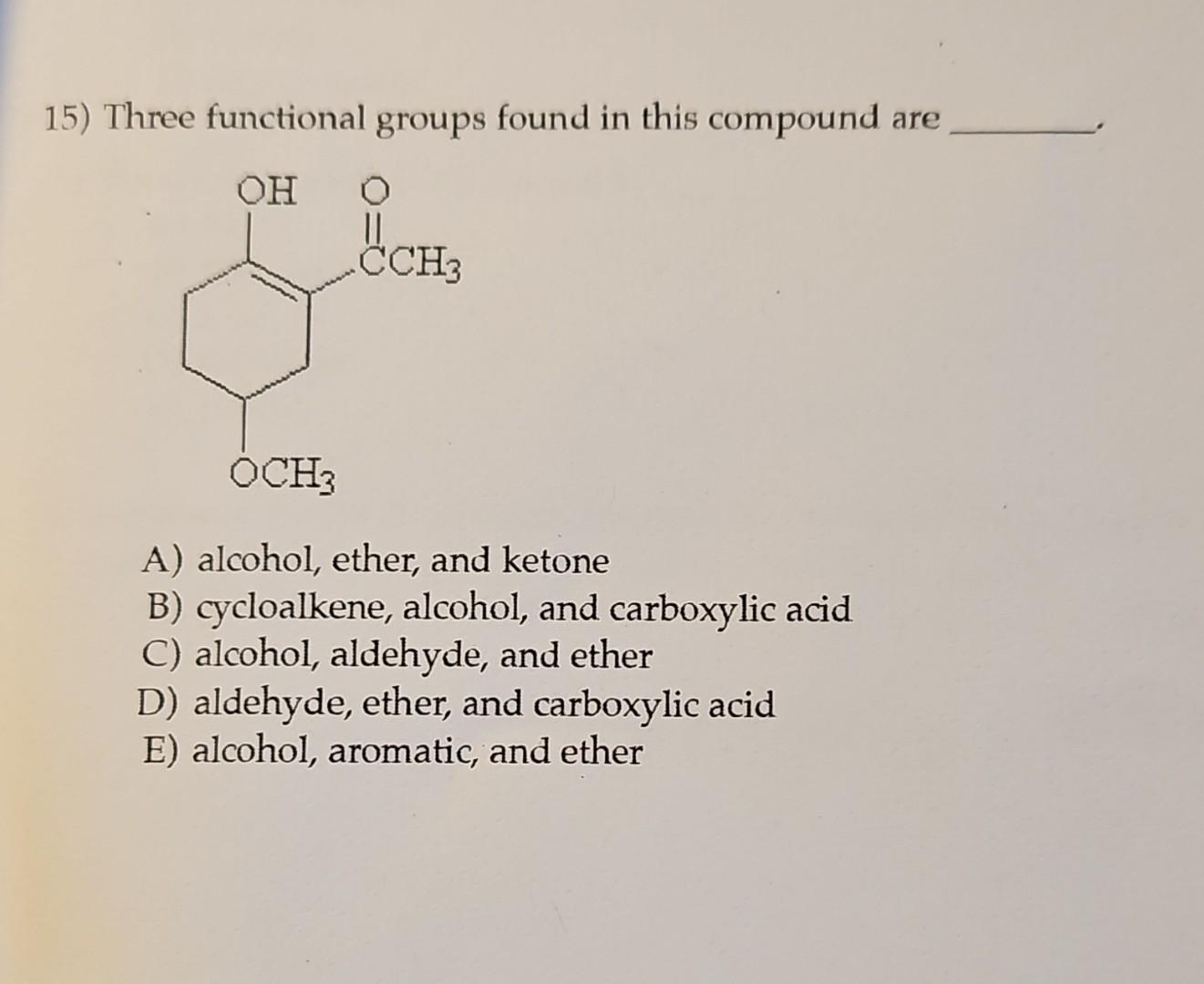
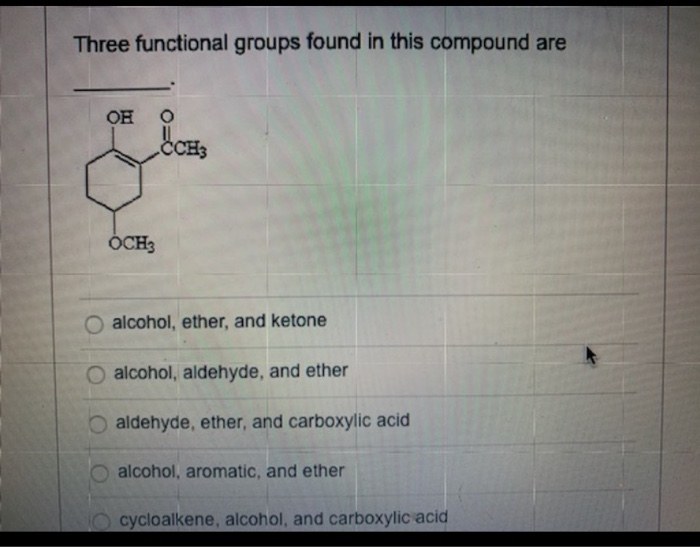
Three Functional Groups Found In This Compound Are ________.
