Use Retrosynthetic Analysis To Suggest A Way To Synthesize 1-phenyl-1-propanol
Hey there, budding chemists and curious minds! Ever looked at a molecule and thought, "How on earth did they make that?!" Well, today we're going to pull back the curtain on a super cool trick called retrosynthetic analysis. Think of it like being a detective, but instead of solving a crime, we're solving a synthesis puzzle. And our target today? A rather handsome fellow called 1-phenyl-1-propanol. Don't let the fancy name scare you, it's basically a fancy way of saying a three-carbon chain with a fancy benzene ring stuck to the first carbon, and a hydroxyl group (that's the 'ol' part, like in alcohol) also chilling on that first carbon. Pretty neat, right?
So, what's the deal with retrosynthetic analysis? Imagine you've just baked an amazing cake, and someone asks you, "How did you make this deliciousness?" You wouldn't just start listing off ingredients randomly. You'd probably think backward: "Well, to get this fluffy cake, I needed flour, eggs, and sugar. To get the eggs, I needed a chicken... and so on." Retrosynthesis is exactly that, but for molecules. We start with our target molecule and work backward, breaking it down into simpler, more readily available starting materials. It's like dismantling a Lego castle piece by piece until you have all the original bricks. Except, you know, way more scientific and with less chance of losing tiny plastic bits under the sofa.
Our star for today, 1-phenyl-1-propanol, looks like this:
Ph-CH(OH)-CH2-CH3
Where 'Ph' is our friendly benzene ring (that's the phenyl part) and the '-CH(OH)-CH2-CH3' is our propanol bit. See? Not so scary when you break it down. Now, let's put our detective hats on and start figuring out how to build this guy.
The Backward Journey Begins!
The core idea of retrosynthesis is to identify disconnections. These are bonds that we can imagine breaking to simplify our molecule. When we break a bond, we often create new functional groups. The trick is to break bonds in ways that lead us to known reactions and starting materials. It’s like finding the “undo” button for chemical reactions.
Let's look at our 1-phenyl-1-propanol. We've got a carbon atom (let's call it C1) that's bonded to a phenyl group, a hydroxyl group, a hydrogen atom, and then the rest of our propyl chain (CH2-CH3). That C1 is a pretty busy bee, isn't it?
One of the most common and powerful retrosynthetic moves involves breaking a carbon-carbon bond that's adjacent to a functional group. In our case, that hydroxyl group is a big hint! It's attached to C1. So, let's think about disconnecting the bond between C1 and the phenyl group. Why this bond? Because phenyl groups are usually pretty stable and often come from readily available starting materials like benzene itself, or perhaps a bromobenzene.
So, if we imagine breaking that bond between C1 and the phenyl ring, what do we get? Well, breaking a bond often implies that the two pieces could have been joined together by a reaction. If we break the C-Ph bond, we could think of the phenyl group having come in as a nucleophile (electron-rich, looking to donate electrons) or an electrophile (electron-poor, looking to accept electrons). Similarly, the other piece, the propyl chain with the -OH, could have been the opposite. This is where things get exciting!

Let's rewind the clock. If we break the C-Ph bond, and we consider that the hydroxyl group is already present on the carbon that was attached to the phenyl, then we might imagine a precursor where the phenyl group was added to a carbonyl group. Think about it: how do we often form C-C bonds next to oxygen-containing functional groups? One of the classic ways is using Grignard reagents or organolithium reagents. These are super handy tools that allow us to add carbon chains to other molecules. And guess what? They often react with carbonyl compounds like aldehydes and ketones!
The Grignard Gadget!
So, let's try this disconnection: we'll break the C-Ph bond. This leaves us with a fragment containing the phenyl group, and another fragment containing the propanol part. If we think about forming this bond, a common way to attach an aryl (phenyl) group to a carbon bearing an alcohol is through the reaction of an aryl Grignard reagent with an aldehyde or ketone. This is a huge clue, folks! This is like finding a footprint at a crime scene!
