This Figure Reviews The Structure Of A Phospholipid.

Hey there, science explorers and couch commanders! Ever find yourself staring at, well, anything and wondering, "What’s it made of?" Today, we're diving into the microscopic world, specifically into something called a phospholipid. Now, before your eyes glaze over like a perfectly baked donut, think of it this way: phospholipids are the tiny, unsung heroes that build the walls of pretty much every cell in your body. Yeah, you heard that right. The things that make you, you, are held together by these little molecular marvels.
Imagine your body is like a massive, bustling city. Cells are the individual buildings, from towering skyscrapers to cozy little bungalows. And the walls of these buildings? They’re mostly made of phospholipids. If they weren't there, your cells would be like a deflated bouncy castle after a kid’s party – all saggy and spread out. Not a good look, trust me.
So, what's the deal with these phospholipids? Let's break them down, but in a way that won't make you reach for the nearest science textbook and use it as a doorstop. Think of a phospholipid as having two main personalities, or parts. It’s like that friend who’s super outgoing at parties but also enjoys a quiet night in with a good book. This duality is what makes them so darn important.
The first part is the "head." This head is a bit of a drama queen. It absolutely loves water. We call this part hydrophilic, which is just a fancy way of saying "water-loving." Think of it like a golden retriever absolutely losing its mind when you bring out the sprinkler on a hot day. Pure, unadulterated joy and a desperate need to be close to the water. This hydrophilic head is also a bit of a show-off; it likes to stick its head out and say "hello!" to the watery environment both inside and outside your cells.
Now, the second part of our phospholipid friend is the "tail." And let me tell you, this tail is the opposite of the head. It’s shy. It’s introverted. It actively hates water. We call this part hydrophobic, or "water-fearing." Imagine this tail is like a cat who’s just had a bath. It wants to hide, to get away from all that wetness. It’d much rather hang out with other tails in a cozy, dry little huddle. So, while the heads are waving their watery greetings, the tails are doing the opposite, trying to escape the splash zone.
Because of this split personality – one half loving water, the other hating it – phospholipids do something really cool when you have a bunch of them floating around. They spontaneously arrange themselves into a structure called a lipid bilayer. It’s like a tiny molecular dance party where everyone finds their perfect partner. The hydrophilic heads all face outwards, towards the water, and the hydrophobic tails all cuddle up together in the middle, creating a water-free zone. It's like a perfectly organized buffet line, but with molecules.

The Structure: A Simple Analogy
Let's try another analogy. Imagine you're at a crowded beach. You want to build a sandcastle, but you also want to keep your picnic basket dry. What do you do? You probably dig a moat around your castle, right? The moat is the water. The sandcastle is the inside of your cell. And the phospholipids? They’re like the tiny, determined little builders who form the walls of your sandcastle.
The heads are like little umbrellas, happily sticking out into the wet sand (the outside of the cell) and the damp air around the moat (the inside of the cell). They’re okay with getting a little wet. But the tails? They’re like the tiny, scared little squirrels who are trying to burrow as deep as they can into the dry sand, away from the water. They bunch up in the middle, forming a barrier. This barrier is the key! It keeps the water from rushing in and making your sandcastle crumble, or in our case, keeping the cell's contents contained.
So, the phospholipid bilayer is essentially a double layer of these molecules, with the tails of one layer facing the tails of the other layer in the middle. This creates a boundary, a sort of molecular fence. This fence is incredibly important because it controls what gets in and out of the cell. It's like the bouncer at a really exclusive club, deciding who gets to party inside and who has to stay outside.
Why This Matters (Beyond Just Being Cool)
Now, why should you care about this whole water-loving, water-hating molecular arrangement? Because this is what forms the cell membrane. The cell membrane is the gatekeeper, the security system, the delivery service, and the recycling center of your cell. Without it, your cells would just… cease to exist as distinct entities. They’d be a big, watery mess.