Ever looked at a molecule and felt a spark of curiosity? It's like deciphering a secret code, and the key to unlocking its secrets often lies in its functional groups! These aren't just random bits of atoms; they're the superstars of the chemical world, dictating how molecules behave, what reactions they participate in, and even what unique properties they possess. Think of them as the building blocks that give compounds their personality. Understanding these groups is like learning a new language, and once you grasp it, a whole new universe of chemistry opens up. It's incredibly useful, whether you're a student diving into organic chemistry for the first time, a curious mind wanting to understand how everyday substances work, or even a professional looking to innovate. The beauty of functional groups is their universality – the same groups pop up in everything from life-saving medicines to the flavors in your favorite foods!
The "Holy Trinity" of Functional Groups
Let's dive into a particularly interesting compound and spotlight three of its defining functional groups. These aren't just any three; they're a powerful combination that contributes significantly to the molecule's characteristics. When we talk about the functional groups found in this compound, we're talking about the alcohol, the carboxylic acid, and the ester. These three, often seen working together or influencing each other, are fundamental to understanding a vast array of chemical reactions and applications.
The Versatile Alcohol: More Than Just a Drink
First up, we have the alcohol group. You might immediately think of beverages, but chemically speaking, an alcohol is characterized by a hydroxyl (-OH) group attached to a saturated carbon atom. This seemingly simple group is incredibly versatile. Alcohols can act as solvents, fuels, and are crucial intermediates in the synthesis of many important compounds. The hydroxyl group is polar, meaning it can form hydrogen bonds. This property is what makes many alcohols soluble in water and gives them their ability to interact with other polar molecules. Think about how rubbing alcohol disinfects – that's the power of the -OH group at play! In our compound, the presence of an alcohol group suggests potential for reactions like oxidation or esterification, making it a reactive and adaptable part of the molecule.
The Acidic Powerhouse: Carboxylic Acid
Next, we encounter the carboxylic acid group, represented as -COOH. This group is defined by a carbonyl group (C=O) bonded to a hydroxyl group (-OH). The "acidic" in its name is a big clue to its behavior. Carboxylic acids are, well, acidic! This means they can donate a proton (H+) in solution, forming a carboxylate ion. This acidity is vital for many biological processes and industrial applications. Think of the familiar acetic acid in vinegar – that sour taste is courtesy of the carboxylic acid group. In our compound, the -COOH group brings a distinct level of reactivity and acidity. It can readily participate in neutralization reactions, and it's a key player in forming amides and, importantly, esters!
The Sweet and Savory Ester
Finally, we have the ester group, typically represented as -COO-. An ester is formed when the hydrogen atom of the carboxylic acid's hydroxyl group is replaced by an alkyl or aryl group. Esters are incredibly important because they are often responsible for the pleasant fragrances and flavors we associate with fruits and flowers. That's right, the sweet scent of strawberries or the tangy zest of a lemon often comes from ester compounds! Beyond their olfactory contributions, esters are used as solvents, in the production of plastics (like polyester), and as components in many pharmaceuticals. The formation of an ester, often from an alcohol and a carboxylic acid, is a classic reaction in organic chemistry, and its presence in our compound points to its potential in flavoring, fragrancing, or even as a building block for polymers.
The Synergy of Three
The truly fascinating aspect of our compound is the interplay between these three functional groups. The proximity and interaction of the alcohol, carboxylic acid, and ester groups can lead to unique chemical behaviors. For instance, a molecule might have an alcohol group and a carboxylic acid group that can react with each other intramolecularly (within the same molecule) to form a cyclic ester. Alternatively, these groups can influence each other's reactivity, making certain reactions more or less likely. Understanding these three functional groups isn't just about memorizing structures; it's about appreciating how they work together to give a molecule its identity and purpose. It's this intricate dance of atoms that makes chemistry so endlessly engaging and so profoundly impactful on our world.
