What Happens To The Protons Received From Nadh In Bacteria

So, you know how we all need energy to, like, live? Yeah, bacteria are totally the same. Except they're way more hardcore about it, and their whole energy-making process is a little less "cup of coffee, maybe a bagel" and a lot more "intense biochemical rave." Today, we're diving into something super specific, but honestly, it's pretty cool. We're talking about what happens to all those protons that NADH hands over in bacteria. Big question, right? Let's spill the (proton-y) tea!
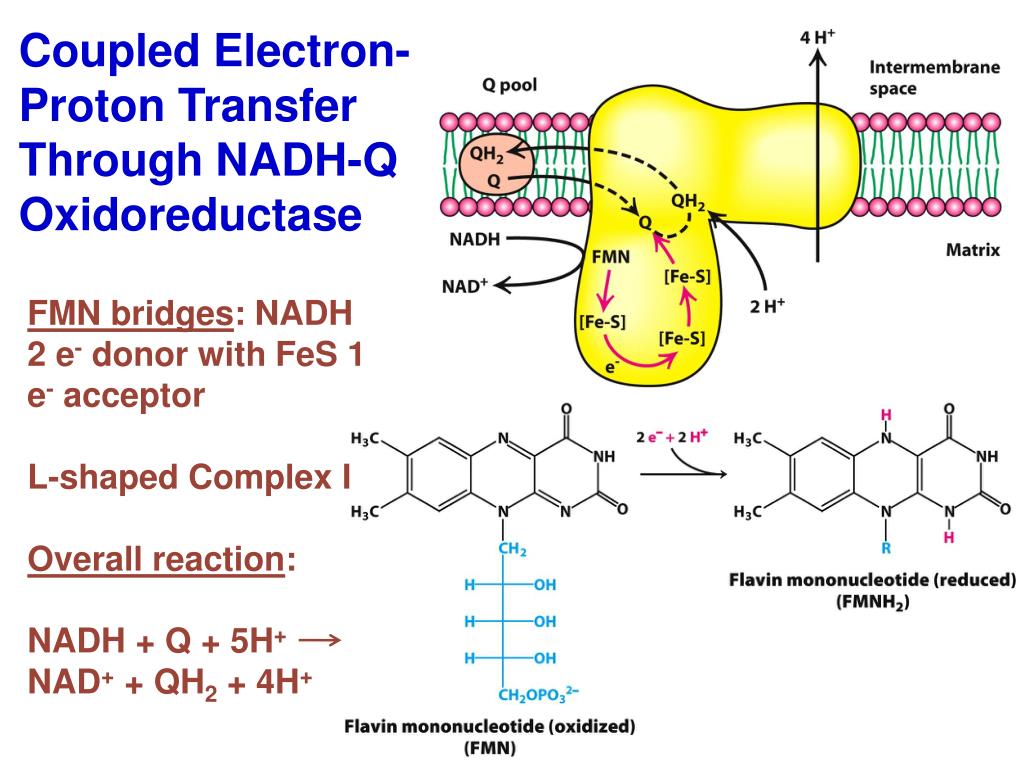
First off, who's NADH? Think of NADH as this little rechargeable battery, but for electrons. It picks up a couple of electrons and a hydrogen ion – that's a proton for us sciencey folks – during, you know, the food-breakdown party called cellular respiration. It's like, "Here you go, world! Take these energy-packed things!" It’s basically carrying around precious cargo, ready to deliver it for some serious energy production. Pretty important job, wouldn't you say?
Now, in us humans and other fancy eukaryotes (that's just a fancy word for anything with a proper nucleus, like your dog or a particularly well-organized mushroom), these protons get tossed into the mitochondria. You know, the "powerhouse of the cell"? Totally lives up to its name. They then get used in this epic chain reaction called the electron transport chain. It’s like a microscopic waterslide of energy, and the protons are the water pushing the ride.
