What Energy Difference Is Calculated For Axial Vs Equatorial Conformations
.png)
Hey there, curious minds! Ever thought about the tiny, invisible dances happening inside molecules? You know, those super-tiny building blocks of everything around us? Well, buckle up, because we're about to dive into a topic that sounds a bit science-y but is actually as fun and fascinating as figuring out the best way to arrange your furniture to maximize coziness. Today, we're chatting about energy differences and why a molecule might prefer to stick its "arms" out sideways or up and down. Intriguing, right?
Imagine a molecule like a tiny, flexible stick figure. It can bend and twist, and sometimes, it can even flip its shape! This is where the idea of conformations comes in. Think of it like a yoga pose for molecules. They can twist into different positions, and these different positions are called conformations. For some molecules, especially those with rings (like the super common cyclohexane, which is shaped like a hexagon – how neat is that?!), these yoga poses come in a few distinct flavors.
The two big stars of the show for these ring molecules are the axial and equatorial conformations. Now, let's picture our little hexagon molecule. If we draw imaginary lines going straight up and down through the center of the ring, any "arms" sticking out along those lines are in the axial position. Think of it like someone standing perfectly straight with their arms rigidly by their sides. It’s a very… upright position.
On the other hand, the equatorial position is like the molecule saying, "Let's chill!" The "arms" here are sticking out more sideways, almost parallel to the plane of the ring itself. Imagine your arms outstretched at your sides, relaxed. It’s a much more spread-out, breezy feel, wouldn't you agree?
Now, here's the super cool part that makes all the difference: energy. Everything in the universe, from you reading this to the tiniest atom, has energy. And just like you feel more energized after a good night's sleep or drained after a long day, molecules have preferred energy states. They tend to gravitate towards the positions that require the least amount of energy to maintain. It's like wanting to be in the most comfortable chair, not one that's wobbly or poking you!
