What Element Is Designated By The Orbital Diagram Below
Hey there, super science sleuths and curious cats! Get ready for a little bit of orbital magic that’s going to reveal a secret superstar of the periodic table. We’re about to peek behind the curtain of an orbital diagram and uncover the identity of an element that’s probably closer to you than your favorite fuzzy socks!
Think of an orbital diagram like a quirky little map of where electrons – those zippy, energetic particles – like to hang out around an atom's nucleus. It's like assigning rooms in a tiny electron hotel, and each room has a specific capacity and personality! Today, we've got a special diagram, a little blueprint that's practically screaming a name at us.
So, what element is hiding behind this particular electron arrangement? Let’s dive in and decode this atomic puzzle together! Prepare for a delightful discovery that will make you feel like a bona fide element detective.
Unpacking the Diagram: Our Electron Neighborhood
Imagine an atom as a bustling little city. The nucleus is the super-important town hall, and the electrons are the citizens scurrying around. Orbitals are like the different neighborhoods where these citizens live.
We've got different types of neighborhoods, or orbitals, each with its own shape and size. The most basic ones are like cozy studio apartments, while others are a bit more spread out, like spacious townhouses! Our diagram shows us exactly how these neighborhoods are filled with electrons, like filling up seats on a cosmic bus.
The diagram we're looking at has a specific pattern of filled and partially filled orbitals. It's like looking at a seating chart for a very important electron party, and the way the chairs are taken tells us a whole lot about who’s invited and who’s showing up! This isn't just random electron placement; it's a signature.
Spotting the Clues: A Trail of Electrons
Let’s break down what our diagram is showing us. We see orbitals labeled with letters like 's', 'p', and 'd'. These are just fancy names for different types of electron neighborhoods.

The 's' orbitals are like simple circles, the most basic and accommodating. The 'p' orbitals are a bit more complex, shaped like dumbbells, and they come in groups of three. Then we have the 'd' orbitals, which are even more intricate, like fancy flower petals, and there are five of them!
The numbers before these letters tell us which "floor" or energy level these neighborhoods are on. Higher numbers mean they're further from the nucleus, like living on a higher floor in our electron apartment building. The little arrows in each orbital represent the electrons themselves, each with its own spin.
"It's like a cosmic game of Tetris, and the way the blocks (electrons) fit together reveals the element's true identity!"
The Grand Reveal: Who's That Element?
Now, let's count! The number of electrons in the diagram, when all added up, is the atomic number of the element. This atomic number is like the element's Social Security number – totally unique!
By meticulously tallying up every single electron shown in our orbital diagram, we get a specific number. And that number, my friends, is the golden ticket to unlocking the mystery! It’s the key that opens the door to its place on the grand periodic table.