But here's where it gets interesting. Bacteria? They don't have mitochondria. Nope, no fancy internal compartments for them. So, where do all those precious protons from NADH end up going? Do they just, like, chill in the cytoplasm? Get thrown out the back door? Nah, they're way too smart for that. They've got their own, equally effective, system.
The Bacterial Proton Pump Party
Instead of a dedicated organelle, bacteria use their cell membrane. This is like their entire operating system, and it’s where all the magic happens. Think of the cell membrane as this super-thin, super-important barrier. It separates the inside of the cell from the outside world. And it’s not just a wall; it’s a bustling highway of proteins and other molecules doing important work.
When NADH gives up its protons, they don't just float around. Oh no. They get actively pumped. Yeah, pumped! It's like a bouncer at a club, but instead of kicking people out, it's pushing protons out of the cell. This happens across that trusty cell membrane.
This whole process is powered by enzymes, these little biological machines. They take the energy from the electrons NADH delivered and use it to, you guessed it, shove those protons to the other side. It’s a one-way ticket to the outside!
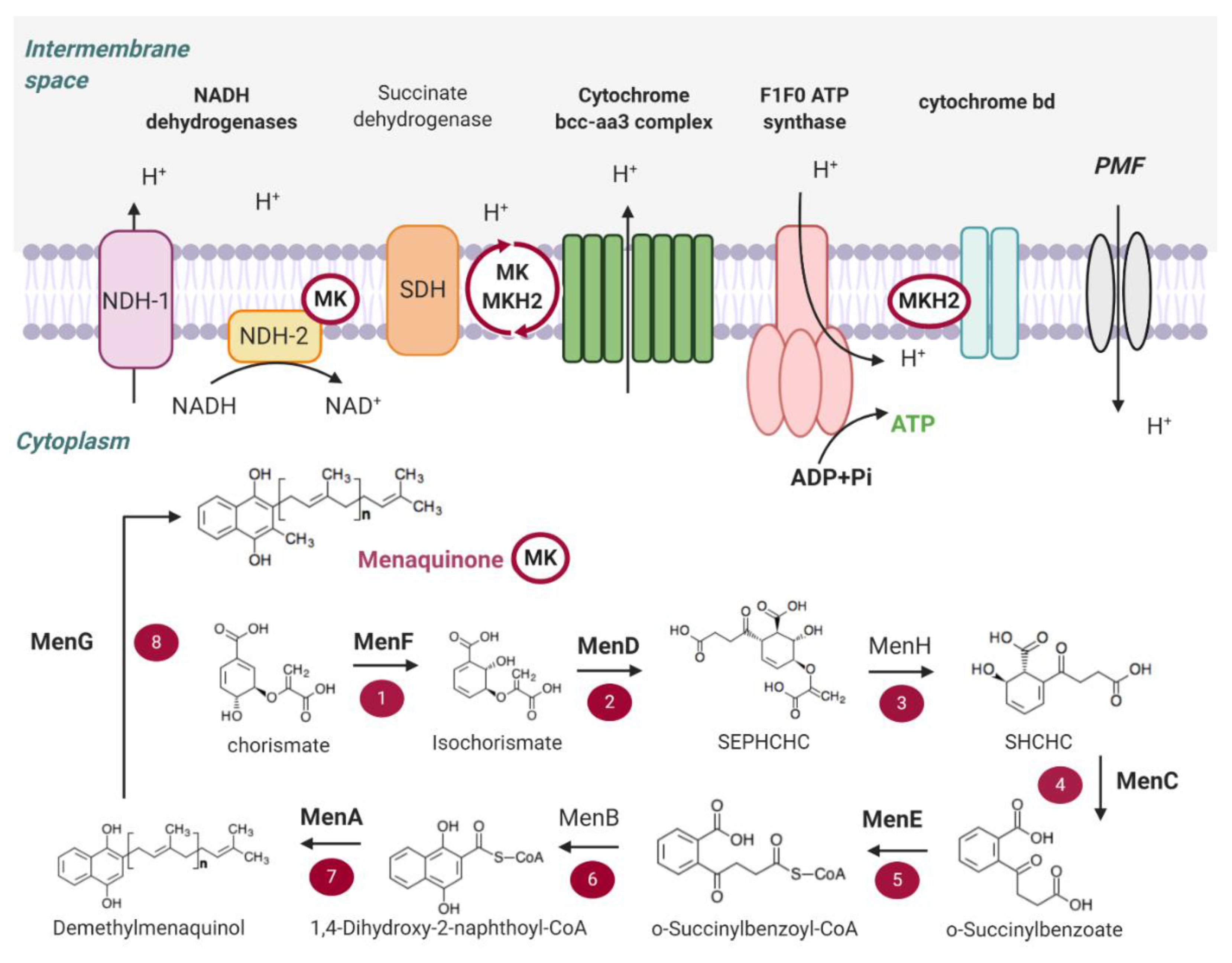
So, what's the point of all this outbound proton traffic? Why would a bacterium want to make its outside all proton-y?
