Unit Chemical Bonding Polarity Ws 5 Answer Key

Ever wondered why some things stick together and others don't? Or why water can dissolve so many different substances? The secret often lies in something called chemical bonding, and more specifically, the fascinating concept of polarity. Think of it like a tiny dance between atoms, where they share or transfer electrons, and this dance dictates how molecules behave. And if you've stumbled upon "Unit Chemical Bonding Polarity Ws 5 Answer Key," you're likely diving into a world that's both incredibly useful and surprisingly fun to understand!
This topic might sound a bit "science-y," but its principles are at play everywhere, from the food we eat to the cleaning products we use. For beginners, understanding polarity is a fantastic stepping stone into the broader world of chemistry. It helps demystify why certain reactions happen and makes grasping more complex concepts much easier. For families looking to explore science together, it's a great way to spark curiosity. Imagine explaining to your kids why oil and water don't mix using the idea of polarity – it makes learning interactive and memorable! And for hobbyists, whether you're into gardening, cooking, or even making your own soaps, understanding how substances interact due to their polarity can lead to more successful and creative projects. It’s about understanding the "why" behind everyday phenomena.
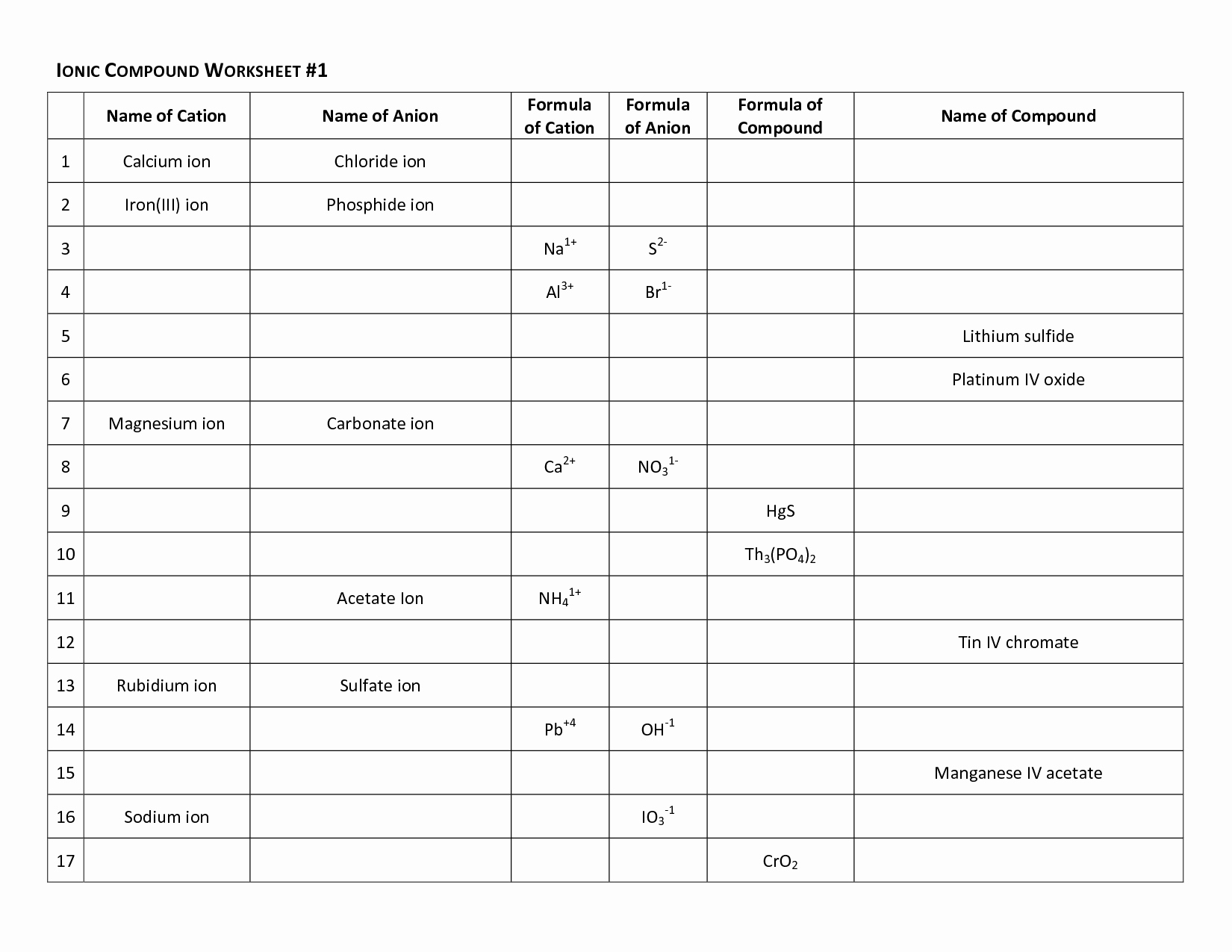
So, what exactly is this "polarity" we're talking about? In simple terms, some molecules have a slight positive charge on one end and a slight negative charge on the other, like a tiny magnet. This is called a polar molecule. Water is a classic example! Other molecules are more evenly balanced, with no distinct charges – these are nonpolar molecules. The "Ws 5 Answer Key" you might be looking at is simply a guide to help you check your understanding of these concepts, likely through practice problems. It’s like a tutor that confirms you're on the right track.
Think about common examples. Why does salt (an ionic compound, a form of strong bonding) dissolve so readily in water (a polar molecule)? Because the charged ends of water molecules are attracted to the charged ions in salt. But try dissolving oil (a nonpolar molecule) in water – it just beads up. This is because polar and nonpolar substances generally don't mix well. It's the rule of "like dissolves like." Variations could involve looking at different types of bonds, like covalent bonds (where electrons are shared) and ionic bonds (where electrons are transferred), and how these lead to different degrees of polarity or complete charge separation.
Getting started is easier than you think! If you have a "Ws 5 Answer Key," the best way to begin is by working through the problems it's designed for. Don't be afraid to look up definitions for terms you don't understand. You can also search for simple diagrams or animations online that visually represent how electrons are shared or distributed in different molecules. Visual aids can make the abstract concept of electron distribution much more concrete. Try to identify everyday substances around your home and think about whether they might be polar or nonpolar. This active engagement is key to solidifying your understanding.
Ultimately, exploring chemical bonding and polarity isn't just about memorizing facts; it's about gaining a deeper appreciation for the molecular world that surrounds us. It's a journey of discovery that can unlock a new way of seeing and understanding everything from a simple glass of water to the complex biological processes happening within us. So, dive in and have fun with it – the answers are waiting!
