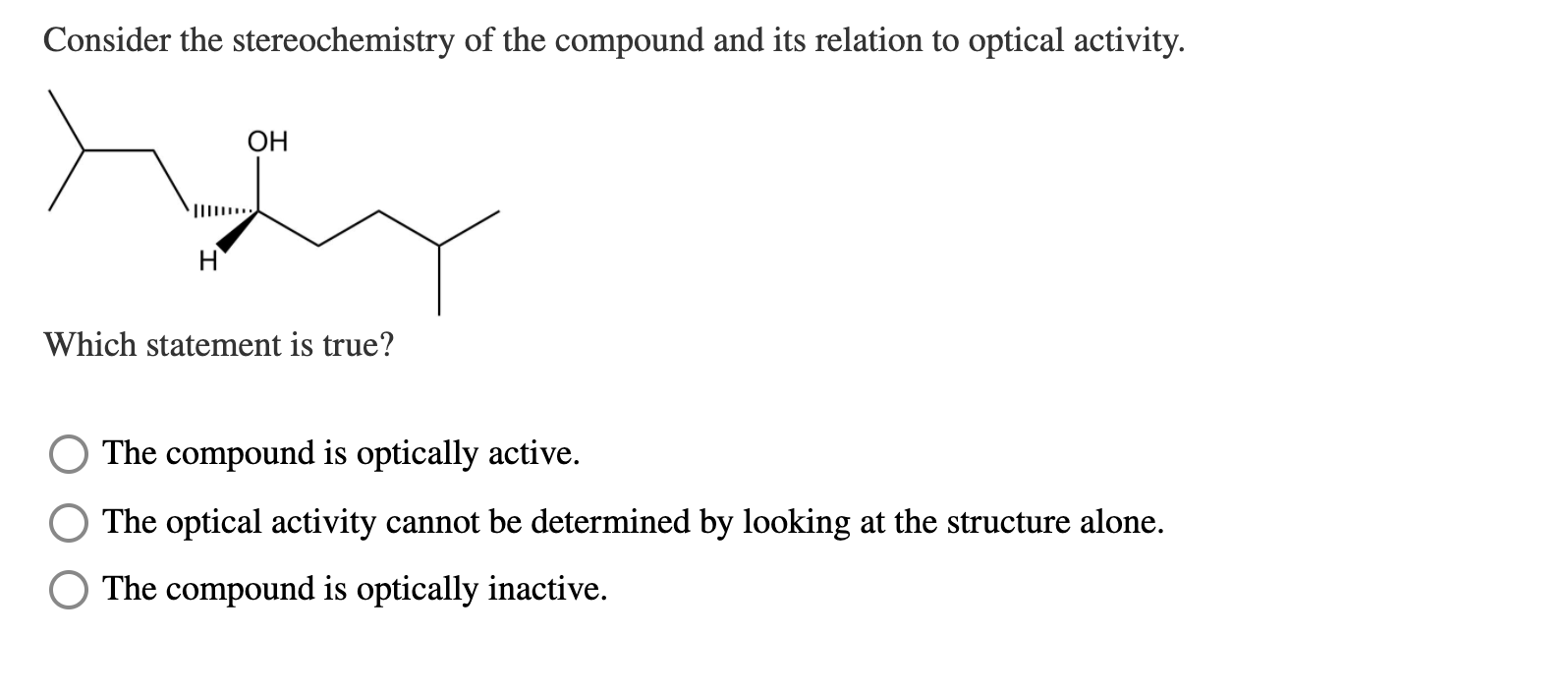
To Possess Optical Activity A Compound Must Be

