What Electron Configuration Has The Greatest Stability
So, let’s chat about something kinda cool, right? Like, what makes an atom super chill? What’s its deal? We’re diving into the wild world of electron configurations, and specifically, what makes some of them, well, super stable. Think of it like your favorite comfy couch, or that perfect cup of coffee that just hits the spot. Atoms want that feeling too, you know?
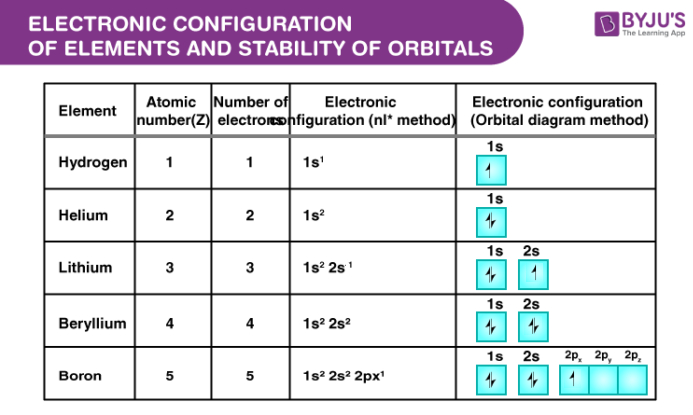
Imagine electrons as little buzzy things zipping around the nucleus. They don't just go anywhere, oh no. They have their favorite spots, their own little energy levels, kind of like apartments in a building. These spots are called orbitals. And the way these electrons fill up these orbitals? That’s the electron configuration. It’s like the building’s floor plan, but for atoms. Pretty neat, huh?
Now, why are we even talking about stability? Because, my friends, stability is the name of the game for atoms. They’re always trying to reach this low-energy, happy state. It’s like us after a long day, right? We crave that chill. Atoms are no different. They’ll do whatever it takes to get there. It’s their whole life’s mission!
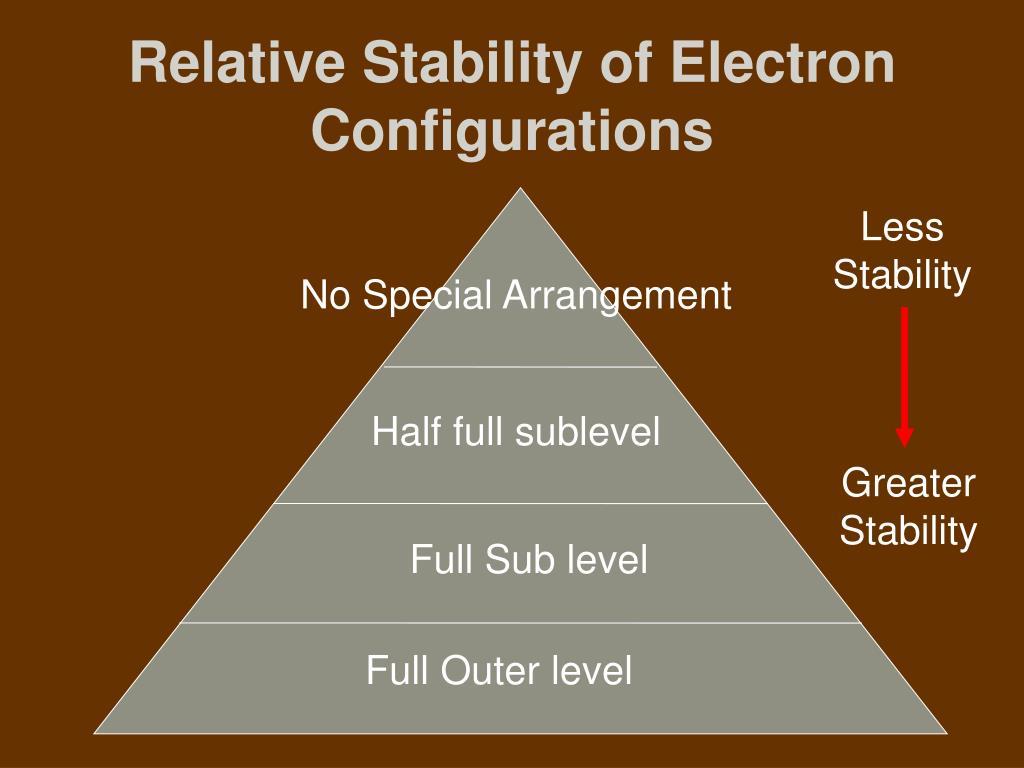
So, what kind of electron configurations are the MVPs of stability? Get ready for this: it’s all about full and half-full orbitals. Mind. Blown. Right?
