Under Acid Hydrolysis Conditions Starch Is Converted To
Ever wondered what happens to that fluffy pile of starch when things get a little… intense? We’re talking about a situation that sounds like a culinary science experiment gone wild, but trust me, it’s way more fascinating (and delicious) than you might think! Imagine your favorite starchy foods – the glorious potatoes, the comforting bread, the perfectly fluffy rice – all ready to undergo a magical transformation. And the secret ingredient to this transformation? A splash of acid!
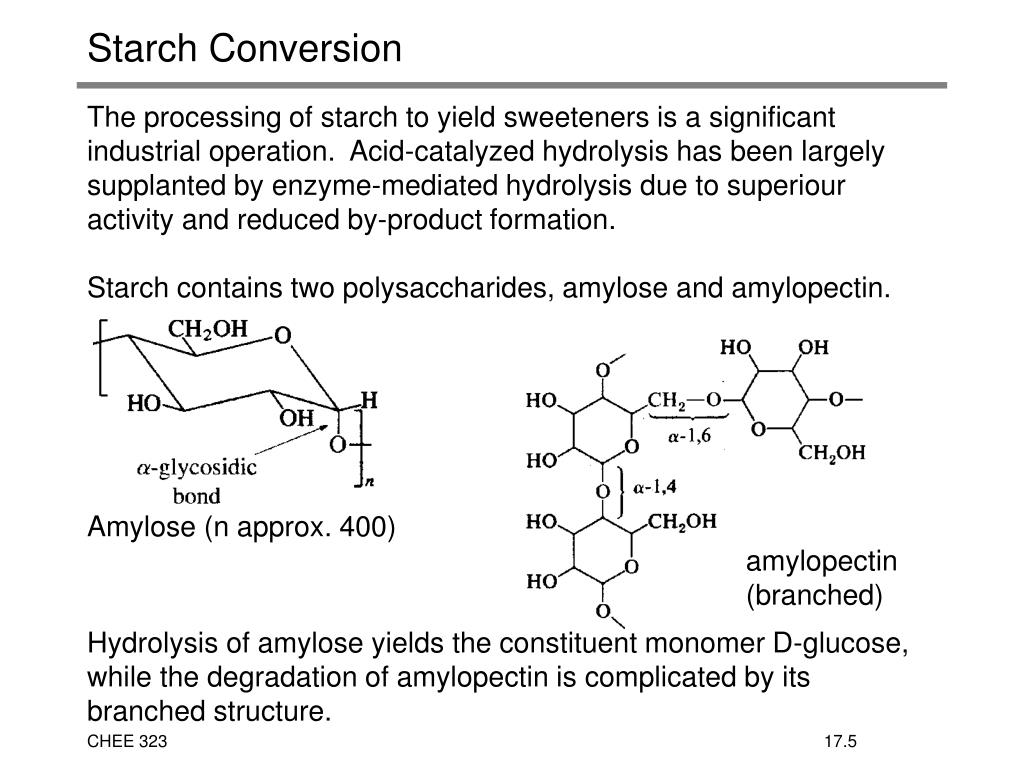
So, picture this: we’ve got a big, ol’ pile of starch. It’s like a giant Lego castle, made up of countless tiny, identical building blocks all linked together in a super-long chain. These blocks, in the grand scheme of the carbohydrate world, are called glucose units. Now, this starch castle is pretty solid. It’s what gives our foods their structure and provides us with slow-burning energy. But sometimes, we need to break it down, right? We need to liberate those individual glucose units so our bodies can gobble them up super-fast, or maybe so we can whip up something even more delightful in the kitchen.
Enter the superhero of our story: acid! When we introduce a bit of acid into the mix – think of it like a tiny, but very determined, demolition crew – it starts to gently, but persistently, snip away at the bonds holding those glucose units together. It’s not a violent explosion, mind you. It’s more like a very polite, yet incredibly effective, unzipping. The acid works its magic, and one by one, those long starch chains start to shorten. It’s like watching a giant rope fray until it’s just a bunch of individual threads.
And what do these liberated threads become? Drumroll, please… they become glucose! That’s right, the very same building blocks that made up the starch castle are set free. But it doesn't stop there! Depending on how long our acid demolition crew has been at it, and how much acid we’ve thrown into the mix, we can get a whole spectrum of goodies.
