What Happens To Sucrase When It Binds To Sucrose

So, you’re sipping on your sweet tea, right? Or maybe munching on a cookie? Yum! Ever wonder what’s really going on in your body to make all that sugary goodness digestible? It’s not just magic, though it kinda feels like it sometimes. It’s all about tiny molecular matchmakers, and one of the coolest is called sucrase. Ever heard of it? Probably not, unless you're into biochemistry for fun (no judgment!).
But seriously, this little enzyme is a superhero in disguise. Its main gig? Breaking down sucrose. You know, that fancy word for table sugar? Yep, the stuff you sprinkle on your cereal or stir into your coffee. Sucrose is a disaccharide, which basically means it’s made of two smaller sugar units stuck together. Think of it like a LEGO brick set that's snapped together. Too big to be useful on its own for your body’s energy needs, you see.
And that’s where our star player, sucrase, swoops in. What a name, right? Sucrase. Sounds like it’s designed for the job. And it totally is! It’s like the perfect key for the sucrose lock. But what exactly happens when this dynamic duo, sucrase and sucrose, decide to get together? It's a whole process, and honestly, it's kind of adorable when you think about it.
Imagine sucrase as this tiny, highly specific worker. It’s not just grabbing any sugar molecule; it’s got a very particular target: sucrose. It's like a bouncer at a super exclusive club, only letting in the right VIPs. And sucrose, well, it’s that VIP. It’s got a specific shape, a certain vibe, that only sucrase recognizes. Without that perfect fit, nothing happens. No breakdown, no energy boost, nada.
So, sucrase is chilling, doing its enzyme thing. It's got a special spot on its structure, a place called the active site. Think of the active site as the enzyme’s handshake zone, or maybe its tiny little mouth. It’s shaped just so to welcome sucrose. It’s all about that precise molecular shape. Nature is pretty amazing with these details, isn't it?
When a sucrose molecule drifts by – and believe me, in your digestive tract, there are tons of them floating around – sucrase is ready. It’s like a magnet, but a really, really specific magnet. The sucrose molecule, with its own unique 3D shape, glides right into this active site. Voila! The binding happens.
This isn't just a casual hug, mind you. It's a tight, snug fit. They lock in together, like two puzzle pieces that finally found their home. This temporary union is called an enzyme-substrate complex. Pretty fancy, right? Substrate? That’s just biochemistry talk for the molecule the enzyme works on. So, sucrose is the substrate for sucrase. See, it's not so scary when you break it down!
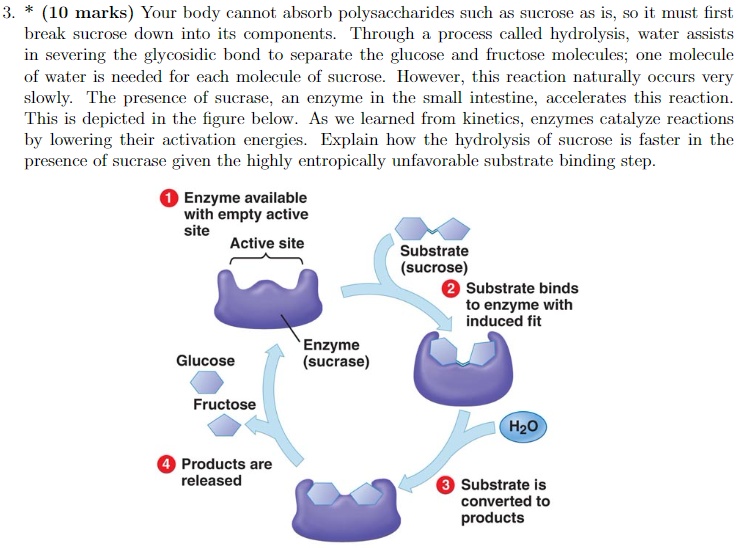
Once they’re bound, it’s like the whole world for them narrows down to just this interaction. The active site of sucrase is now cradling the sucrose molecule. And within this cozy little complex, the magic truly begins. Sucrase isn't just holding sucrose; it's actively changing it.
How does it do that? Well, the active site has some special chemical groups, like little chemical tools, that are perfectly positioned to interact with the bonds holding sucrose together. Sucrose, as we said, is made of two simpler sugars: glucose and fructose. They're linked by a special bond called a glycosidic bond. Think of it as the glue holding the two LEGO bricks together. This bond is strong, but not strong enough to resist a determined sucrase.
Inside the active site, sucrase uses its chemical tools to weaken and break this glycosidic bond. It's like applying just the right amount of pressure, or maybe a little chemical nudge, to snap that glue. The enzyme provides the perfect environment for this reaction to occur. It might even involve water molecules playing a little role, helping to pry the bond apart. This whole process is called hydrolysis. Sounds complicated, but it just means "splitting with water." Super helpful, right?
So, the sucrase is busy doing its thing, and snap! The glycosidic bond is broken. What was once a sucrose molecule is now two separate, simpler sugar molecules: one glucose and one fructose. They’re like the two LEGO bricks that have been successfully separated and are now ready to be used in other creations – or, in this case, to be absorbed into your bloodstream.
And here’s the really cool part: after it’s done its job, after it’s facilitated the breakdown of sucrose, sucrase lets go. It doesn't get all clingy or stuck to the products. Nope! Once the glucose and fructose are free, they detach from the active site. The enzyme is now free and ready to go find another sucrose molecule to work its magic on. It’s like a delivery person dropping off a package and then heading out for the next delivery. No strings attached!