Full Orbitals: The Ultimate Chill Zone
Let’s break down these full orbitals. Think of an orbital as a tiny room that can hold a maximum of two electrons. These electrons are like roommates, and they gotta have opposite spins. It’s a whole thing. When a room is completely packed, with two electrons snuggled up, that’s a full orbital. And full orbitals? They are the absolute kings and queens of stability. Seriously, they are living their best life.
Why are they so chill? Because having that perfect pair, those opposite spins, creates a kind of balance. It’s like a perfectly weighted seesaw. Everything is just… right. There’s no extra energy hanging around, no desire to grab another electron or ditch one. They’re content. Utterly, completely content. It’s the atomic equivalent of a Sunday morning, no obligations, just pure bliss.
When an atom has its outermost electron shell, you know, the one that’s all about interacting with other atoms, totally filled up, that atom is super unreactive. It’s like, “Nah, I’m good.” It’s got everything it needs, so why bother with all the drama of chemical reactions? It’s the hermit of the periodic table, and it’s perfectly happy in its solitude. Or, it’s like the person who’s just won the lottery and is now retired on a private island. They’ve got it made!

The most famous examples of these super stable, full-shelled atoms are the noble gases. You know, Helium, Neon, Argon, Krypton, Xenon, Radon. These guys? They are the poster children for stability. Their outer shells are completely full, and that’s why they rarely form chemical bonds. They’re already at the top of the mountain, looking down at everyone else. They’re the ultimate cool kids.
So, if you see an atom with a full outermost electron shell, you can practically hear it sighing with contentment. It’s the atomic version of a deep, satisfied breath. Ahhh, peace.
Half-Full Orbitals: The Almost There Heroes
But wait, there’s more! It’s not just about being completely full. There’s another level of stability that’s pretty darn good, too. And that’s the half-full orbital. Think of it like this: if a full orbital is a complete meal, a half-full orbital is like that perfectly portioned appetizer that still leaves you feeling pretty satisfied. It’s not quite the ultimate bliss, but it’s definitely better than being starving!
What’s the deal with half-full? Well, orbitals come in different shapes and sizes, remember? We’ve got the s orbital (super simple, holds two), the p orbitals (three of them, holding six total), the d orbitals (five of them, holding ten total), and so on. When you have exactly half the maximum number of electrons in a subshell, that’s where the magic happens.
For example, a p subshell can hold up to six electrons. If it has three electrons, one in each of the three p orbitals, that’s a half-full p subshell. It’s a state of excellent equilibrium. The electrons are spread out, each occupying its own orbital, and they’re all pointing in the same direction, which is a particularly stable arrangement. It’s like having three friends each in their own room, all agreeing to wear the same color. Harmony!

Why is this half-full situation so stable? It's all about symmetry and minimizing electron-electron repulsion. When electrons are spread out as much as possible, they don't bump into each other as much. It's like a crowded bus versus an empty one. Everyone prefers the empty one, right? Electrons feel the same way. Plus, that uniform distribution of electrons leads to a lower overall energy state.
These half-full configurations aren’t quite as unreactive as the full shells, but they are definitely more stable than orbitals that are only partially filled in a non-uniform way. They’re like the athletes who are always at the top of their game, but might still compete occasionally. They’re strong, they’re balanced, and they’re definitely not looking for trouble.
