What Are The Bond Angles In A Typical Carbonyl Group
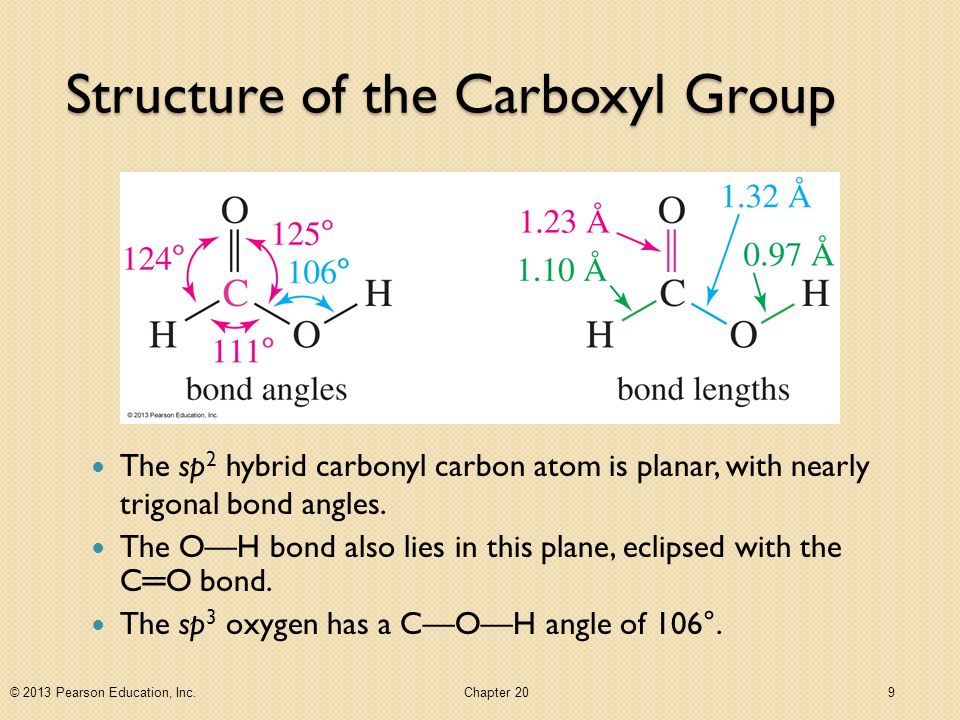
Hey there! So, you’re curious about those little carbon-oxygen double bonds, huh? The ones that pop up everywhere in organic chemistry? Yeah, those carbonyl groups! They’re seriously the rockstars of so many molecules. Think sugars, fats, even the stuff that makes your favorite perfume smell so good. Pretty cool, right?
And the big question on everyone's mind, or at least the minds of us chemistry nerds, is: what are the bond angles in these bad boys? It's like asking the perfect party host how they arrange their seating chart, you know? Gotta get it just right!
So, grab your imaginary coffee, pull up a chair, and let’s spill the tea on these angles. No intimidating textbooks here, promise! Just good old-fashioned chemical gossip.
The Heart of the Matter: The Carbonyl Carbon
Alright, so let’s zero in on the star of the show: the carbonyl carbon. This little guy is bonded to a whole bunch of stuff. Usually, it's got an oxygen atom hooked up with a double bond – the classic carbonyl sign. And then, it’s typically also attached to two other atoms or groups. These could be other carbons, hydrogens, or even other fancy bits and bobs.
Now, here's where the magic (or the science, depending on your mood) happens. Because this carbon is involved in a double bond, it’s a bit of a diva. It doesn't want to just sit there randomly. It wants things to be neat and tidy. You know how some people just need their pencils aligned perfectly? This carbon is like that, but with its atoms.
SP2 Hybridization: The Secret Sauce
The reason for this neatness? It’s all thanks to something called sp2 hybridization. Don't let the fancy name scare you! Think of it like this: the carbon atom is getting ready for a big event. It takes one of its s orbitals and two of its p orbitals and mixes them all up to create three new, perfectly equal hybrid orbitals. It’s like taking three slightly different colored paints and blending them into three identical shades. Voila!
These three sp2 orbitals are positioned as far away from each other as possible to minimize that awkward, squished-together feeling. And where do they end up? In a perfectly flat arrangement, like a perfectly arranged bouquet of flowers. This flat arrangement is called trigonal planar. Isn’t that a fancy word for just… flat? So, the three things attached to the carbonyl carbon are spread out in a plane.
The Famous 120 Degrees!
So, if you have three things arranged in a flat, trigonal planar shape, what do you think the angles between them are? Think of cutting a pizza into three equal slices. Each slice has a nice, even angle at the center, right? That's exactly what we've got here!

The bond angles around that sp2 hybridized carbonyl carbon are all approximately 120 degrees. Yep, 120 degrees! It’s like the chemical world's idea of perfect symmetry. It’s not exactly 120 degrees all the time, of course. Chemistry is rarely that simple, is it? There are always little deviations, like that one friend who’s always a few minutes late to the party.
But 120 degrees is our magic number, our ideal. It’s the geometry that makes everything most stable and happy. Think of it as the Goldilocks zone of bond angles – not too cramped, not too stretched out, just right!
What About That Oxygen Double Bond?
Now, you might be thinking, “But what about the double bond to the oxygen? Doesn't that mess things up?” Great question! It’s like asking if the extra sparkle on a celebrity’s outfit changes their overall silhouette. Not really, but it definitely adds something!