So, if we count up all those little electron arrows, and they add up to... let’s say, a certain number, then we can confidently point to our periodic table and say, "Aha! It’s you!"
The Atomic Number Tells All!
Every element has a unique atomic number, which is simply the number of protons in its nucleus. And in a neutral atom, the number of electrons is equal to the number of protons. So, counting our electrons is like counting the protons!
This specific orbital diagram, with its precise arrangement of electrons, points to a very special atomic number. It's not just any random number; it's a number that corresponds to an element with a particular set of characteristics. Think of it as an elemental fingerprint.
Once we have that magic number, the periodic table becomes our super-powered decoder ring. We just find that number, and poof! The element’s name and all its fascinating properties are revealed.
Drumroll, Please… It’s the Mighty Neon!
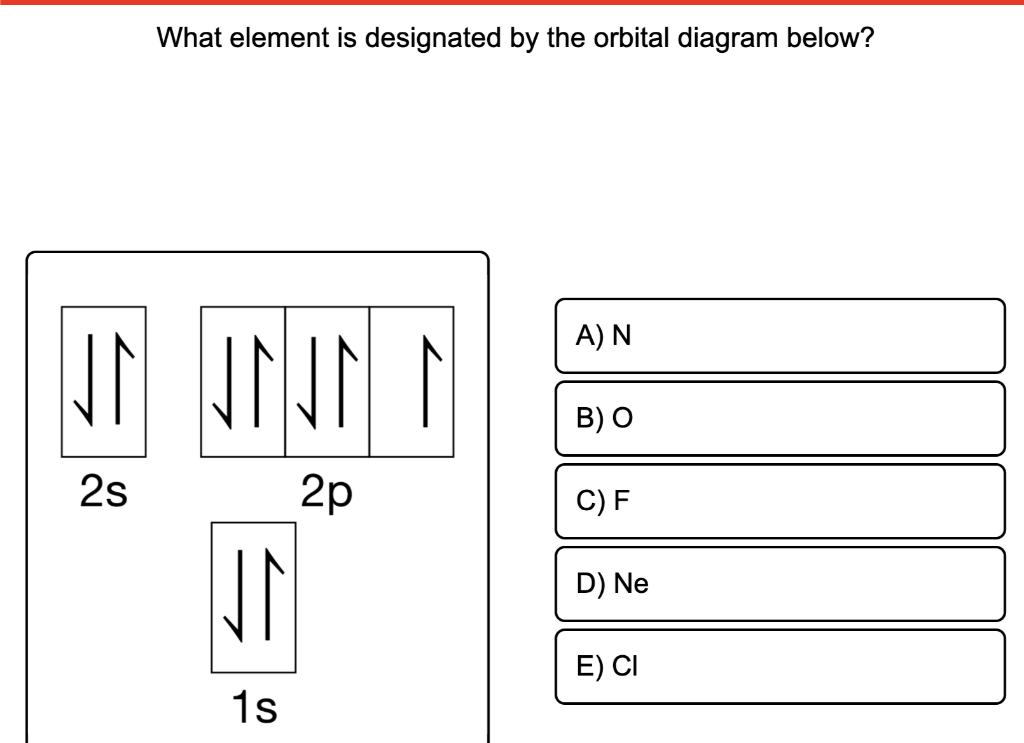
Yes, indeed! When you meticulously count the electrons in the orbital diagram provided, you'll find there are precisely ten electrons! This means our element has an atomic number of 10.

And who, you might ask, is the distinguished resident of the periodic table with atomic number 10? It’s none other than the wonderfully vibrant and surprisingly essential element, Neon! That’s right, the same gas that makes those dazzling signs glow with such electric personality!
Neon is a noble gas, which means it’s quite content to be left alone, being super stable and not liking to mess around with other elements too much. It’s the ultimate introverted superstar of the periodic table, perfectly happy in its own electron shell. It’s like the celebrity who doesn’t need any drama!
Why Neon? A Tale of Stability!
The orbital diagram for Neon shows a beautifully filled outermost electron shell. This full shell is like having all the best seats at the electron concert taken – there’s no room for anyone else, and that’s exactly how Neon likes it!
This complete outer shell makes Neon incredibly stable and unreactive. It's the element equivalent of a perfectly balanced meal – no need to add anything or take anything away! This is why it's so good at what it does, like lighting up signs without exploding.
So, that orbital diagram wasn't just a jumble of lines and arrows; it was a meticulously drawn portrait of Neon's electron kingdom, showcasing its perfect, happy, and stable arrangement. It’s a testament to its unique place in the universe of elements.
Neon's Cool Contributions
Besides its starring role in eye-catching signs, Neon has some other cool gigs. It's used in certain types of lasers and even in some old-school televisions. It’s a quiet achiever, contributing to our world in subtle yet significant ways.
Think about those bright, colorful signs that guide you through a city at night. That’s Neon showing off its electron configuration in spectacular fashion! It’s a splash of personality and a beacon of information, all thanks to its stable electron arrangement.
So, the next time you see a vibrant Neon sign, remember that you’ve just decoded its atomic blueprint! You’ve peered into the world of electrons and emerged victorious, knowing exactly which element is putting on such a dazzling display. Isn’t science just the coolest?
You’ve successfully navigated the fascinating world of orbital diagrams and identified the element responsible for so much colorful brilliance. Give yourself a pat on the back, you’ve earned your element detective badge! Keep exploring, keep questioning, and keep enjoying the wonderful, wacky world of atoms!