The Proton-Motive Force: It's All About the Gradient!
This is where things get really clever. By pumping protons out, the bacteria create a difference. A big difference. On the outside of the cell, you end up with a higher concentration of protons. Think of it like a crowded party room (inside) versus a quiet hallway (outside). The protons are like the partygoers, and they want to get back to the less crowded space.
This difference in proton concentration, and also a difference in electrical charge (because protons are positive, duh!), creates something called the proton-motive force. Isn't that a cool name? Proton-motive force. It sounds like it could be a superhero move. "Fear not, for the proton-motive force is here!"
This proton-motive force is basically stored energy. It's like a coiled-up spring, or a dam holding back a huge amount of water. All those protons are just waiting for an opportunity to flow back into the cell.
ATP Synthase: The Tiny Energy Factory
And guess what? They get their chance! Bacteria have these amazing molecular machines embedded in their cell membrane called ATP synthase. You might have heard of ATP – that's adenosine triphosphate, the universal energy currency of all life. It's like the cash of the cellular world. Bacteria absolutely need it to do anything and everything.
ATP synthase is like a tiny turbine or a water wheel. As the protons from the outside, driven by the proton-motive force, rush back into the cell through this enzyme, they spin a part of ATP synthase. It’s like a little mini-generator!
And this spinning action? It’s what powers the synthesis of ATP. Boom! Energy made. All from those protons that NADH brought over and then were pumped out. It’s a beautiful, efficient cycle.
So, the protons don't just disappear. They’re part of a carefully orchestrated symphony of molecular events. NADH provides the initial energy boost, the protons get shipped out to build up a charge, and then they flow back through ATP synthase to create the ATP that keeps the whole bacterial operation running.
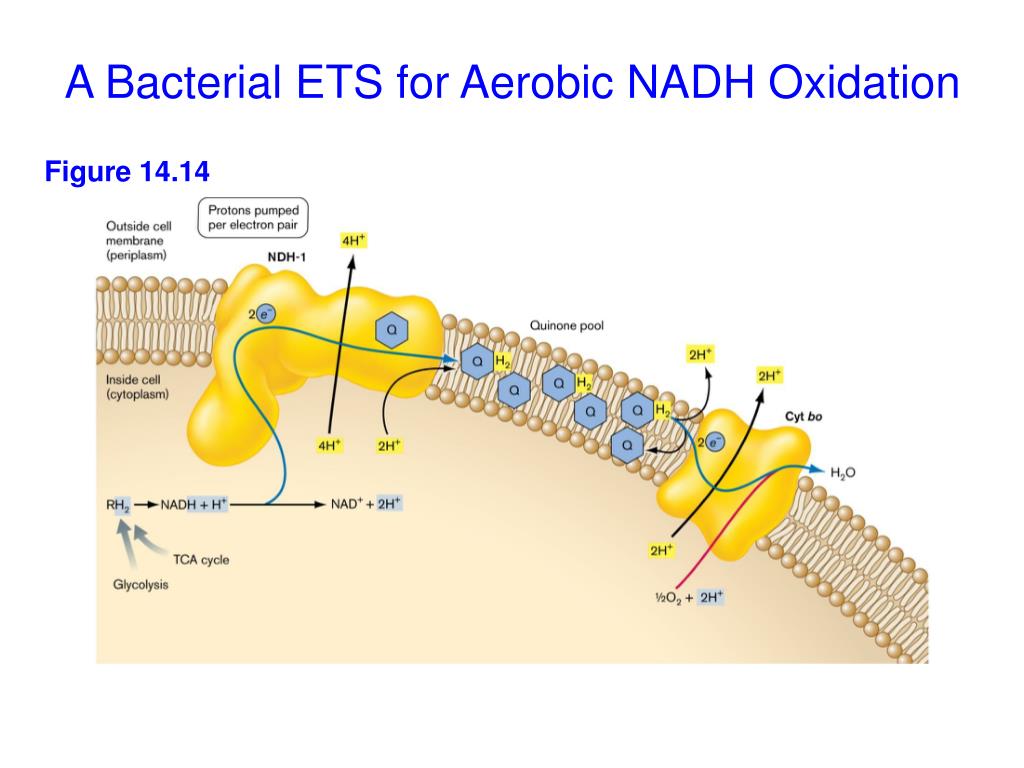
The Electron Transport Chain in Bacteria: Same Idea, Different Location
Now, you might be wondering, "What about the electron transport chain itself?" It's still there, folks! In bacteria, the electron transport chain is also located in the cell membrane. It's a series of protein complexes that pass those electrons from NADH along. As the electrons get passed, energy is released. And what do they do with that released energy? You guessed it – they use it to pump those protons across the membrane!