Let's represent this disconnection with a wavy arrow, signifying our "undo" move:
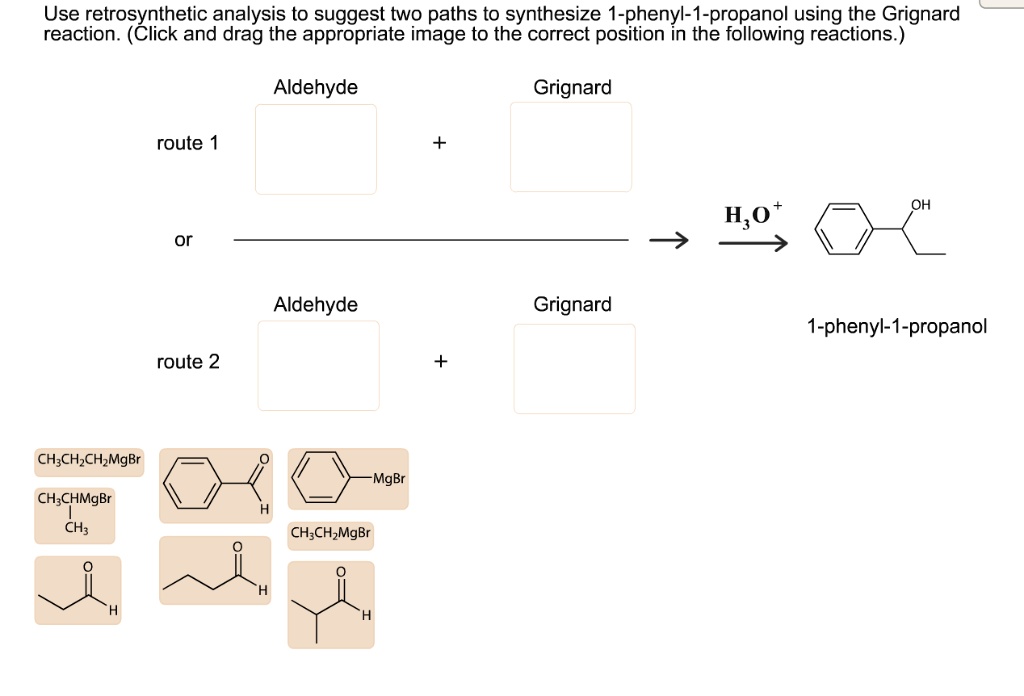
Ph-CH(OH)-CH2-CH3 <= Ph-MgBr + OHC-CH2-CH3
Whoa, what happened there? We took our target molecule and imagined it came from reacting phenylmagnesium bromide (Ph-MgBr), which is a Grignard reagent, with propanal (OHC-CH2-CH3). Propanal is a simple three-carbon aldehyde. See how the oxygen in the hydroxyl group of our target molecule now sits on the carbon that was the carbonyl carbon of the propanal? And the phenyl group from the Grignard reagent has attached itself to that same carbon.
This is a fantastic disconnection because both Ph-MgBr and propanal are relatively easy to make or buy. Phenylmagnesium bromide can be prepared from bromobenzene (which itself comes from benzene – easy peasy) and magnesium metal. Propanal is a common aldehyde. So, we're looking pretty good!

Alternative Paths to Explore (Because Variety is the Spice of Synthesis!)
Now, a good detective doesn't just settle for the first suspect, right? Let's explore some other possibilities. What if we decided to break a different bond? What if we disconnected the bond between C1 (the one with the -OH and Ph) and the next carbon (the CH2)?
Let's visualize this:
Ph-CH(OH)-CH2-CH3 <= Ph-CH(OH)-X + [CH2-CH3]-
Here, 'X' could be a leaving group (like a halide, e.g., Cl, Br, I), and '[CH2-CH3]-' represents an ethyl anion. Now, while ethyl anions are a thing, they're not the easiest to work with directly. This disconnection suggests an SN2-type reaction where an ethyl nucleophile attacks a carbon bearing the phenyl and hydroxyl groups. This feels a bit more complicated than our first idea.
However, we can reframe this. What if we think about creating the C-C bond before adding the hydroxyl group? This is a common strategy: build the carbon skeleton first, then add the functional groups. Let's try disconnecting the bond between C1 and the phenyl group, but this time, let's imagine the carbon chain already has a functional group that can become an alcohol.
Consider this:
Ph-CH(OH)-CH2-CH3 <= Ph-X + [CH3-CH2-C=O]- (or similar electrophile)
This suggests adding the phenyl group to a molecule that will eventually become our alcohol. This is a bit of a stretch. Let's think more practically. What if we disconnect the C1-C2 bond (the one between the carbon with the phenyl and OH, and the next carbon)?