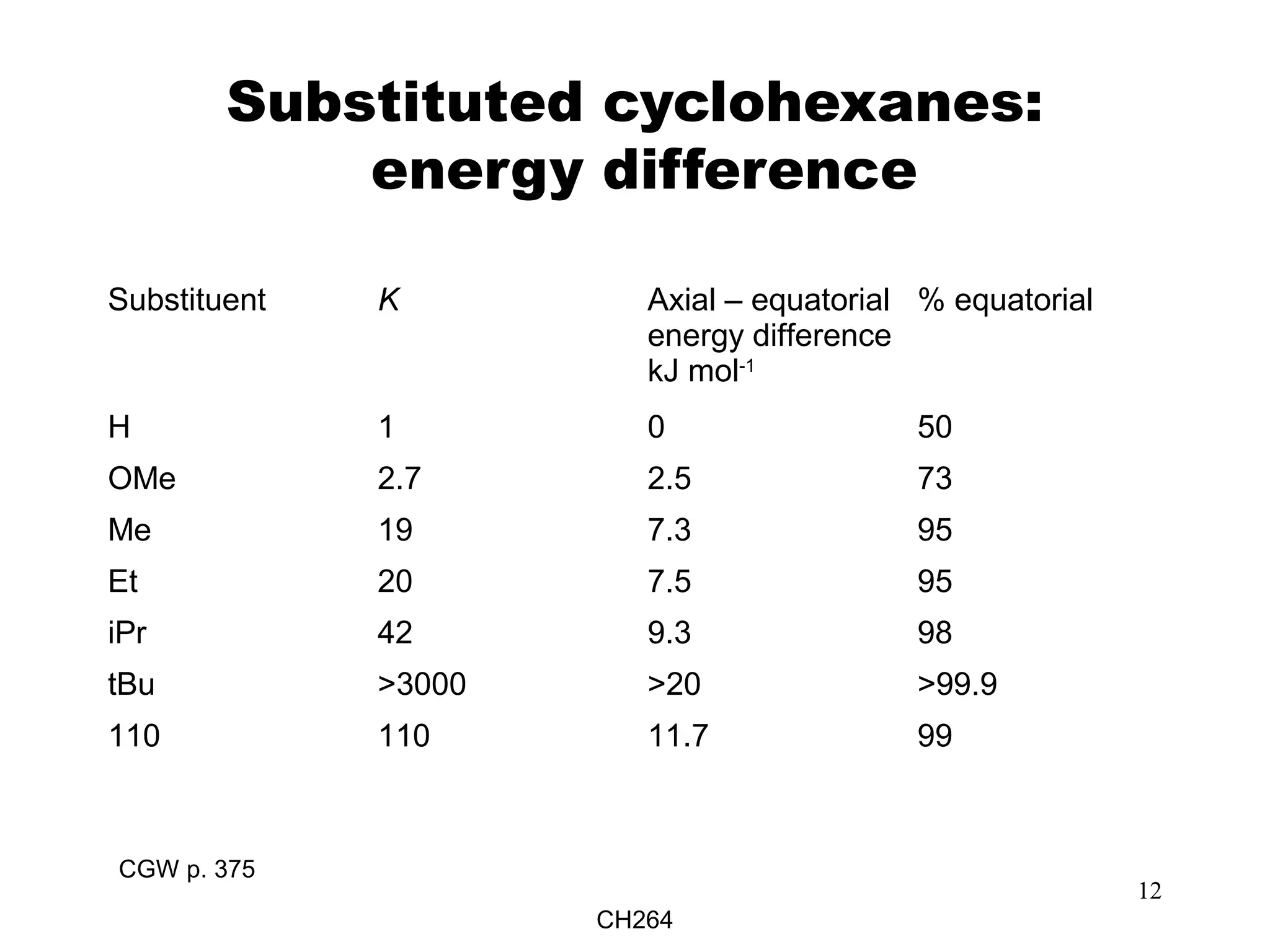
So, when we're talking about axial versus equatorial conformations, what we're really asking is: Which yoga pose is the most comfortable for the molecule? Which one makes it feel the least stressed out and most stable?
The answer, more often than not, is the equatorial position. Why? Well, it's all about a little phenomenon called steric hindrance. Don't let the fancy name scare you! It just means that sometimes, when things are too close together, they bump into each other and cause a bit of unpleasantness. Think of trying to cram three friends onto a small couch – it's going to be a bit of a squeeze, and everyone will feel a little cramped and unhappy.

In the axial position, the "arms" sticking straight up and down can sometimes get a bit too cozy with other axial "arms" on the opposite side of the ring. This close proximity leads to repulsion, which is like a tiny molecular argument. This repulsion means the molecule has to expend more energy to keep those arms in place, making the axial conformation higher in energy. It's like the molecule is constantly saying, "Ouch, get off me!"
The equatorial position, however, offers a lot more breathing room. The "arms" sticking out sideways are generally further away from each other. There's less bumping, less arguing, and therefore, less energy needed to maintain that shape. It’s the molecular equivalent of everyone having their own comfortable space on the couch. This makes the equatorial conformation lower in energy and, consequently, more stable.
So, the energy difference calculated for axial vs. equatorial conformations is essentially a measure of how much "unhappiness" or "stress" the molecule experiences in the axial position compared to the more relaxed equatorial position. Scientists can actually calculate this difference, and it's a really valuable piece of information for understanding how molecules behave.
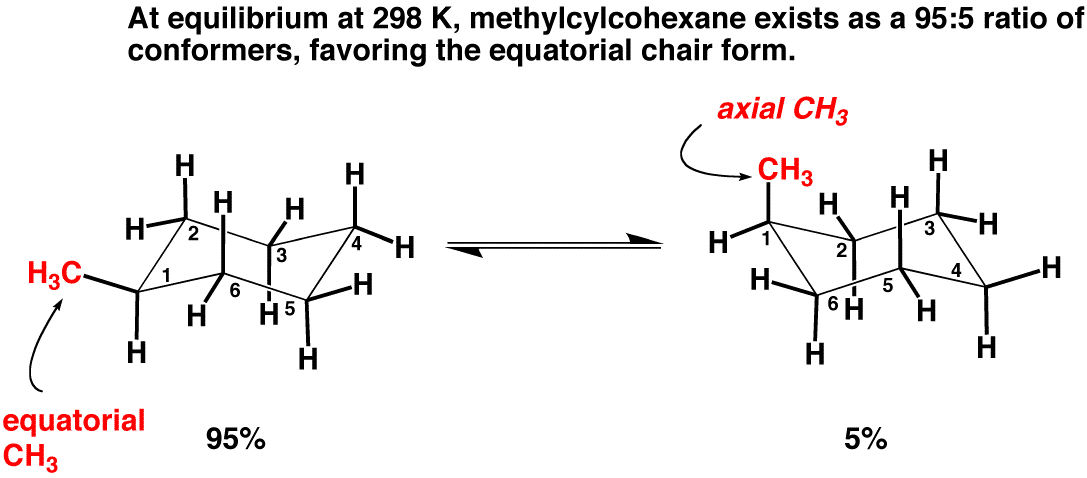
Think about it: if a molecule prefers to be in its equatorial pose, it’s going to spend most of its time there. This preference influences everything from how it reacts with other molecules to how it fits into biological systems (like inside your own body!). It's like knowing your favorite spot on the sofa – you’ll naturally gravitate there!
