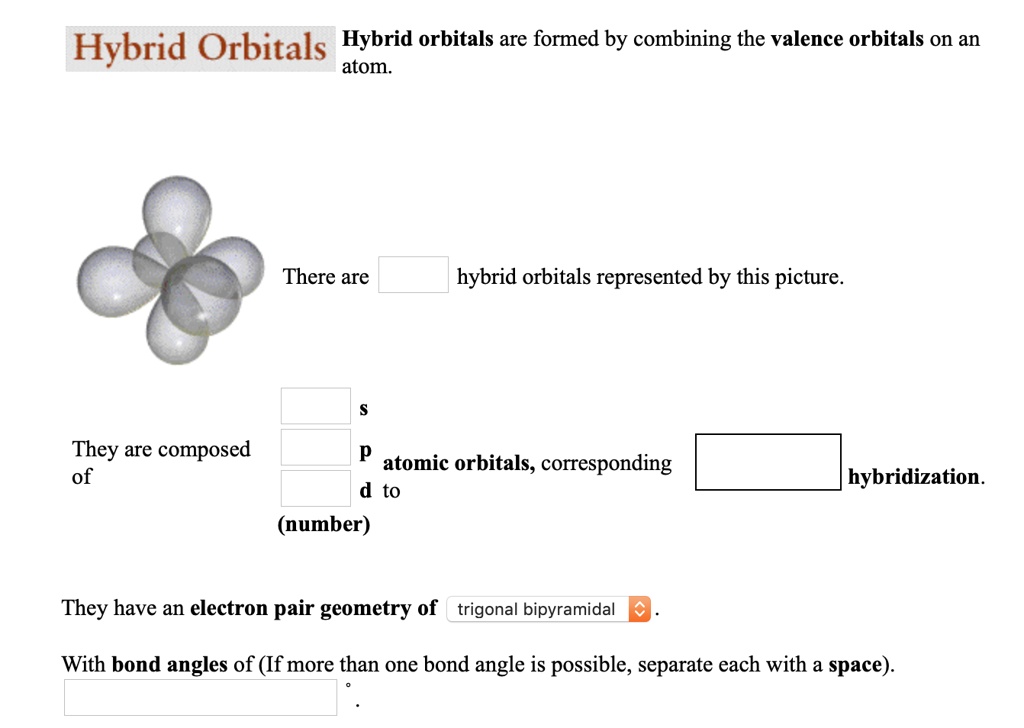
There Are Hybrid Orbitals Represented By This Picture
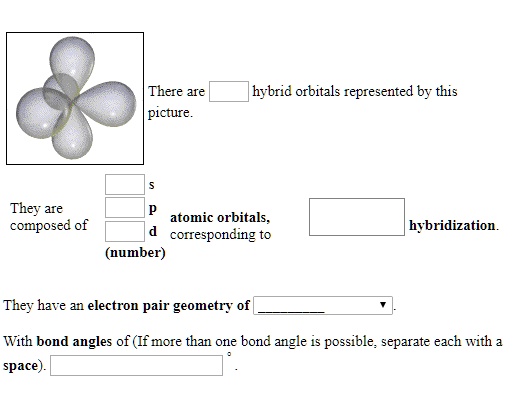
I remember this one time, in high school chemistry, feeling like I’d just been handed a secret decoder ring. We were talking about atoms, those tiny building blocks of everything, and how they stick together to make molecules. Simple enough, right? Until Mrs. Henderson, bless her enthusiastic heart, pulled out this diagram. It looked less like an atom and more like a slightly squashed, multi-lobed amoeba having a bad hair day. And she said, with a twinkle in her eye, “These, class, are hybrid orbitals.”
My brain immediately went into panic mode. Hybrid? Like a hybrid car? What was being mashed together? Was it like when my dog accidentally ate a whole bag of Doritos and looked like a particularly colourful, slightly chunky sausage? I had SO many questions, and frankly, I was a little terrified. But eventually, as the fog of confusion cleared (and after a lot of frantic note-taking and re-reading the textbook), I started to get it. It wasn't an amoeba. It was a beautifully messy compromise.
And that, my friends, is where we're heading today. We're going to dive into the wonderful world of hybrid orbitals, the invisible architects of molecular bonds that you see represented in that picture. Because honestly, if you've ever looked at a molecule and wondered, "How on earth do those atoms arrange themselves like that?", the answer often lies within these quirky, mixed-up atomic spaces.
When Atoms Get Creative: The Hybrid Orbital Story
So, let’s ditch the amoeba image for a second and think about what’s actually going on. Atoms, in their pure, unadulterated form, have these nice, tidy electron shells and subshells. We talk about s orbitals, which are basically perfect little spheres, and p orbitals, which are like dumbbells. You know, the basic shapes. And when atoms get together to form molecules, their electrons, those tiny, negative charges that are constantly zipping around, are what create the glue. But here’s the catch: the way these pure orbitals should interact doesn't always explain the shapes of the molecules we actually observe. Mind-blowing, right?