Elements that have these half-full configurations in their outermost shells are often quite stable, though they might be a bit more prone to certain types of chemical reactions than the noble gases. Think of them as being in a really good, but not completely final, state of calm.
The Exceptions and the Near Misses
Now, chemistry, like life, isn’t always perfectly neat and tidy, is it? There are always a few curveballs. While full and half-full orbitals are the golden rules for stability, sometimes you see some weird stuff happening. Especially with those d and f orbitals.
You know, the d orbitals can hold ten electrons. So, a full d orbital has ten electrons. A half-full one has five. Easy peasy. But sometimes, an element that should have, say, nine electrons in its d subshell, will actually have eight. And an element that should have four, will have five. Huh?

This happens because of a phenomenon called orbital promotion and the energetic stability gained by achieving that half-full or full configuration. Sometimes, an electron from a higher energy level will jump down to fill a d orbital to make it half-full or full. It’s like an electron saying, “You know what? I’m going to make a little sacrifice and move to a slightly lower energy level if it means we all become way more stable as a group.” That’s teamwork for you!
For example, chromium (Cr) and copper (Cu) are famous for this. Chromium’s electron configuration is a bit of a cheat code. Instead of ending up with a 4s² 3d⁴ configuration, it’s actually 4s¹ 3d⁵. That extra electron in the 3d subshell makes it half-full, and that stability is just too good to pass up. It’s like a salesperson who can offer an amazing discount to close the deal – it’s worth the small initial cost.
Copper is similar. Instead of 4s² 3d⁹, it’s 4s¹ 3d¹⁰. That full 3d subshell is incredibly stable, so the atom rearranges itself to achieve it. These are the rebels of the electron configuration world, bending the rules to achieve ultimate chill. They’re the ones who know a shortcut to happiness, and they’re not afraid to take it.
So, while full and half-full are the general superpowers of stability, remember that atoms are clever. They’ll rearrange things a bit to get that extra dose of peace. It's all about minimizing that overall energy. They’re always striving for the lowest possible point on the energy landscape. Think of it as a ball rolling down a hill – it wants to get to the bottom!
Why Does This Matter?
Okay, so we’ve talked about full and half-full orbitals being super chill. But why should you even care? Well, my friends, this is the fundamental reason behind a huge chunk of chemistry! It explains why certain elements bond with each other, why some are so reactive, and why others are as aloof as a cat who’s just had its dinner.
When atoms are not stable, they want to become stable. How do they do that? By interacting with other atoms! They’ll share electrons (covalent bonds), give away or take electrons (ionic bonds), or just hang out with each other in metallic bonds. It’s all in the pursuit of that perfect, stable electron configuration.
Atoms that already have full outer shells (the noble gases) are like the lone wolves of the universe. They don't need anyone else. Atoms that are close to a full shell, like halogens (fluorine, chlorine, etc.) with just one electron missing, are desperate to grab that electron from someone else. That’s why they’re so reactive!
And elements with half-full shells? They’re often involved in forming some pretty strong and stable compounds because they’re already in a pretty good state. They might form bonds to get to that even more stable full state, or they might be content to just be where they are, providing a solid foundation for a molecule.
Understanding electron configurations, and specifically what makes them stable, is like having a secret decoder ring for the entire periodic table. You can start to predict how elements will behave, what kind of compounds they’ll form, and even why some reactions happen and others don’t. It's the bedrock of understanding how matter works!
So, next time you look at an atom, don’t just see a fuzzy cloud of electrons. Think about its inner life. Is it crammed into a full orbital, living the dream? Is it balanced in a half-full state, feeling pretty good? Or is it still trying to figure things out, looking for that perfect arrangement? It’s a whole little drama playing out on a microscopic scale, all driven by the quest for ultimate stability. Pretty wild, right? Pass the coffee, this is fascinating stuff!
