The Valence Electron Configuration For Alkali Metals Is
Ever wondered what makes some elements super reactive, eager to jump into a chemical hug with other atoms? It all comes down to a tiny but mighty concept in the world of chemistry: valence electron configuration. And when we talk about the alkali metals, this concept gets particularly exciting and, dare I say, fun!
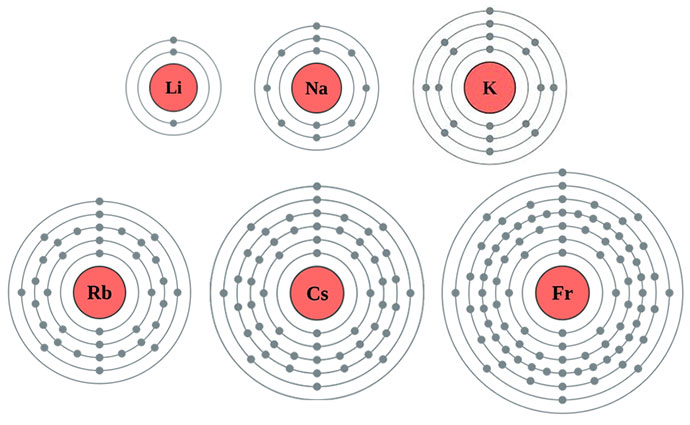
You might have seen them on the periodic table – a neat little row on the far left, looking all innocent. We're talking about elements like lithium (Li), sodium (Na), potassium (K), rubidium (Rb), cesium (Cs), and francium (Fr). These guys are the rockstars of the periodic table when it comes to reactivity. But why? The secret sauce, the real magic, lies in their valence electron configuration.
The Superpower of a Single Valence Electron
So, what exactly is a valence electron? Think of an atom like a tiny solar system. The nucleus is the sun, and electrons are the planets orbiting around it. The valence electrons are those outermost planets, the ones on the very edge of the solar system. These are the VIPs, the ones that get to interact with other atoms and form chemical bonds. They are the key players in all the chemical reactions you learn about.
Now, for our alkali metals, there's a universal truth about their outermost shell: it contains exactly one precious valence electron. Just one! This is their defining characteristic, their superpower, and the reason for their incredible eagerness to participate in chemistry. It’s like they have one extra party favor they just can't wait to share.
Why is having just one so significant? Atoms generally like to be stable, and stability often comes from having a full outermost electron shell. For most atoms, this means having eight electrons in their valence shell (this is often referred to as the octet rule). Our alkali metals are so close to achieving this stability. They only need to get rid of that single, lonely valence electron. And boy, do they want to get rid of it!

Imagine you're at a party, and you're holding one single balloon. Everyone else has a big, happy bunch of balloons. You'd probably be pretty eager to give away your single balloon to someone who wants it, just to feel like you're part of the group with full bunches, right? That's a bit like our alkali metals!
The Benefits of Being So Generous
This intense desire to shed that single valence electron has some pretty amazing benefits and consequences:
- Extreme Reactivity: Because they're so keen to lose an electron, alkali metals react very vigorously with other elements. They readily give up their single valence electron to become positively charged ions (called cations). This makes them fantastic reducing agents – they help other substances gain electrons and get reduced. This property is incredibly useful in many industrial and laboratory processes.
- Formation of Ionic Compounds: When an alkali metal atom loses its valence electron, it becomes a positive ion. This positive ion is then very attracted to atoms that want to gain electrons (like the nonmetals in Group 17, the halogens, which have seven valence electrons and desperately want one more). The strong electrostatic attraction between these oppositely charged ions forms a stable ionic compound. Think of sodium chloride (NaCl) – common table salt! It’s a classic example of an alkali metal (sodium) bonding with a halogen (chlorine).
- Softness and Low Melting Points: The way the atoms are arranged in solid alkali metals is relatively simple, and the metallic bonds holding them together aren't as strong as in many other metals. This is largely due to the limited number of valence electrons involved in the bonding. As a result, alkali metals are surprisingly soft and have low melting points. You can actually cut sodium with a knife, which is quite unusual for a metal!
- Conductivity: Like most metals, alkali metals are excellent conductors of electricity and heat. This is thanks to their delocalized valence electrons, which are free to move throughout the metal structure, carrying electrical charge and thermal energy.
Understanding the valence electron configuration of alkali metals isn't just about memorizing a fact; it's about unlocking the 'why' behind their fascinating behavior. It explains why they are so essential in everything from making medicines and batteries to producing clean water. They might seem simple with just one electron to offer, but that one electron is the key to a world of chemical wonders!