So, it's not like they just randomly toss protons around. It's a very specific, sequential process. NADH hands off electrons to the chain, the chain does its thing, and that powers the proton pumps. It's all interconnected. You can't have one without the other.
Think of it like a factory assembly line. NADH is the raw material. The electron transport chain is the first few stations, processing the material. The proton pumps are the next stations, using the processed material to build up a reserve. And ATP synthase? That's the final station, where the finished product – ATP – is made.
What if something goes wrong? (Spoiler: It's usually bad for the bacteria.)
What happens if this proton-pumping system gets messed up? Well, the bacteria would have a really hard time making ATP. If you can't make energy, you can't do much. You can't grow, you can't reproduce, you can't even move (if you’re one of the mobile types).

Imagine trying to run a city without electricity. Everything just grinds to a halt. Same for bacteria. This is actually why some antibiotics work. They target specific parts of this energy production system in bacteria, effectively shutting down their power supply. Pretty neat, if a bit grim for the bacteria involved.
Different Bacteria, Different Systems?
While the basic principle of proton pumping and the proton-motive force is pretty universal for bacteria that do respiration, there can be some variations. Different species might have slightly different versions of the electron transport chain or ATP synthase. Some might use different molecules to accept the electrons at the very end of the chain. But the core idea of using those protons to generate energy? That's a pretty solid bet.
Some bacteria, especially those living in oxygen-free environments (anaerobic bacteria), have even more complex ways of doing things, sometimes using things like sulfate or nitrate instead of oxygen at the end of the chain. But even then, the proton gradient is often the key to unlocking that ATP-generating power.
The Big Takeaway: Protons are Key!
So, to sum it all up, the protons that NADH receives (or rather, donates, after picking up electrons) in bacteria are crucial players. They don't just go anywhere. They are actively pumped out of the cell, creating a vital proton-motive force across the cell membrane. This force then drives the incredible ATP synthase enzyme, which spins like a tiny dynamo to produce the ATP bacteria need to survive and thrive.
It's a testament to the elegance and efficiency of life at the microscopic level. No mitochondria? No problem! Bacteria have figured out their own ingenious way to harness energy, and it all hinges on managing those tiny, energetic protons. Pretty cool, right? Next time you think about energy, remember those hardworking bacteria and their proton-powered ATP factories!