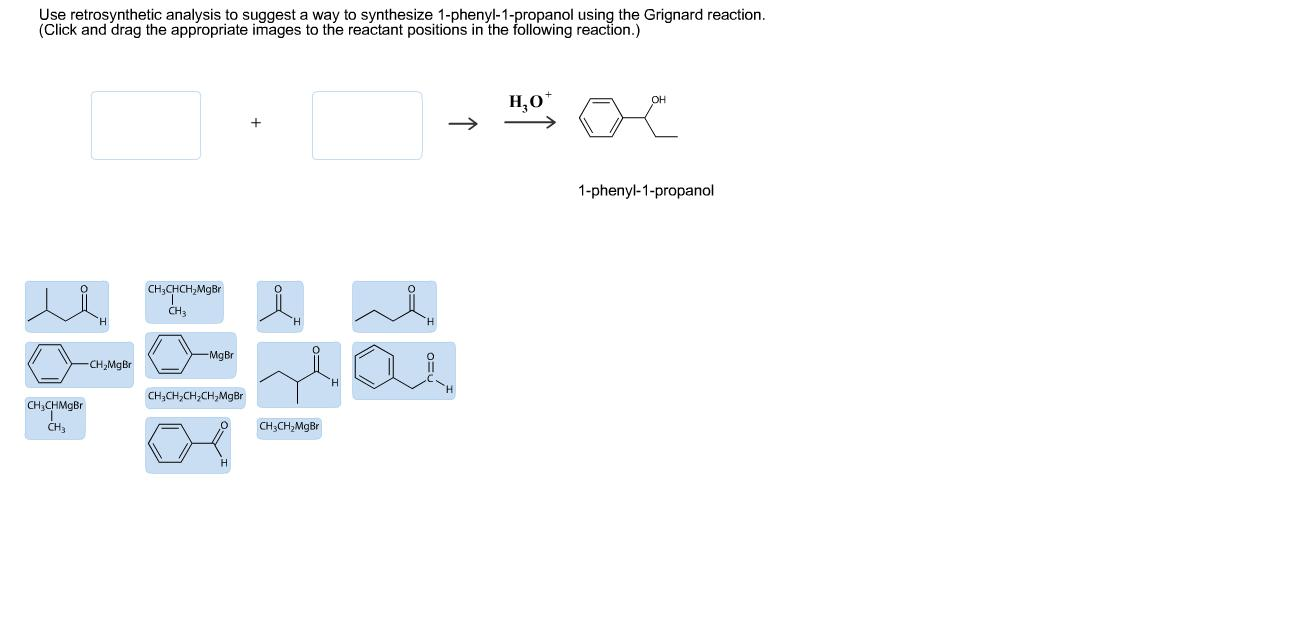
Ph-CH(OH)-CH2-CH3 <= Ph-CHO + CH3-CH2-MgBr
Eureka! This looks very promising, just like our first idea, but with the roles reversed! Here, we're suggesting that our target molecule could be formed by reacting benzaldehyde (Ph-CHO) with ethylmagnesium bromide (CH3-CH2-MgBr). Benzaldehyde is a common aromatic aldehyde, and ethylmagnesium bromide is another readily available Grignard reagent. When the ethyl group from the Grignard reagent attacks the carbonyl carbon of benzaldehyde, and then we do a workup with acid (to protonate the alkoxide intermediate), we get our desired 1-phenyl-1-propanol!
This is a classic example of using Grignard reagents to form secondary alcohols. The beauty of retrosynthesis is that it allows us to see multiple potential pathways, and then we can evaluate which ones are the most practical and efficient.
Evaluating Our Options
So, we have two really strong contenders emerging from our retrosynthetic analysis:
- Phenylmagnesium bromide + Propanal
- Benzaldehyde + Ethylmagnesium bromide
Both pathways involve a Grignard reaction, a workhorse of organic synthesis. Both use readily available starting materials. In a real-world lab setting, a chemist might choose one over the other based on factors like cost, ease of handling, or potential side reactions. But for our purposes, both are perfectly valid and fun ways to think about making 1-phenyl-1-propanol.
Let's briefly walk through the forward synthesis for option 2 (Benzaldehyde + Ethylmagnesium bromide), because it's a great illustration of how it works:
Forward Synthesis Sketch:
- Making the Grignard: Start with bromoethane (CH3-CH2-Br) and react it with magnesium metal in a dry ether solvent (like diethyl ether or THF). This gives you ethylmagnesium bromide (CH3-CH2-MgBr). You have to be careful here, as Grignard reagents are sensitive to water and air. It's like having a pampered celebrity reagent that needs special treatment!
- The Grignard Attack: Add benzaldehyde (Ph-CHO) to your solution of ethylmagnesium bromide. The nucleophilic ethyl group from the Grignard will attack the electrophilic carbonyl carbon of benzaldehyde. This forms an alkoxide intermediate. Think of it as the ethyl group giving the carbonyl carbon a big, welcoming hug.
- Workup: After the reaction is complete, you add a dilute acid (like HCl or H2SO4) to protonate the alkoxide. This step liberates the alcohol and gives you your final product, 1-phenyl-1-propanol! It's like giving the molecule a little pat on the head to finalize its transformation.
And there you have it! A molecule that might have seemed complex at first glance is now within our synthetic reach, all thanks to the magical art of working backward.
Why is This So Cool?
Retrosynthetic analysis isn't just about finding one way to make a molecule. It's about developing a strategic mindset. It teaches you to look at a molecule and break it down into its fundamental building blocks, understanding how different functional groups can be formed and how carbon-carbon bonds can be created. It's like learning the language of chemistry at a deeper level.
When you can perform retrosynthesis, you unlock the ability to design your own synthetic routes for entirely new molecules. You become the architect of molecular construction, not just a builder following a pre-written blueprint. And that, my friends, is incredibly empowering!
So, the next time you see a molecule, don't just stare at it. Imagine breaking it apart. Imagine the reactions that could have put it together. You might be surprised at how much you can figure out, and how much fun it can be. Keep exploring, keep questioning, and keep synthesizing. The world of molecules is vast and full of wonders, and you've got the tools to explore it. Go forth and create something amazing!
