Thermodynamics Of The Dissolution Of Borax Lab Report

Alright, so imagine this: you're chilling at your kitchen counter, a little bored, and you stumble upon this dusty old box. Inside? Borax. Yeah, that stuff your grandma probably used to whiten her laundry and maybe even make some questionable slime back in the day. But hold up, this isn't just about sparkling whites. We're about to dive headfirst into the wild world of thermodynamics and how it applies to dissolving this mysterious white powder in water. No, seriously, it’s way more exciting than it sounds. Think of it as a tiny, kitchen-based science adventure.
So, what exactly is thermodynamics? Basically, it’s the science of heat, energy, and work. It’s like the universe’s ultimate rulebook for how things interact and transform. And in our little borax experiment, we're looking at how heat affects the process of dissolving. Is it going to suck up heat like a black hole, or will it let it go like a celebrity at a press conference?
Our main character here is borax, also known as sodium tetraborate decahydrate. Fancy, right? It’s got a whole decahydrate party going on, meaning it’s practically dripping with water molecules. When you toss it into water, these water molecules decide it’s time for a rave, and they start pulling the borax apart. It’s a whole molecular fiesta!
Now, the big question in this lab report adventure was whether dissolving borax is endothermic (meaning it absorbs heat, making the solution feel colder) or exothermic (meaning it releases heat, making it warmer). It’s like a chemical temperature check. Will our borax be a cold-blooded killer or a warm-blooded buddy?
We’re talking about a few key players in this thermodynamic drama: enthalpy, entropy, and Gibbs Free Energy. Don't let those big words scare you! Think of enthalpy (ΔH) as the heat content of the reaction. If it’s negative, heat is released (exothermic). If it’s positive, heat is absorbed (endothermic). Entropy (ΔS) is all about disorder or randomness. When things dissolve, they tend to get messier, so entropy usually increases (becomes more positive). And Gibbs Free Energy (ΔG)? This is the ultimate decider, the Simon Cowell of chemical reactions. It tells us if a reaction will happen spontaneously. If ΔG is negative, it’s a “yes, please!” If it’s positive, it’s a “nope, not happening.”
The formula that ties it all together is a classic: ΔG = ΔH - TΔS. That 'T' there? That’s the temperature in Kelvin. So, as the temperature goes up, the entropy term (TΔS) gets bigger. This is like adding more fuel to the disorder fire.
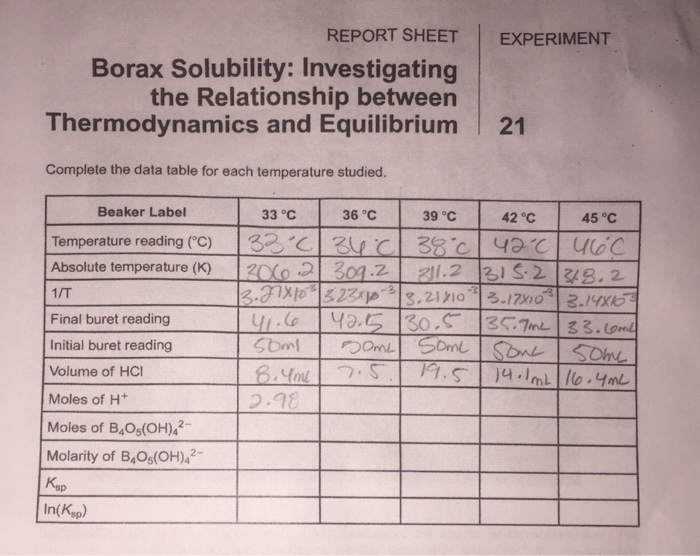
In our lab, we probably did something like this: We took a beaker, added some water, and measured its initial temperature. Then, with the precision of a brain surgeon (or at least someone who’s had their coffee), we added a measured amount of borax. We stirred like our lives depended on it, or at least until it dissolved. Then, we eagerly stuck a thermometer back in and measured the final temperature. Was it colder? Warmer? Or exactly the same? That little temperature change is our big clue.
Most of the time, when you dissolve borax in water, you’ll notice the beaker gets a little chilly. This is your first hint that the process is likely endothermic. It’s like the borax is saying, “Hey, can I borrow some of your heat energy to get this party started?” It’s not being greedy; it just needs a little boost to break those bonds and mingle with the water molecules.

So, if the temperature drops, our ΔH is probably positive. But what about entropy? When solid borax turns into dissolved ions swimming around in water, things get way more spread out and disordered. Imagine a neatly organized bookshelf versus a room after a toddler’s birthday party. Yeah, that’s entropy! So, our ΔS is almost certainly positive.
Now, back to the Gibbs Free Energy equation: ΔG = ΔH - TΔS. We have a positive ΔH (endothermic) and a positive ΔS (increasing disorder). The spontaneity of the reaction, therefore, becomes heavily dependent on that temperature (T). At lower temperatures, the positive ΔH might be the boss, making ΔG positive (not spontaneous). But as you crank up the heat, that -TΔS term becomes more and more negative. Eventually, it can overpower the positive ΔH, making ΔG negative, and bam! The reaction becomes spontaneous. So, borax is more likely to dissolve readily in hot water than in cold. It’s like it needs a warm embrace to truly let loose.

This is why, when you're making, say, a super-saturated borax solution for crystal growing (which is basically a science experiment disguised as art), you always use hot water. The higher temperature gives the borax the energy boost it needs to dissolve more of itself, setting the stage for those magnificent, geometric masterpieces.
Think about it: if it were exothermic, it would be like a chemical hand warmer, giving off heat. But it’s the opposite! It’s a heat thief. It’s not malicious, it’s just doing its thermodynamic thing. And understanding this little dance of heat and disorder helps us predict how substances will behave. It's not just about making things disappear into water; it's about the fundamental forces at play.
So, the next time you’re staring down a box of borax, remember this little café chat. You’re not just looking at a cleaning product; you’re looking at a participant in a grand thermodynamic ballet. It’s a testament to how even the most ordinary household items can hold secrets of the universe, waiting for a curious mind (and maybe a thermometer) to uncover them. Who knew that a little bit of powder could teach us so much about the laws of energy? Pretty neat, huh? It’s enough to make you want to go buy some borax and a thermometer, just to prove it yourself. Just try not to spill any on your clean whites… unless you’re testing its cleaning powers, of course!