Hey there, science explorers! Ever wonder why some molecules are total show-offs, while others are just…meh?
We’re talking about optical activity today. Sounds fancy, right? But it’s actually super cool. Think of it like this: some molecules have a secret handshake. They can twist and turn light, and that’s what makes them optically active.
So, what’s the magic trick? What’s the secret ingredient that makes a compound do this light-bending dance?
The Key Ingredient: Chirality!
It all boils down to something called chirality. Don't let the big word scare you. It's actually pretty intuitive once you get it. Think of your hands. They look almost identical, right? But you can’t put your left glove on your right hand, can you?
They are mirror images of each other, but they aren’t the same. That’s chirality in a nutshell. Molecules can be like that too. They can exist as two different forms, called enantiomers, which are non-superimposable mirror images of each other.
So, to possess optical activity, a compound must be chiral. If a molecule isn’t chiral, it’s just going to sit there, doing nothing with that light. Boring!
Why Chirality Matters (Besides Twisting Light)
This isn't just some abstract chemistry concept. Chirality is everywhere! Your DNA? Chiral. Amino acids, the building blocks of proteins? Chiral. Sugars? Yep, chiral.
Our bodies are basically a giant chiral playground. And because of this, the way our bodies interact with molecules often depends on their specific chiral form. It’s like trying to fit a square peg in a round hole – some molecules just won’t fit where they’re supposed to if they have the wrong “hand.”
The Mirror Image Mishap
Imagine you have a molecule that’s chiral. Let’s call it "Righty." It has a mirror image, "Lefty." These two are like a pair of gloves. They look the same but are flipped.
When plane-polarized light comes along, "Righty" will rotate it one way. Let's say clockwise. "Lefty," being the opposite, will rotate it the exact same amount but counter-clockwise. They’re perfectly balanced opposites.
But here’s the kicker: if you have a mixture containing equal amounts of "Righty" and "Lefty," what happens?
The Racemic Ruckus
When you have a 50/50 mix of a chiral compound and its mirror image, it’s called a racemic mixture. And guess what? This mixture is optically inactive. Poof! The light-twisting power disappears.
It’s like having a party where everyone is doing a dance. If half the people are doing the macarena clockwise and the other half are doing it counter-clockwise at the same speed, the overall effect is… nothing. It all cancels out.
So, even if a compound can be chiral, if it’s in a racemic mixture, it won’t show optical activity. It’s like having the potential for greatness but being held back by your twin!
What Makes a Molecule Chiral?
Okay, so we know chirality is the ticket. But what’s the architectural feature that gives a molecule its chiral personality?

The most common culprit is a chiral center. This is usually a carbon atom that’s bonded to four different groups. Think of a carbon atom as a little hub with four spokes. If all four spokes are attached to different things, then that carbon atom is a chiral center.
It’s like a four-pronged star. If all the points are different, you can't flip it and make it look exactly the same as the original in all orientations. You’d have to pick it up and rotate it, and then you’d see the difference.
So, a carbon with four different atoms or groups attached? Bingo! That’s a prime candidate for chirality.
Not Always So Simple
Now, don’t get too comfortable. Chemistry loves to throw curveballs. While a chiral center is the classic example, there are other ways a molecule can be chiral. Some molecules are chiral due to their helical shape (like a spiral staircase) or even due to the arrangement of atoms around a bond (called axial chirality).
It’s like saying, "Okay, a chiral center is the most common way to be quirky, but we also have these other funky architectural styles that can make you chiral."
The Light-Bending Spectacle
So, when polarized light hits a chiral molecule, it’s not just passing through. The molecule’s chiral structure interacts with the light’s electric field. It’s a tiny dance between the molecule and the light waves.

Imagine the light waves as tiny wiggling ropes. A chiral molecule is like a cleverly designed obstacle course. It forces the ropes to wiggle in a particular direction. If the molecule is "Righty," it nudges the ropes clockwise. If it's "Lefty," it nudges them counter-clockwise.
This rotation is what we measure. Chemists use a device called a polarimeter to see how much a compound rotates plane-polarized light. The amount of rotation tells us about the specific enantiomer and its concentration.
A Sweet and Sour Tale
Here’s a classic, slightly tragic, example: Thalidomide. This drug was prescribed to pregnant women in the late 1950s and early 1960s to combat morning sickness. It was sold as a single compound, but it actually existed as two enantiomers.
One enantiomer was effective. It did its job. But the other enantiomer, the mirror image? It caused severe birth defects. This was a harsh lesson in the importance of chirality. The body’s chiral machinery treated the two enantiomers very differently.
It really drives home why understanding optical activity and chirality is so vital in fields like medicine and pharmacology. What seems like a tiny difference in molecular shape can have huge consequences.
The Fun Side of Optical Activity
Beyond life-and-death scenarios, there’s a lot of fun to be had with optical activity. Think about flavors and smells.

Carvone is a great example. One enantiomer of carvone smells like spearmint. The other enantiomer? It smells like caraway (you know, the seeds in rye bread).
Seriously! Same molecule, just a different "hand," and your nose picks up a completely different scent. It’s like having a molecule that can magically transform into two different perfumes. How cool is that?
And what about taste? Aspartame, the artificial sweetener, is another chiral compound. Only one of its enantiomers has that sweet taste. The other tastes bitter.
So, the next time you enjoy spearmint gum or your diet soda, you can think about the fascinating world of chiral molecules and their optical activity. They’re not just passive observers; they’re active participants in shaping our world, one light twist at a time.
The Takeaway
So, there you have it. To possess optical activity, a compound must be chiral. It needs that special, non-superimposable mirror image quality. It needs to be able to interact with polarized light in a way that its mirror image (or a mix of both) doesn't.
It’s a fundamental concept, but it’s also a gateway to understanding the intricate molecular dance that goes on all around us, and even inside us. It’s a reminder that sometimes, the smallest differences can make the biggest impact. Pretty neat, huh?