If the acid is feeling a little more aggressive, and it works its way through most of the starch castle, we end up with a whole lot of pure, unadulterated glucose. This is like getting a whole bag of those individual Lego bricks, ready to be used for anything! And when I say anything, I mean anything. This is the sweet stuff that our bodies crave for immediate energy. Think of that sugary rush you get from a piece of fruit – a lot of that sweetness comes from glucose! In the food industry, this pure glucose is a star player. It’s used in everything from candies and baked goods to energy drinks and even pharmaceuticals. It’s the ultimate quick-release energy source, and the acid hydrolysis process is a fantastic way to get it.
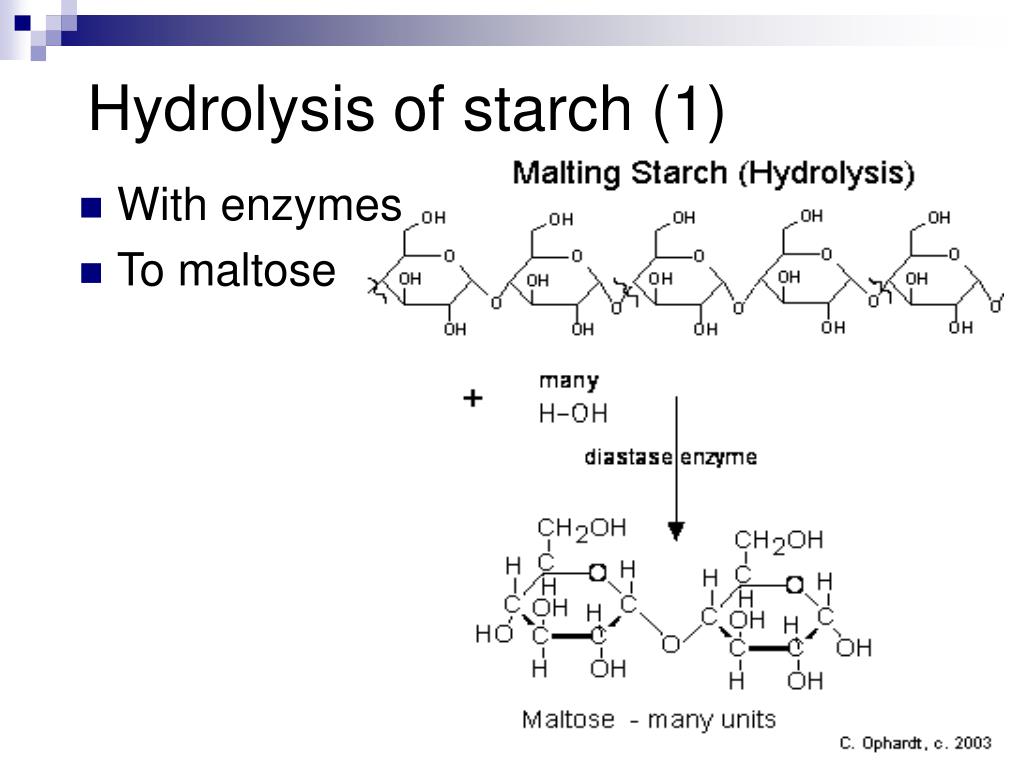
But what if the acid is feeling a bit more laid-back? What if it only manages to break off a few glucose units at a time? Well, then we get something else really cool: maltose and dextrins!

Maltose is like a tiny duo, two glucose units holding hands. And dextrins? They’re like small clusters of glucose units, still a bit connected, but much shorter than the original starch.
These guys are also super useful. Maltose, for instance, is what gives malted milkshakes their distinctive, slightly sweet, malty flavor. It’s also a key ingredient in brewing beer – the yeast munch on maltose and produce that lovely alcoholic beverage we all know and… well, some of us enjoy! And dextrins? They’re fantastic for giving food that satisfying texture. They can act as thickeners in sauces and gravies, or as stabilizers in processed foods. They're the unsung heroes of our pantry, making our meals just a little bit better, a little bit smoother, a little bit more… right.
So, under these acid hydrolysis conditions, that seemingly inert, bulky starch doesn't just disappear. Oh no! It’s like a shy giant revealing its true, versatile nature. It transforms into the simple, sweet, and incredibly useful building blocks that fuel our bodies and tantalize our taste buds. It’s a process that takes something humble and turns it into something truly spectacular, ready to be used in a million different ways. It’s chemistry, yes, but it’s also a little bit of everyday magic happening right before our eyes, or at least, right within our food! It’s a testament to how a little bit of intensity, like a controlled splash of acid, can unlock a world of sweetness and energy from the most unexpected places. It’s enough to make you want to hug a potato, isn’t it? And maybe thank that humble bit of acid for its hard work!
