Understanding Periodic Trends In Atomic Ionizability
Ever looked at a bunch of stuff, like, say, all the kids in a school, and noticed they have certain things in common? Maybe the older ones are taller. Or the ones who eat their veggies are generally healthier. Well, atoms are kinda like that, but with their own little quirks. We're diving into how "ionizable" they are, which is just a fancy way of saying how easy it is to snatch an electron away from them.
Think of an electron as that one toy your sibling really wants. Sometimes it's super easy to grab. Other times, it's practically glued to the atom's hand. This "grab-ability" changes as we hop around the periodic table, which is basically a big family tree for atoms. It's like trying to swipe candy from a baby versus a grumpy teenager guarding their stash.
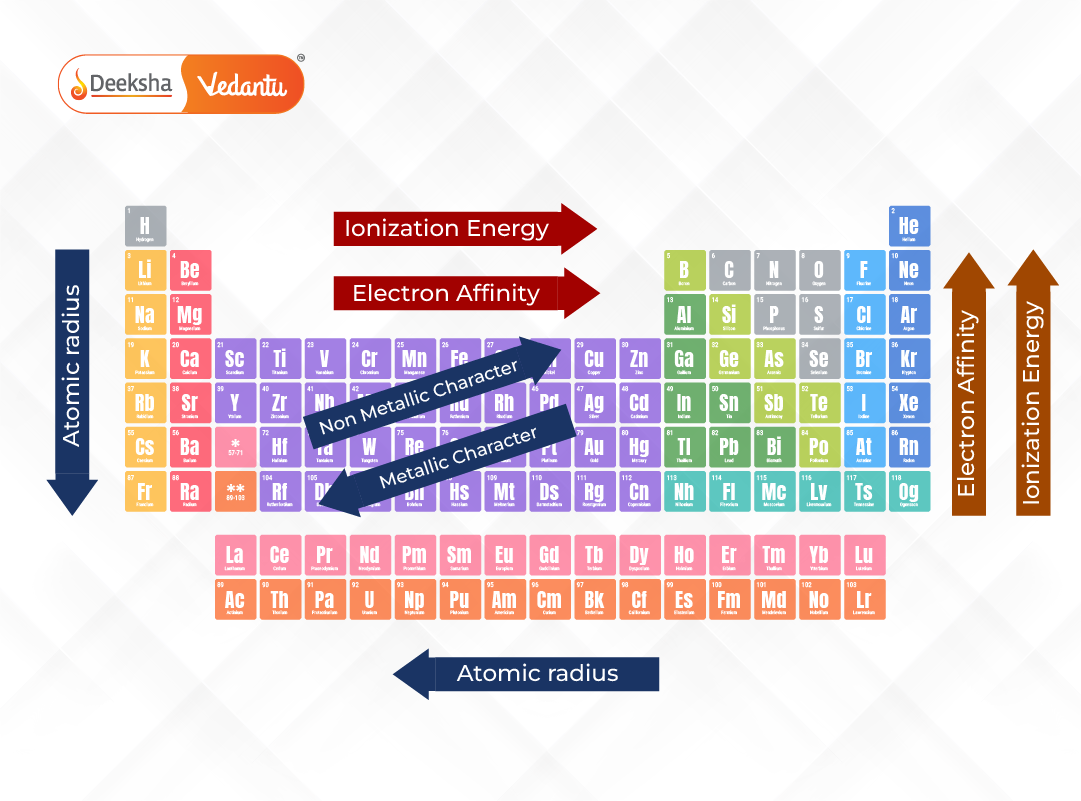
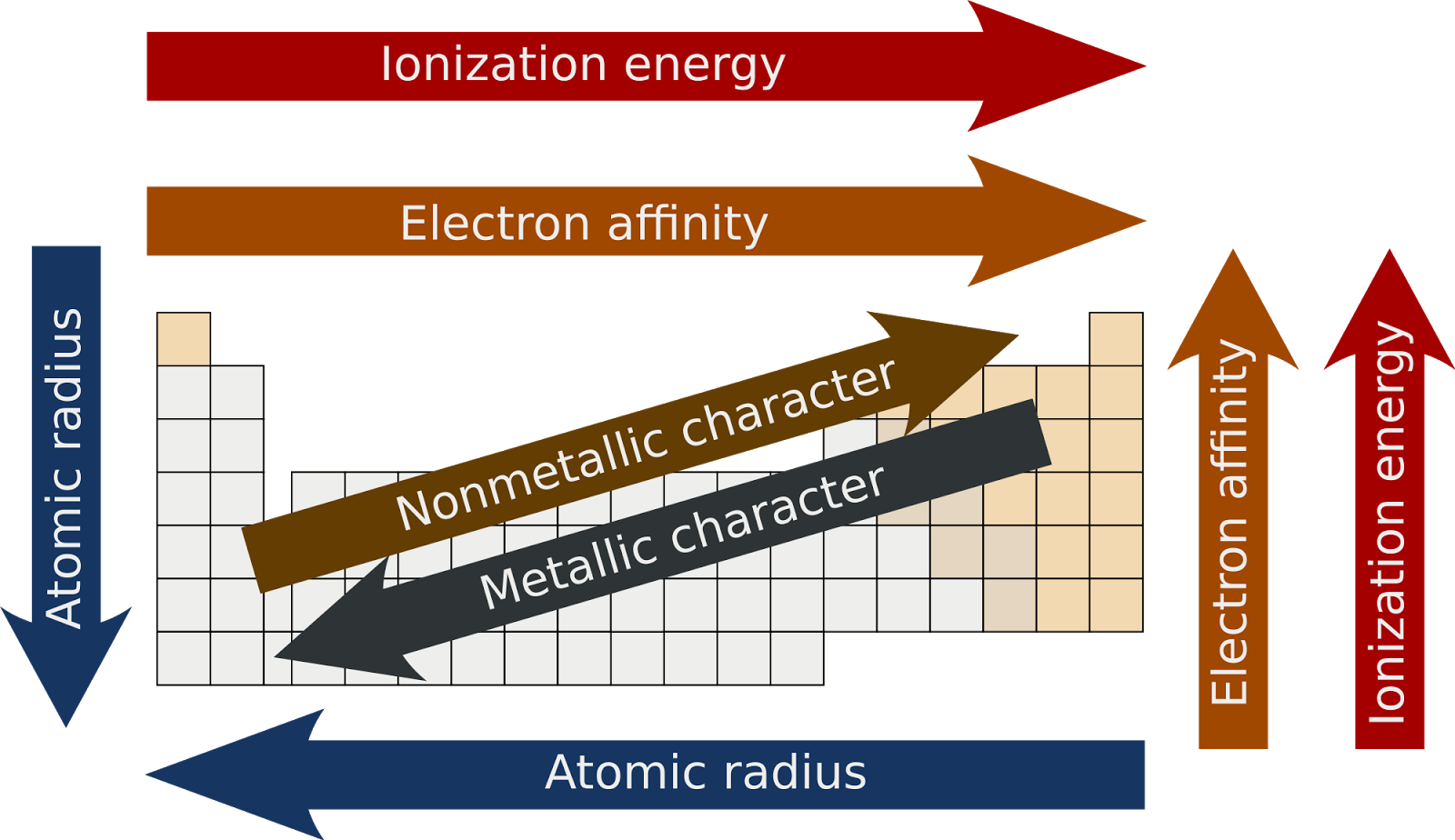
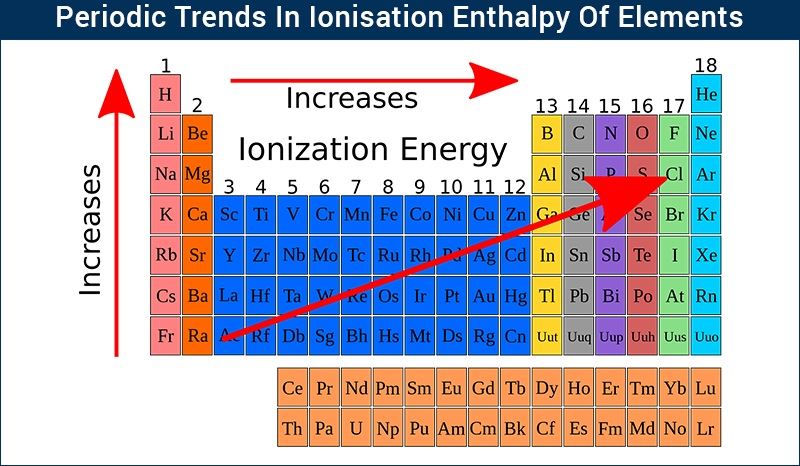
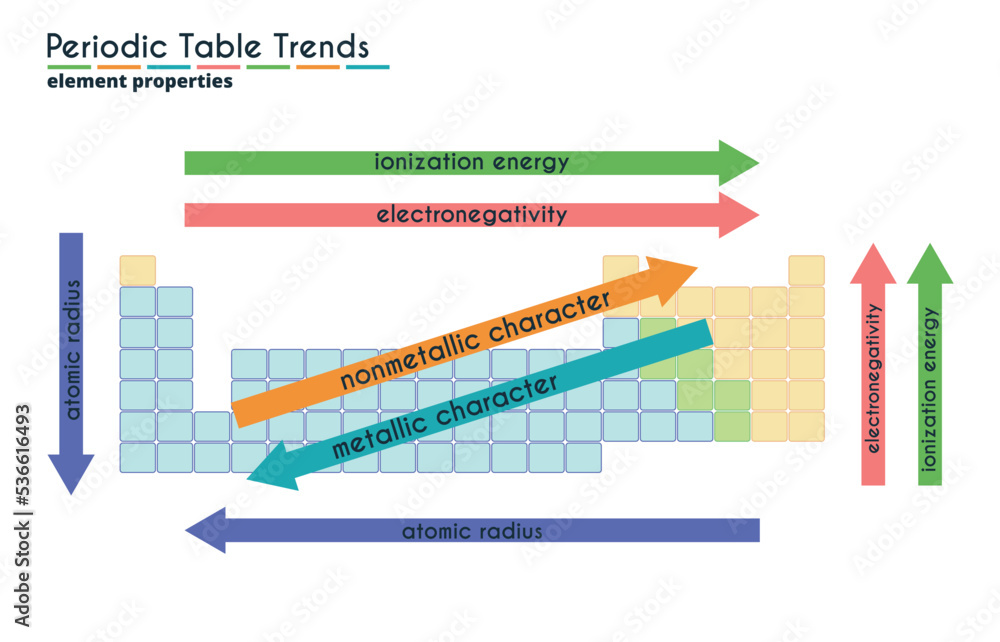
Let's talk about our first trend: Across a row. Imagine you're walking down a street, and each house is an atom. As you go from left to right, the atom gets a little more stubborn. It's like each house on the street has a slightly stronger lock on its door.
Why? Well, there's this thing called nuclear charge. Think of the nucleus as the really strict parent in the atom house. As you move across a row, this parent gets a bit stronger, pulling on all the electrons a bit tighter. So, those outer electrons, the ones we're trying to nab, are held more securely.
It's like trying to pull a balloon away from someone. If they're really holding onto it, it's tough. If they're just loosely holding it, poof, it's yours.
So, generally, as you go from the left side of the periodic table to the right, atoms get less ionizable. They're like, "Nope, not giving up my electrons easily, thank you very much." This is kind of an unpopular opinion, I know. Everyone loves the easily-give-away-ers, but the stubbornly-holding-on-ers have their own charm, right?