Think about it. Everything that happens inside your body – your thoughts, your sneezes, your ability to digest that delicious pizza you had last night – it all happens thanks to these individual cells working together. And the foundation of all that cellular activity is the phospholipid bilayer.
When scientists talk about the structure of a phospholipid, they’re essentially describing the blueprint for life itself. It’s not just some abstract concept; it's the very stuff that holds you together. It’s like understanding how bricks are made to appreciate the grandeur of a castle. Except, in this case, the castle is you!
Let's get a little more specific about the parts. The "head" isn't just a blob. It usually consists of a phosphate group and often a glycerol molecule. The phosphate group is the part that's super keen on water. It's got a bit of a charge, which makes it attracted to the polar nature of water molecules (water molecules are like tiny magnets, with a positive end and a negative end). This attraction is what makes it hydrophilic. It’s like a magnet for H2O!
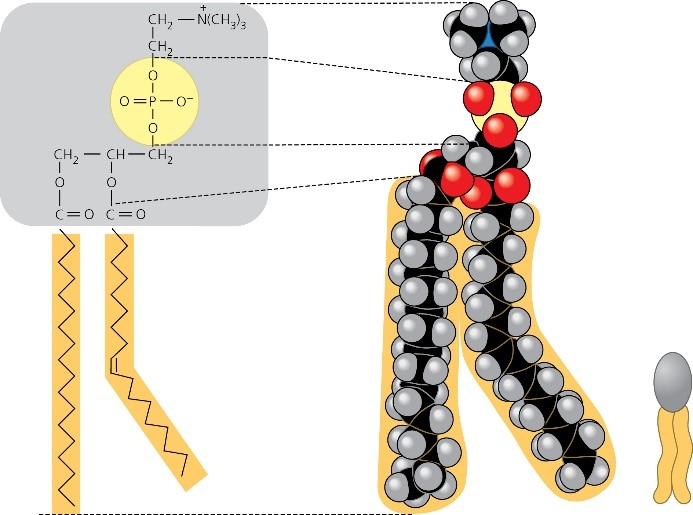
The "tail" is typically made up of two long chains of fatty acids. Fatty acids are basically long strings of carbon and hydrogen atoms. This arrangement makes them nonpolar, meaning they don’t have that positive and negative charge. Because of this, they don’t interact well with water. They’re the hydrophobic introverts of the molecule, preferring to stick together in their own little fatty acid clique.
So, when you have a bunch of these phospholipids in an aqueous solution (that's just a fancy term for "watery stuff"), they naturally organize themselves. It's not like someone is telling them what to do; it's just their inherent nature. It's like how oil and water don't mix – the oil (hydrophobic) clumps together, and the water stays separate. Phospholipids are kind of like that, but they form a very specific, organized structure.
This structure, the lipid bilayer, is like a very thin, flexible sheet. It’s not rigid like a brick wall; it’s more like a soapy film. This flexibility is crucial because cells need to be able to change shape, to move, and to expand and contract. Imagine trying to run a marathon if your body was made of stiff, unmoving bricks! You wouldn't get very far.
The cell membrane isn't just phospholipids, though. It's like a skillfully decorated cake. The phospholipids are the cake batter, forming the basic structure. But then you have other ingredients mixed in and decorating the top, like proteins and cholesterol. These other components have their own jobs. Some proteins act as channels, allowing specific things to pass through the membrane. Others are like antennas, receiving signals from the outside world. Cholesterol is like the little extra bit of fat that helps keep the membrane from getting too floppy or too stiff, depending on the temperature.

Think of it like the walls of your house. The basic structure is the wood and drywall (the phospholipids). But then you have windows (protein channels), doors (other protein transporters), light switches (receptor proteins), and insulation (cholesterol). All these parts work together to make your house functional and comfortable. The same applies to your cells!
So, when you’re looking at a diagram of a phospholipid, you’re essentially looking at the fundamental building block of every membrane in your body. It’s the reason you have a distinct inside and outside to your cells. It’s the reason your organs can stay put and do their specialized jobs.
It's quite mind-boggling, really. These tiny, seemingly simple molecules, with their love-hate relationship with water, are the architects of our very existence. They are the unsung heroes working tirelessly, day in and day out, to keep us alive and kicking. The next time you look in the mirror, take a moment to appreciate the incredible molecular dance happening within every single one of your trillions of cells. It's a testament to the elegant and often surprisingly simple solutions nature has come up with to build the complex marvel that is life.
And that, my friends, is the basic rundown of a phospholipid’s structure. It’s a bit of a dual personality, with a water-loving head and a water-hating tail, that comes together to form the essential barrier of life. Pretty neat, huh? Now, if you'll excuse me, I think my own phospholipids are calling for some water. And maybe a snack.
