- Compost your organic waste. When you compost, you’re helping those decomposer microbes do their job, returning valuable nitrogen compounds to the soil. It's like giving the nitrogen cycle a little nudge in the right direction.
- Appreciate your green spaces. Plants are the direct beneficiaries of the nitrogen cycle, turning it into the stuff of life. Spending time in nature is a subtle nod to the incredible work nitrogen is doing all around you.
- Support sustainable agriculture. Practices that promote healthy soil and reduce reliance on synthetic fertilizers help maintain the natural balance of the nitrogen cycle.
It's a subtle science, this atomic stability. It's about finding that perfect balance, that state of contentment where you don’t need to be constantly striving or reacting.
A Moment of Reflection
When nitrogen fills its valence shell, it achieves a state of profound stability. It’s not inert; it’s simply at peace with its electron configuration. And isn’t that something we can all relate to? That feeling of finally being settled, of having found your equilibrium, is incredibly powerful.
In our fast-paced, always-on world, it’s easy to feel like we're constantly chasing more electrons, more achievements, more connections, without ever quite reaching that satisfying completeness. But maybe, just maybe, we can learn a thing or two from nitrogen. Perhaps true strength isn't in constant striving, but in finding that inner stability, that contentedness that comes from being exactly where you need to be, with what you have. It's the ultimate low-key power move, and it’s happening all around us, from the air we breathe to the very essence of life itself.