Imagine you’ve got two pure p orbitals, looking like those classic ‘8’ shapes, ready to bond with something. Now, picture a central atom with a bunch of these pure orbitals. If it bonded using just those pure orbitals, the angles between the bonds would be… well, a bit predictable. You’d expect them to be at 90 degrees, given the dumbbell shape. But then you look at, say, methane (CH4), that stuff they use for natural gas. Carbon in the middle, four hydrogens sticking out. And those hydrogens aren’t at 90 degrees! They’re at a glorious 109.5 degrees, arranged like the points of a tetrahedron. How? That’s where our hybrid heroes come in.
This is where the idea of hybridization was born – this brilliant concept to explain why molecules have the shapes they do. It’s basically saying that before the atoms even get serious about bonding, they get together and do a little atomic makeover. They mix their original, pure atomic orbitals to create new, hybrid orbitals. Think of it like a chef taking different ingredients (pure orbitals) and blending them into a new, unique sauce (hybrid orbitals) that’s perfect for the dish (the molecule).
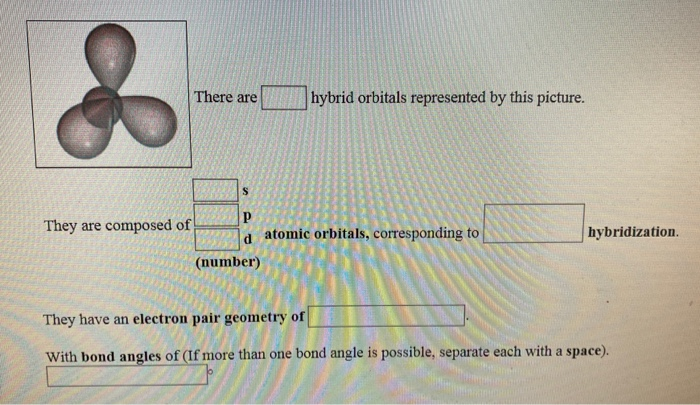
The Different Blends: sp, sp², and sp³
The picture you're looking at likely shows examples of these hybrid orbitals. The most common ones you’ll encounter are formed by mixing s and p orbitals. Let's break them down. You’ve got a few main recipes:
- sp³ Hybridization: This is like the "all-purpose" blend. One s orbital and three p orbitals get mushed together. What do you get? Four identical hybrid orbitals! And here's the cool part: these four sp³ orbitals are arranged in space to be as far away from each other as possible. What shape does that create? You guessed it – a tetrahedron. This is the hybridization you see in molecules like methane (CH4) and ethane (C2H6). It’s super common and responsible for those classic tetrahedral angles of 109.5 degrees. Pretty neat, huh?
- sp² Hybridization: Now, this is a bit of a different mix. Here, one s orbital gets combined with two p orbitals. This gives you three equivalent sp² hybrid orbitals. What about the leftover p orbital? It stays put, unmixed. These three sp² orbitals are arranged in a flat, triangular shape, known as a trigonal planar arrangement, with angles of 120 degrees. The leftover p orbital sits above and below this plane. This type of hybridization is crucial for molecules with double bonds, like ethene (C2H4). The sp² orbitals form the sigma (σ) bonds, the strong, direct connection, and the unmixed p orbitals overlap sideways to form the pi (π) bond, the weaker, but equally important, second connection in the double bond.
- sp Hybridization: This is the minimalist blend. One s orbital mixes with just one p orbital. This results in two equivalent sp hybrid orbitals. You’re left with two unmixed p orbitals. These two sp hybrid orbitals are arranged in a straight line, creating a linear geometry with bond angles of 180 degrees. The two leftover p orbitals are perpendicular to each other and to the sp hybrid orbitals. This is the hybridization you find in molecules with triple bonds, like ethyne (acetylene, C2H2), or carbon dioxide (CO2). Again, the sp orbitals form the sigma bonds, and the unmixed p orbitals form the pi bonds.
See the pattern? The number of hybrid orbitals you create is equal to the number of atomic orbitals you mixed. And these hybrid orbitals have specific shapes and orientations that dictate the geometry of the molecule. It’s like nature saying, "Okay, electrons, you can't just be in your own little spheres and dumbbells anymore. You need to cooperate and form these new, more efficient bonding shapes."
What the Picture is Really Showing You
So, when you look at that picture, what are you seeing? You’re probably seeing a visual representation of these hybrid orbitals. For example, if it shows a tetrahedral arrangement, you’re looking at sp³ hybrid orbitals. If it shows a trigonal planar arrangement, it’s likely sp² hybridization. And for a linear setup, it’s sp.
