Trans 1 4 Dimethylcyclohexane Chair Conformation
Okay, let’s talk about something truly thrilling. Get ready for a rollercoaster of excitement! We're diving deep into the wild and wacky world of trans 1,4 dimethylcyclohexane. Yes, you heard that right. Prepare yourselves for the adventure of a lifetime.
Now, you might be thinking, "What in the world is that?" Don't worry, I’m right there with you. It sounds like something you’d find at the bottom of a cereal box, or maybe a secret code for spies. But it’s actually… a molecule. A rather chunky molecule, if you ask me.
And this molecule, my friends, has a favorite way to hang out. It likes to get comfy. It has this thing called a chair conformation. Imagine a tiny, perfectly formed armchair. That’s kind of what this molecule does to itself.
But here's where it gets really interesting. It’s not just any old chair. It’s a trans chair. This means the two little methyl groups, which are like the molecule's little accessories, are on opposite sides. Like two friends standing on opposite sides of a dance floor, trying to catch each other's eye.
So, you have this dimethylcyclohexane ring, all bendy and wiggly. Then you slap on two methyl groups. And because it's trans, they have to be on opposite sides. It’s like a polite rule in molecule town.
Now, the chair conformation itself is pretty neat. The ring isn't flat, oh no. That would be too boring. It folds itself into this shape that’s less stressed. Think of a gentle slump instead of a stiff poker back.
The two methyl groups can either be sticking straight up (axial) or kind of poking out to the side (equatorial). It's like deciding whether to wear your hat forwards or backwards. Both are valid, but one might feel a bit more… natural.
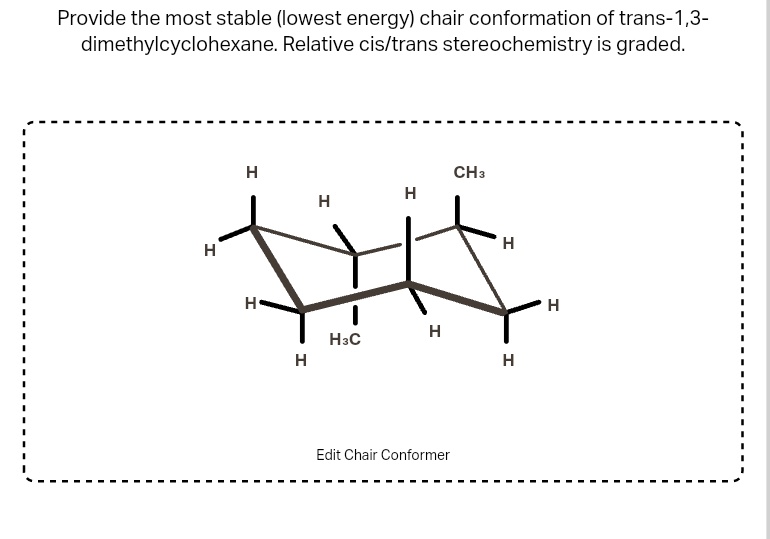
For trans 1,4 dimethylcyclohexane, one of the most stable ways it likes to exist is when both methyl groups are equatorial. This is the molecule’s ultimate comfy pose. It's like finding that perfect spot on the sofa where you can just… be.
Why is this the best? Because the equatorial positions are less crowded. Imagine trying to fit two big suitcases next to each other on a tiny shelf. It’s a tight squeeze! But if you put them on opposite shelves, much better, right?
The axial positions are a bit more… in your face. They are closer to other parts of the molecule, leading to what chemists call steric hindrance. Which, in plain English, means "ouch, that's crowded."
So, our trans 1,4 dimethylcyclohexane, the polite molecule, prefers to have its two methyl groups in the chill, equatorial spots. It’s like two guests at a party who decide to occupy separate, less crowded corners, just to have some breathing room.
But here’s the kicker, and this is my little unpopular opinion. While everyone else is raving about the all-equatorial conformation being the ultimate win, I sometimes wonder… is it too perfect? Is it too predictable?
What if, just sometimes, our trans 1,4 dimethylcyclohexane gets a little bored? What if it decides to switch things up? The molecule can flip its chair, you see. It’s like doing a little shimmy and turning your armchair upside down. Quite the acrobatic feat!

When it flips, the axial positions become equatorial, and the equatorial positions become axial. It's a complete role reversal. The molecules that were sticking out sideways are now trying to stand up straight, and vice versa.
And in this flipped state, for trans 1,4 dimethylcyclohexane, one methyl group will be axial and the other will be equatorial. It's not the absolute most stable, but it’s still a thing. It’s like deciding to wear your hat backwards for a change, just because you can.
It might be a bit less comfortable, a little more cramped, but it’s a valid conformation. It's a reminder that even in the predictable world of molecules, there’s room for a little bit of… variation. A little bit of spice.
So, while textbooks might point to the all-equatorial arrangement as the undisputed champion, I’ll be over here, appreciating the other possibilities. The slightly less perfect, but equally fascinating, states of being for our trans 1,4 dimethylcyclohexane.
It’s like looking at a family photo. Everyone might be smiling perfectly in the classic pose, but then there’s that one slightly awkward, slightly goofy face in the background. And you know what? That’s often the face that makes the picture memorable.
This molecule, with its chair conformation, its trans arrangement, and its ever-so-slight preference for lounging, is just trying its best. It’s navigating the world of molecular forces, trying to find its most comfortable fit.
And honestly, who among us can’t relate to that? We all have our preferred ways of relaxing, our favorite spots to unwind. We all have moments where we’re perfectly poised, and moments where we’re a little… less so.
So, the next time you hear about trans 1,4 dimethylcyclohexane and its chair conformation, remember this. It’s not just a dry chemical concept. It’s a tiny, bending, folding entity just trying to find its groove. And sometimes, the groove isn't perfectly straight and tidy.
Sometimes, the groove involves a little bit of leaning, a little bit of stretching, and a whole lot of molecular personality. And that, my friends, is something worth a little smile.
So yes, the all-equatorial is likely the most stable. But the other conformation, the one with one axial and one equatorial methyl group? That one has character. That one tells a story. That one is, dare I say, more interesting?
Let’s give a round of applause for trans 1,4 dimethylcyclohexane. It’s more than just a name; it’s a wonderfully complex little world, all happening in a tiny armchair. A world where even slight imperfections can be truly… charming.
And as for me? I’ll be over here, contemplating the sheer, unadulterated joy of a molecule finally finding a comfortable, albeit slightly quirky, way to sit.
The universe is made of stories, not atoms.
It's a simple truth, really. Even the smallest things have their tales to tell. And the tale of trans 1,4 dimethylcyclohexane in its chair conformation? It's a story of shape, space, and a subtle dance of preferences.
So, while the scientists diligently calculate the energy differences, let’s just appreciate the visual. The little molecule, doing its best impression of a very comfy chair, with its accessories strategically placed. It’s a miniature drama playing out in the world of the very, very small.
And that, I think, is pretty darn cool. No matter how you arrange those methyl groups, it’s still a fascinating little structure. It’s still trans 1,4 dimethylcyclohexane, doing its thing.
So there you have it. My little ode to a molecule. My perhaps unconventional take on its preferred seating arrangements. May your own chair conformations always be as entertaining!
