What Effect Does Temperature Have On Reaction Rate Apex

Ever notice how some things just happen faster when they're warm? Like, really warm. Think about your morning coffee. When it's piping hot, it's a whole different beast compared to that lukewarm stuff you forgot about for an hour. That, my friends, is the magic of temperature at play, and we're about to dive into its rather enthusiastic relationship with reaction rates. Prepare yourselves, because this might be an unpopular opinion, but I’m here to tell you that temperature is basically the ultimate hype-man for chemistry.
Let’s be real. When it’s chilly, everything feels like it’s moving in slow motion. Your brain feels fuzzy, your limbs feel heavy, and even the simplest tasks take an age. Imagine trying to bake a cake when your kitchen is practically an icebox. You’d be waiting forever for that dough to rise, wouldn’t you? It's like the ingredients themselves are saying, "Nah, too cold. We're gonna nap." That’s because, on a microscopic level, things are just sluggish. The tiny particles that make up your ingredients are shuffling around like they’re wearing snowshoes. They bump into each other, sure, but not with much gusto. It’s a polite little tap, tap, tap, not the enthusiastic smash we need for a good reaction.
But then, introduce some heat! Suddenly, it’s a party. Those same little particles are zipping around like they’ve had way too much caffeine. They’re bouncing off each other with wild abandon, like toddlers who’ve just discovered a trampoline. BAM! WHIZZ! POP! This is where the action is. The more energy they have, the harder and more often they collide. And when they collide with enough force, poof! A new substance is born. It’s like the difference between a gentle nudge and a full-on hug. One is polite; the other usually gets things moving.
Think about cooking. When you’re searing a steak, that high heat is crucial. You want that beautiful crust, that Maillard reaction, to happen fast. If you just left a raw steak in a lukewarm pan, you’d probably just end up with a sad, grey, slightly cooked piece of meat. Not exactly a culinary triumph. The heat is the catalyst, the encourager, the ultimate instigator of deliciousness. It’s turning a passive ingredient into a flavorful masterpiece in what feels like record time.
And it's not just cooking. Consider your own body. When you’re feeling a bit under the weather and your temperature goes up – that’s your immune system kicking into high gear! Your internal chemical reactions are being boosted to fight off those pesky invaders. It’s your body’s way of saying, "Alright, time to get serious, let's crank up the speed!" Of course, there's a limit. Too hot, and things start to fall apart. Proteins denature, enzymes lose their shape, and suddenly your body's internal processes are going from a speedy race car to a sputtering, broken-down jalopy. It's a delicate dance, but the general rule of thumb? Warmer is usually faster.

This is why chemists love their Bunsen burners. They’re not just for show, you know. That little blue flame is a powerful tool for speeding up reactions that would otherwise take ages. Imagine trying to make a chemical compound by just leaving the ingredients in a beaker on a cold day. You’d need a calendar, maybe even a historical timeline. But with a bit of heat? Suddenly, you’re talking minutes instead of millennia. It’s the difference between watching paint dry and watching a time-lapse video of a flower blooming – both are happening, but one is just way more exciting.
It's like temperature is the ultimate cheat code for chemistry. Want things to go faster? Just turn up the heat. Easy peasy.
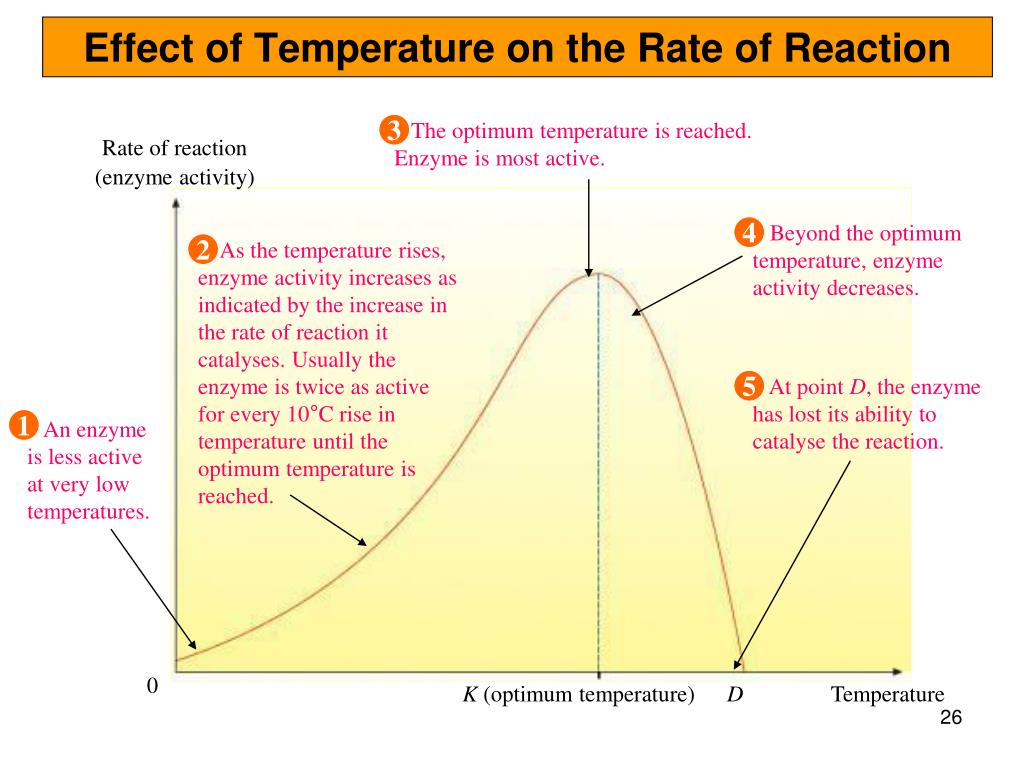
PPT - Chapter 5 - Enzymes PowerPoint Presentation, free download - ID
Now, I know what some of you might be thinking. "But what about freezing things? Doesn't that stop reactions?" And yes, you're right! Freezing is like hitting the pause button. It drastically slows down those energetic particles, making them practically motionless. But that’s not because the cold is actively doing something to speed up the reaction. It’s because the cold is actively preventing the reaction from happening. It's the absence of the hype, the removal of the energy. It’s the opposite of what we're talking about here, which is the acceleration of reactions. Think of it as turning off the music at a party. The party doesn’t stop because the music is bad; it stops because there’s no music at all.
So, the next time you’re waiting for something to happen, and it’s taking an eternity, just remember the power of temperature. It’s not just about feeling comfortable; it’s about influencing the very speed of existence at a molecular level. It’s the unsung hero of rapid transformations, the grand architect of brisk chemical change. While some might prefer a more measured, leisurely pace for their reactions, I’m firmly in the camp that believes a little extra warmth can make all the difference. It’s simple, it’s effective, and frankly, it’s a lot more fun to watch things happen with a bit of pep in their step. So, let's raise a (warm) glass to temperature, the ultimate accelerator!