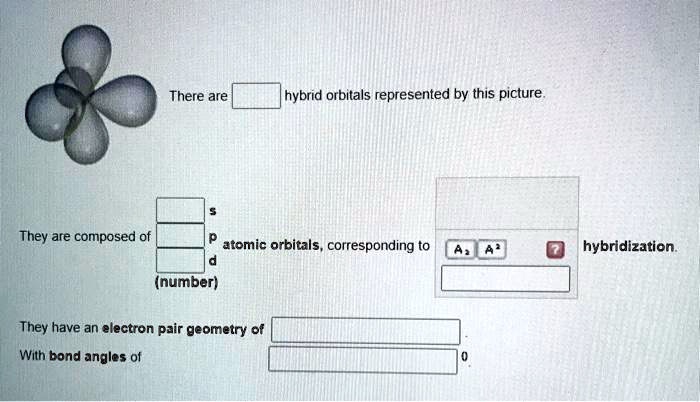
These drawings often depict the hybrid orbitals as lobes, and the size and shape of these lobes are important. They are generally larger than the original s and p orbitals, and they are often asymmetrical, with one large lobe and one smaller lobe. This larger lobe is the one that participates most effectively in bonding, meaning it can overlap better with an orbital from another atom to form a strong sigma bond. Think of it as a more focused, more effective way for electrons to get close enough to stick atoms together.
It’s also crucial to remember that these hybrid orbitals are not some separate, independent entities that exist before the molecule forms. Hybridization is a mathematical model, a way for us chemists to understand and predict molecular shapes and bonding. It's a concept that helps bridge the gap between the simple atomic orbitals we learn about first and the complex, often surprisingly symmetrical, structures of real molecules.
The picture might also illustrate how these hybrid orbitals overlap. You’ll see them shown extending towards each other, or sometimes, you’ll see the remnants of the unmixed p orbitals ready to form those pi bonds. It’s all about visualizing how the electron clouds of different atoms interact to create those stable molecular structures.
Why Does This Even Matter? (Besides Not Failing Chemistry)
Okay, so it’s a cool concept, but why should you care? Well, the shape of a molecule is everything. It determines its properties, how it interacts with other molecules, and ultimately, its function. This is especially true in biology and medicine.
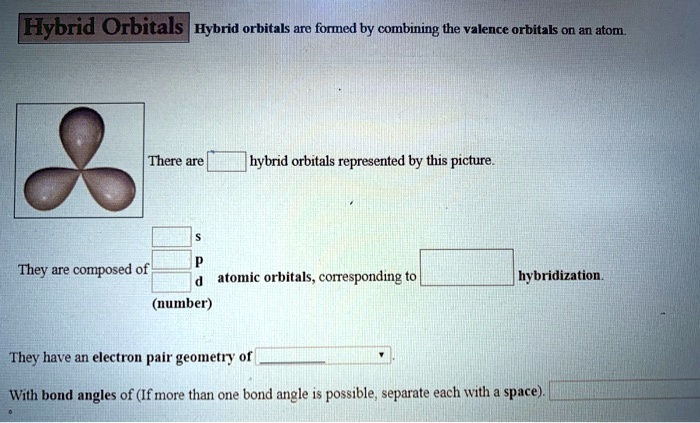
Think about enzymes, those biological catalysts that drive pretty much every reaction in your body. Their active sites, the places where they grab onto other molecules, have very specific shapes. If the enzyme is designed to bind to a certain molecule, that molecule has to fit perfectly into the active site like a key into a lock. And the shape of that molecule is dictated by the hybridization of its atoms and the resulting molecular geometry.
Even something as fundamental as DNA’s double helix structure is a result of specific bonding angles and molecular shapes, which are ultimately rooted in the hybridization of the atoms involved. It’s this intricate dance of electrons and orbital mixing that allows for life as we know it.
And then there’s pharmacology. Drugs work by interacting with specific biological targets, like receptors on cell surfaces. The effectiveness of a drug often depends on its precise three-dimensional shape. Chemists design drugs by understanding the hybridization and geometry of the target molecule and then creating a drug molecule that fits perfectly – or sometimes, not perfectly, to block a receptor. All of this relies on the principles of hybridization!
It's also why you might have different types of plastic or materials with vastly different properties. The way carbon atoms bond in polyethylene (a flexible plastic) versus diamond (incredibly hard) is all about the different ways their orbitals hybridize and arrange themselves. From the simplest gas to the most complex protein, hybrid orbitals are the silent orchestrators.
A Little Irony and a lot of Wonder
It's kind of funny, isn't it? We spend so much time learning about these tiny, invisible things, these abstract concepts like hybrid orbitals. And yet, they are responsible for the solidity of the chair you're sitting on, the taste of your food, the air you breathe. It's this beautiful paradox: the most fundamental aspects of our reality are often the most abstract and the hardest to visualize.
So, the next time you see a diagram of a molecule, or even just look at the world around you, remember the hybrid orbitals. They’re not just weird shapes on a page. They are the fundamental building blocks, the invisible forces that shape our universe. They are the reason atoms can get along, form stable bonds, and create the incredible diversity of matter that surrounds us. And that, my friends, is a pretty cool secret to have decoded.
Don't feel bad if it took a while to click. For a lot of us, it's a concept that requires a bit of wrestling. But once you get it, it's like unlocking a new level of understanding about how everything works. So, give yourself a pat on the back for exploring this fascinating corner of chemistry. You're doing great!