The double bond between the carbon and the oxygen is actually made up of two parts. One is a standard sigma bond, which is like a strong, direct handshake. The other is a pi bond, which is formed by the overlap of leftover p orbitals. This pi bond is a bit more… energetic. It's like a little dance happening above and below the sigma bond.
The pi bond doesn’t really affect the basic arrangement of the atoms directly bonded to the carbon. It’s there, it’s important for reactivity, but the main players in terms of geometry are those three sp2 hybridized orbitals holding hands. So, the 120-degree ideal still holds strong for the atoms directly connected to the carbonyl carbon.
The Varying Players: What Else is Attached?
Okay, so we’ve got our 120-degree baseline. But what happens when the other two things attached to the carbonyl carbon are different? This is where things get a little more interesting, and maybe a tiny bit less perfectly 120. It’s like when you invite a mix of people to a party – sometimes the conversations flow perfectly, and sometimes there are a few awkward silences.
Imagine a simple aldehyde, like formaldehyde. The carbonyl carbon is bonded to an oxygen (double bond) and two hydrogen atoms. All three of those "things" (oxygen and two hydrogens) are roughly equivalent, so you get pretty close to those ideal 120-degree angles. It's a very symmetrical molecule.
Now, let's think about a ketone. Here, the carbonyl carbon is attached to two carbon atoms. For example, acetone. The carbon atoms are a bit bigger and bulkier than hydrogen atoms. They also have their own electron clouds, which can repel each other a little more. This repulsion can cause the angles to be slightly pushed around.
Bulky Groups Mean Bulky Angles
If you have really bulky groups attached to the carbonyl carbon, they’ll want even more space. It's like trying to squeeze three large suitcases into a small car trunk. You have to shift things around to make them fit. These bulkier groups will push the other bonds outwards, making the angles slightly larger than 120 degrees. They’re basically saying, “Excuse me, I need more room!”
Conversely, if you have smaller groups, the angles might be closer to 120, or even slightly less if there's a lot of electron density pulling things in. It’s all about finding that sweet spot of minimal repulsion and maximal stability.
So, while 120 degrees is the target, you might see angles like 118 degrees, 122 degrees, or even slightly more extreme values depending on the specific molecule. It’s a bit like the weather – we expect it to be a certain temperature, but it’s not always exactly that!
The Oxygen Atom's Perspective
What about the oxygen atom itself? Does it have any bond angles? Well, in a carbonyl group, the oxygen is typically double-bonded to the carbon. Oxygen usually likes to be bonded to two things and have two lone pairs of electrons. When it's double-bonded to the carbon, it's using its valence electrons for that strong connection. It doesn't have "bond angles" in the same way the carbon does, because it's not forming multiple distinct single bonds radiating outwards.
However, the oxygen does have its own electron geometry. If you were to consider the electron groups around the oxygen (the double bond counts as one group, and then its lone pairs), it would often adopt a bent geometry, similar to water. But for the purpose of the carbonyl group's shape and the angles between the atoms attached to the carbon, we focus on the trigonal planar arrangement of the carbon and its three neighbors.
The Pi Bond Dance
Let’s circle back to that pi bond for a sec. It's crucial for making the carbonyl group reactive. It’s a higher energy bond, more exposed, and more willing to participate in chemical reactions. Think of it as the energetic younger sibling who’s always up for an adventure.
The plane formed by the sigma bonds (the ones that give us the 120-degree angles) and the pi bond are perpendicular to each other. So, the molecule isn't just flat in one direction; it has a bit of a 3D structure. But for the angles within that planar arrangement, we're sticking with our 120-degree theme.
Why Does This Even Matter?
You might be thinking, “Okay, so the angles are around 120 degrees. Big deal. Why should I care?” Ah, my friend, this is where the real fun begins! These bond angles aren't just pretty numbers; they dictate how molecules behave. They influence everything!
The precise angles affect how molecules interact with each other. Are they going to bump into each other in just the right way to form a new bond? Or are they going to glance off each other like shy strangers at a party?

The 120-degree angles and the trigonal planar shape make the carbonyl group electrophilic at the carbon atom. This means it's a bit of a magnet for electron-rich species (nucleophiles). It's like the carbonyl carbon is saying, “Come here, you electron-rich beauties! I’ve got room for you!” The shape and electron distribution are key to this attraction.
It Affects Reactivity!
So, that slight deviation from 120 degrees? It might seem tiny, but it can have a massive impact on how quickly a reaction happens or which products are formed. It’s like a butterfly flapping its wings in Brazil causing a hurricane in Texas – a small change can have big consequences!
Chemists spend a lot of time studying these angles because understanding them helps us predict and control chemical reactions. We can design new molecules with specific shapes to perform specific jobs, whether it's creating a new drug or a super-strong plastic. It’s all about understanding the geometry, the angles, and how they play together.
In a Nutshell (or a Trigonal Planar Arrangement!)
So, to wrap it all up, in a typical carbonyl group, the carbon atom is sp2 hybridized. This means it likes to arrange the three things attached to it in a flat, trigonal planar geometry. As a result, the bond angles are generally around 120 degrees.
Remember, this is an ideal. Real-world molecules can have slight variations due to the nature of the atoms or groups attached. Bulky groups will push things out, making angles a bit wider. Smaller groups might keep things tighter.
But that 120-degree mark is your go-to answer, your chemical north star for carbonyl bond angles. It’s the fundamental shape that makes these groups so important and so versatile in the world of chemistry. Pretty neat, huh? Now you know the secret behind those super important bonds!
