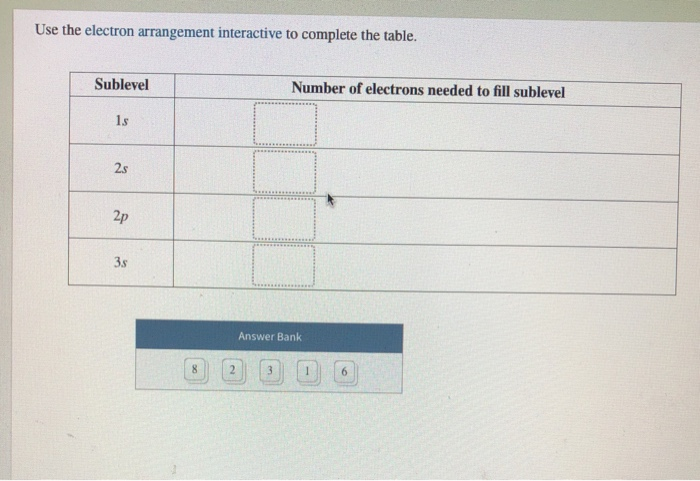
Use The Electron Arrangement Interactive To Complete The Table

Imagine tiny, energetic little characters, all zipping around the center of their own little worlds. We call these characters electrons, and they're basically the lifeblood of everything! Think of them like super enthusiastic puppies that love to play in specific areas around their nucleus (which is like their cozy dog bed). These play areas are called electron shells, and they're arranged in layers, sort of like a multi-story apartment building for these energetic pups.
Now, here's where it gets really fun! We've got this amazing thing called an Electron Arrangement Interactive. Think of it as a digital playground where you can actually see these electron puppies at work. It’s like having a magical window into the heart of atoms, watching them arrange themselves in their designated shells. It’s not just a dry science lesson; it’s more like a fun puzzle game where you’re helping these little guys find their perfect spots.
So, you might be asking, "What's the big deal about where these electron puppies hang out?" Well, it turns out, it's a HUGE deal! The way electrons are arranged in their shells tells us everything about an element. It's like knowing how many toys a puppy has – it gives you a clue about its personality and how it might behave. For example, if an atom has just one electron in its outermost shell, it's probably going to be super eager to share it to make friends, which is how chemical bonds are formed. It's like that one friendly pup always wanting to play fetch with anyone who comes near.
This interactive tool lets you play around with different elements, like famous ones you might have heard of. Take Hydrogen, the simplest of the bunch. It’s got just one electron, and the interactive shows it happily chilling in the first shell. Easy peasy! Then you have something like Helium. This one’s a bit more of a lone wolf, but in a good way. It’s got two electrons, and they’ve both managed to fill up that first shell perfectly. They’re like a perfectly matched pair of socks, all cozy and content. This makes Helium really stable and unlikely to join in any rowdy games, which is why it’s used in balloons that float so cheerfully!
As you move up the elemental ladder, things get more interesting. Take Lithium. It’s got three electrons. The first shell is full with two, and then that third electron has to move up to the next story – the second shell. This little guy is like the enthusiastic newcomer at the party, eager to make an impression. It’s this single electron in the outer shell that makes Lithium so reactive, always looking for a partner to bond with. Imagine it: "Hey, wanna share this outer shell space with me? I've got extra room!"
The fun continues with elements like Carbon, the backbone of so many things we love, including us! Carbon has six electrons. Two fill the first shell, leaving four to arrange themselves in the second shell. This arrangement is like having four eager participants ready for a dance. Carbon is amazing because it can form up to four bonds, creating incredibly complex and diverse molecules. It’s like the ultimate party planner, able to connect with so many different things to create amazing structures!
And what about Oxygen? This life-giving element has eight electrons. Two in the first shell, and then six in the second. It’s like a shell that’s almost full, but not quite. It’s got room for two more electrons. This makes Oxygen very keen to grab those extra electrons from other atoms, which is why it’s so important for breathing and for rust to form on your bike! It’s the ultimate collector, always looking to complete its set.
The Electron Arrangement Interactive is your ticket to exploring these atomic personalities. You’ll see how elements like Neon, with its ten electrons and perfectly filled outer shells, are so incredibly chill. They’re the noble gases, the introverts of the atomic world, content in their own space and rarely bothering to interact. They're the ones who just want to read a book in their quiet corner of the atomic library. Then you have elements like Sodium, which is just like Lithium with one electron in its outermost shell. It’s so eager to get rid of that extra electron, it’s practically throwing it away to become stable, leading to the formation of salt (sodium chloride), that stuff we sprinkle on our fries!
It’s heartwarming to see how these tiny, unseen forces dictate so much of our world. The way these electron puppies play and arrange themselves is the foundation of every chemical reaction, every material, and ultimately, every living thing. So, next time you look at a piece of metal, a puff of smoke, or even take a breath, remember the energetic electron puppies zipping around in their shells. And if you get a chance, jump into the Electron Arrangement Interactive. You might just find yourself with a whole new appreciation for the incredible, tiny universe within everything.