Now, let's switch gears and look Down a column. This is where things get a bit different. Imagine you have a whole stack of boxes, and each box is an atom. As you go down the stack, the boxes get bigger. And the stuff inside is further away from the main action at the bottom.
The "main action" in an atom is its nucleus, that strong parent we talked about. When you go down a column, you add more "shells" of electrons. These are like extra layers of protection around the nucleus.
Think of it like trying to get a toy from someone. If they're right in front of you, it's easy. If they're standing behind a bunch of people, or even behind another person who's holding them, it’s harder to reach them.
Those outer electrons are like the kids on the playground, and the nucleus is the teacher in the classroom. As you go down a column, the playground gets bigger, and the kids are further away from the teacher. The teacher's "pull" on those far-off kids is weaker.
So, as you go down a column, atoms become more ionizable. They're more willing to let go of those outer electrons. It’s like they’re saying, "You know what? They’re pretty far away. Take 'em!"
This is the opposite of what happens across a row. It’s like the universe enjoys keeping us on our toes. Just when you think you've got the hang of it, it throws you a curveball.
Let's talk about some of the big players. On the far left, you have the alkali metals. Think of Lithium, Sodium, and Potassium. These guys are the super-friendly, always-willing-to-share types. They practically throw their one outer electron away.
They're like the people who always offer you a piece of their candy. "Here, have some! You want it? Take it!" Their ionizability is through the roof.
Now, on the far right, you have the noble gases. Think of Helium, Neon, and Argon. These guys are the opposite. They're like the grumpy guardians of their electron shells. They've got a full house, and they're not letting anyone in or out easily.
Their electron configurations are super stable. They're like, "Nope, everything is perfect here. Don't touch my electrons." Their ionizability is super, super low. They’re the ultimate introverts of the atomic world.
And then there are the halogens, right next to the noble gases. Think of Fluorine and Chlorine. These guys are a bit of a mixed bag. They really want an electron to become like a noble gas, so they hang on to their own electrons pretty tightly, but they're not quite as stubborn as the noble gases.
They're like the kids who are almost done with their homework. They're focused, but they might get distracted by a really good offer.
So, to sum it up, ionizability generally increases as you go down a column and decreases as you go across a row. It's like a little trend map for how easily we can cause atomic drama.
It's important to remember that these are trends, not absolute rules set in stone. There are always exceptions to the rule, like that one kid in class who's super tall but is still in the youngest grade. Science loves its little surprises.
Understanding this helps us understand how atoms interact. It’s like knowing which of your friends are most likely to lend you money (high ionizability) and which are going to keep it locked away (low ionizability).
This ability to lose electrons is crucial for forming bonds. It's how atoms decide to become friends and stick together. Some atoms are practically begging to form bonds because they're so happy to give away an electron.
Others are more aloof, preferring to keep their electrons to themselves. It’s all about the electron dance!
So, next time you see the periodic table, don't just see a bunch of boxes. See a family with different personalities, different levels of generosity, and different levels of stubbornness when it comes to their precious electrons. It’s a whole drama unfolding, one electron at a time!

And hey, if you ever feel like you’re being a bit too stubborn with your own “electrons” (maybe your favorite snack), just remember the alkali metals. They’re a good reminder to share sometimes!
Or, if you're always the one giving everything away, maybe channel your inner noble gas for a bit. It's okay to hold onto what's yours sometimes!
It’s all about balance, in atoms and in life. And who knew atomic behavior could be so… relatable?
So, there you have it. A little peek into the world of atomic ionizability. No complicated jargon, just some fun analogies to make sense of it all. Now go forth and impress your friends with your newfound knowledge of electron-snatching tendencies!
It's pretty cool when you think about it. These tiny, invisible things have their own social dynamics. And we get to peek behind the curtain!
And remember, the periodic table isn't just a chart. It's a storybook of elements, and ionizability is one of its most exciting chapters. Enjoy the read!