This catalytic activity – that’s the fancy term for an enzyme speeding up a chemical reaction – is what makes enzymes so incredibly important. They speed things up dramatically. Without sucrase, breaking down sucrose would take ages, if it happened at all. Your body needs these reactions to happen quickly and efficiently so you can get the energy you need to, you know, live and digest cookies. Can you imagine a world without cookie digestion? Tragic.
The specificity of sucrase is also key. It only breaks down sucrose. It doesn't mess with lactose (that's milk sugar, a whole other story!) or other types of sugars. This highly specific binding ensures that only the correct molecules are acted upon, preventing chaos in your metabolic pathways. Imagine if sucrase just went around breaking down any sugar it found? Your blood sugar levels would be all over the place! Not a good look.
So, to recap, when sucrose bumps into sucrase, it's like they're introduced. Sucrose fits perfectly into sucrase's active site, forming a temporary complex. Inside this little molecular dance party, sucrase uses its chemical wizardry to break the bond holding glucose and fructose together. Then, poof, the two simple sugars are released, and sucrase is free to do it all over again. It's a beautiful, efficient, and totally essential process.
Think of it as a perfect partnership. Sucrose provides the material to be broken down, and sucrase provides the specialized tools and the perfect environment to get the job done. It’s a bit like a chef and a specific knife. The chef (sucrase) uses the knife (active site) to chop the ingredients (sucrose) into smaller, more manageable pieces (glucose and fructose) that can then be used in the recipe (your body’s energy needs).
And all this happens in your small intestine, by the way. That’s where most of the magic of digestion really goes down. The lining of your small intestine is covered in these tiny finger-like projections called villi, and on the surface of those villi are even tinier projections called microvilli. This is where you'll find a whole bunch of enzymes, including sucrase, all lined up, ready to do their work.

It's a really busy place, your small intestine. Think of it as a bustling molecular factory floor. And sucrase is just one of the many highly specialized workers on that floor, each with its own specific task. It's an amazing feat of biological engineering, really. The precision involved is just mind-blowing when you stop to think about it.
So, next time you enjoy something sweet, give a little nod to sucrase. It’s working hard behind the scenes, making sure you can actually enjoy that sugary treat. It’s a small enzyme with a huge job, and it does it with remarkable efficiency and specificity. Pretty neat, huh?
And what happens if you don't have enough sucrase? Well, that's where things can get a little… gassy. Yep, undigested sucrose can hang around and get fermented by bacteria in your gut, leading to all sorts of discomfort. So, you really want your sucrase to be on its game! It's not just about enjoying dessert; it's about proper digestion and well-being. Who knew enzymes could be so critical?
It’s a fascinating glimpse into the world of molecular biology, a world where tiny molecules interact in complex ways to keep us alive and functioning. The binding of sucrase to sucrose is a fundamental step in energy metabolism, and understanding it helps us appreciate the intricate machinery that makes our bodies work. So, yeah, next time you're sweetening your tea, remember the unsung hero, sucrase, and its crucial role in making it all possible.
It's a constant cycle of binding, breaking, and releasing. Sucrase doesn't get tired; it's designed for endurance. It's like a tiny, perpetual motion machine for sugar breakdown. And thank goodness for that, because without it, our diets would be a lot less enjoyable, and our energy levels would likely be a lot lower. So, cheers to sucrase, the ultimate sugar-buster!

The interaction isn't just about breaking bonds; it's also about how those bonds are broken. The active site of sucrase has specific amino acid residues that participate in the chemical reaction. They might donate or accept protons, or stabilize the transition state of the reaction, making it easier for the glycosidic bond to break. It’s like the enzyme provides the perfect chemical toolkit and the right atmosphere for the reaction to go off without a hitch. Truly a masterclass in molecular catalysis!
And the energy involved? Enzymes like sucrase lower the activation energy required for a reaction to occur. This is the energy barrier that needs to be overcome for the reaction to start. By lowering this barrier, sucrase allows the breakdown of sucrose to happen at body temperature and at a pace that’s useful for us. Without this lowering of activation energy, the reaction would be so slow it would be practically non-existent in biological terms. It's like trying to push a boulder up a very steep hill versus a gentle slope. Sucrase makes the slope gentle.
So, when sucrose binds to sucrase, it's not just a physical connection. It's an energetic and chemical transformation. The enzyme changes its shape slightly upon binding to the substrate, a phenomenon known as induced fit. This conformational change further optimizes the fit and brings the catalytic groups of the enzyme into the perfect position to perform the chemical reaction. It's like a glove molding around a hand – a perfect, dynamic embrace.
This dance of induced fit and catalysis is what makes enzymes so incredibly effective. And sucrase, in its role of breaking down sucrose, is a perfect example of this fundamental biological process. It’s a testament to the elegance and efficiency of nature’s design. From the initial recognition and binding to the catalytic breakdown and release of products, the entire process is a finely tuned symphony of molecular interactions, all happening in your gut, so you can enjoy that sweet treat.
It's a cycle that repeats over and over again, a tireless effort to unlock the energy stored within sucrose. Each binding event, each breakdown, contributes to your body's energy supply. So, the next time you hear the word "enzyme," remember sucrase and its amazing ability to team up with sucrose and make your digestive life a whole lot sweeter and more efficient. It’s a small but mighty player in the grand scheme of things!
