Triethylamine And Concentrated Hydrochloric Acid Observation
Ever wondered about the dramatic chemical reactions that make science so captivating? It’s like a mini fireworks show right in your lab! One of the most visually striking and delightfully pungent demonstrations involves two common chemicals: triethylamine and concentrated hydrochloric acid. This isn't just a fleeting puff of smoke; it’s a multi-sensory experience that reveals fundamental chemical principles in a way that's both educational and, dare we say, a little bit thrilling.
So, why is this particular combination so popular and frankly, fun? Well, it's a perfect example of a neutralization reaction, a cornerstone of chemistry. When you mix an acid with a base, they cancel each other out, forming salt and water. But with triethylamine and hydrochloric acid, the visual and olfactory payoff is immediate and impressive. It’s a tangible, real-time demonstration of how atoms and molecules rearrange themselves.
The Magic Behind the Mist
Let's dive into what makes this reaction so special. Triethylamine (often abbreviated as TEA) is a tertiary amine, which means it's a type of organic compound. In simpler terms, it’s a basic substance. When you open a bottle of triethylamine, you're greeted with a rather distinct, fishy odor – a characteristic of many amines. It’s not exactly rose-scented, but it’s part of its charm!
On the other side of the ring, we have concentrated hydrochloric acid (HCl). This is a strong acid, meaning it readily releases hydrogen ions when dissolved in water. Handling concentrated HCl requires care because it's corrosive and its fumes can be irritating. But in a controlled setting, it’s an incredibly useful tool for driving chemical reactions.
When these two substances meet, an exciting transformation occurs. The acid (HCl) donates a proton (a positively charged hydrogen ion) to the base (triethylamine). This creates a new compound, triethylammonium chloride. This salt is typically solid, but the magic happens because the reaction is exothermic – it releases heat – and because both reactants can be quite volatile. This means that as the salt is formed, it can also cause the surrounding liquid to vaporize or create tiny solid particles that become suspended in the air.
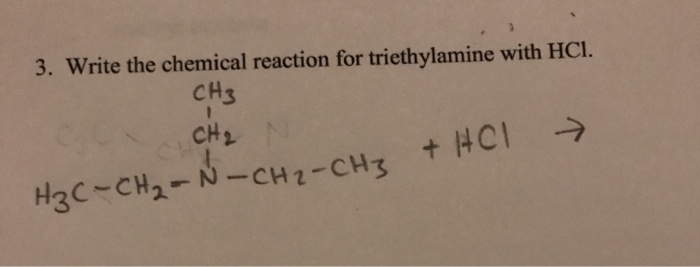
What to Expect: A Spectacle of Senses
The observation itself is where the fun truly lies. Imagine this: you have your triethylamine, and you carefully introduce the concentrated hydrochloric acid. Almost instantly, a dense, white cloud begins to form. This isn't just a wisp; it’s a robust, billowing plume of smoke-like vapor. This phenomenon is often called fuming.
The white cloud is actually tiny solid particles of triethylammonium chloride suspended in the air. These particles are so small they scatter light, making them appear white. The rapid formation of these particles, coupled with the heat generated by the reaction, creates the dramatic visual effect. It's a visual representation of chemical change happening at a rapid pace.
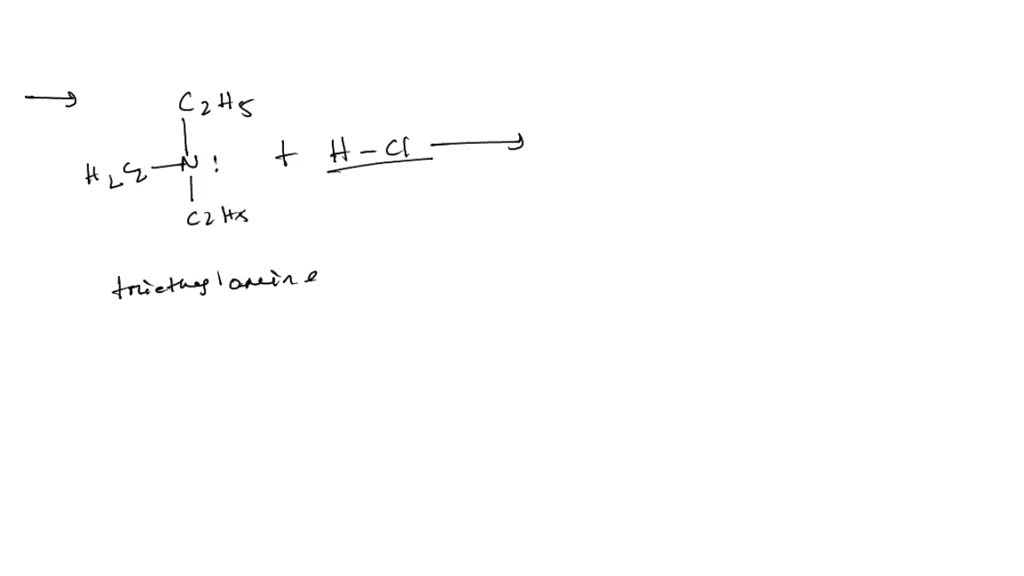
And let's not forget the smell! While the initial fishy odor of triethylamine is present, the reaction also produces ammonium chloride, which has a sharper, more pungent smell. It’s a powerful combination that really drives home the fact that a chemical transformation has taken place. It’s an experience that engages both your eyes and your nose, making it a memorable demonstration.
Why It Matters: More Than Just a Pretty Puff
Beyond the sheer visual appeal, observing the reaction between triethylamine and concentrated hydrochloric acid offers significant benefits for understanding chemistry. It provides a clear and dramatic illustration of:
- Acid-Base Neutralization: It visually reinforces the concept that acids and bases react to form salts.
- Formation of Solids (Precipitation): The white cloud demonstrates how a soluble substance can form solid particles within a reaction.
- Exothermic Reactions: The rapid production of the cloud hints at the heat released during the reaction.
- Volatility of Reactants: The way the vapors form shows that these chemicals can easily turn into gases under certain conditions.
This demonstration is frequently used in educational settings, from high school chemistry labs to university introductory courses, precisely because it’s so effective. It’s a "wow" moment that sparks curiosity and helps solidify abstract chemical concepts in the minds of students. It’s a safe, albeit pungent, way to explore the fundamental building blocks of matter and how they interact.
So, the next time you hear about triethylamine and concentrated hydrochloric acid, don’t just think of two chemicals. Think of a mini chemical spectacle, a powerful demonstration of neutralization, and a fun, engaging way to learn about the wonders of chemistry. It’s a reminder that even the most complex science can be presented in a way that’s accessible and, most importantly, enjoyable.