This might sound like a small detail, but understanding these energy differences is crucial in fields like drug design. If you want to create a medicine that works by latching onto a specific molecule, you need to know which shape that molecule is most likely to be in! It's like knowing whether your friend prefers a handshake or a hug to greet them properly.
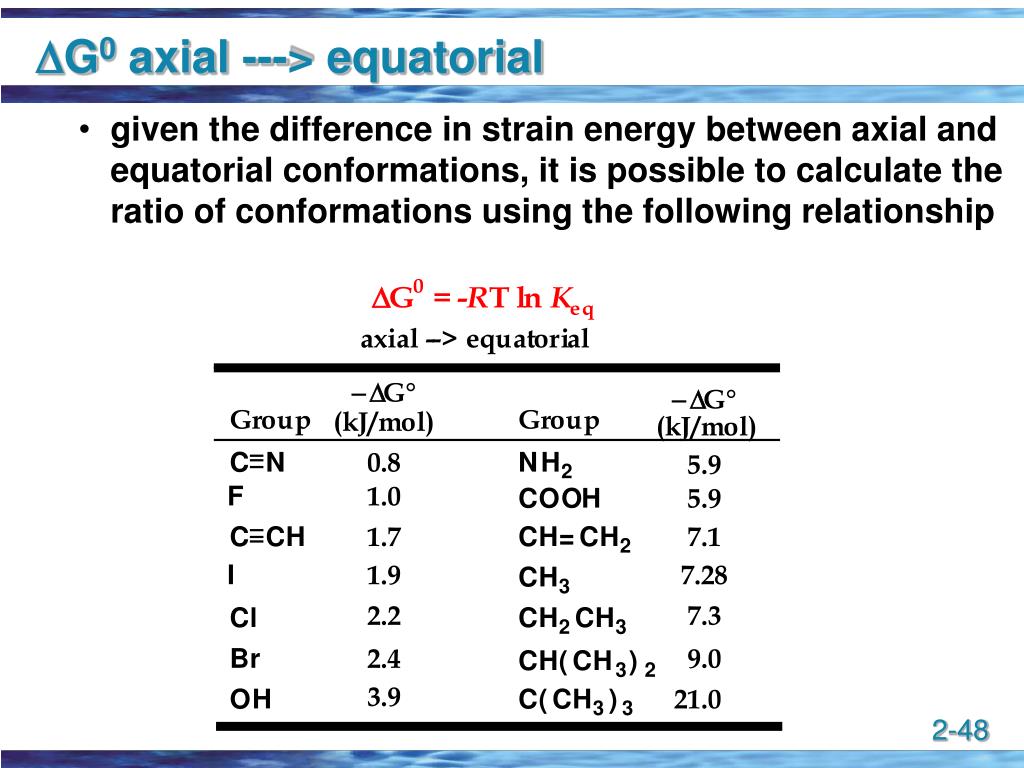
And honestly, isn't it just fun to think about this miniature molecular world? It’s a hidden universe of shapes and preferences, all governed by simple principles of comfort and energy. It's like a constant, silent game of musical chairs, but with molecules!
Learning about these concepts isn't just about memorizing facts; it's about unlocking a deeper appreciation for the intricate beauty of the world around us. It’s about seeing the logic and elegance in the smallest of things. Every molecule is out there, doing its best to exist in its most comfortable state, and we get to be the curious observers.
So, next time you hear about axial and equatorial conformations, don't just think of it as a boring science term. Think of it as a molecular preference, a choice for comfort, and a tiny, energetic dance that shapes our universe. The world of chemistry is brimming with these fascinating puzzles, just waiting for you to explore them. Keep asking questions, keep being curious, and you'll find that even the most complex-sounding topics can lead to moments of pure, inspiring wonder. Go forth and be amazed!
