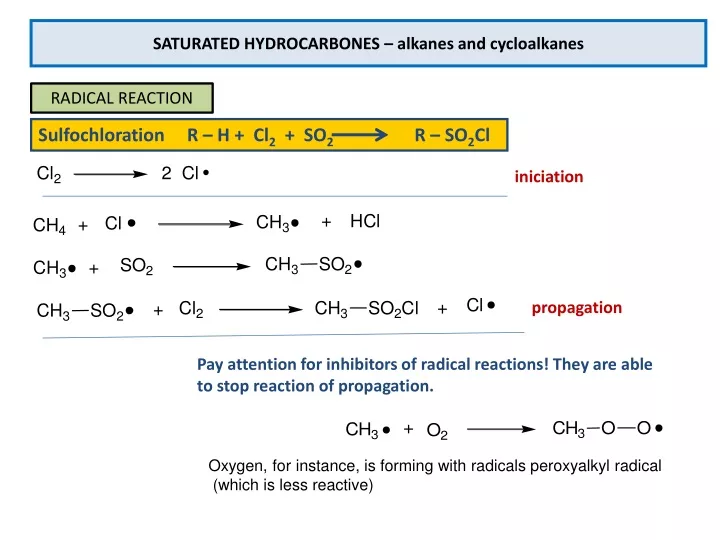
The Typical Reaction Of The Saturated Hydrocarbons Is
You know those things that make your car go vroom? Or the stuff that keeps your stove burning for a cozy dinner? Yep, we're talking about hydrocarbons, the backbone of so many things we use every single day. And today, we're going to dive into the wonderfully predictable, dare I say, chill personalities of the saturated hydrocarbons. Think of them as the laid-back cousins in the big hydrocarbon family. They're not looking for drama, they're just happy to exist and do their thing.
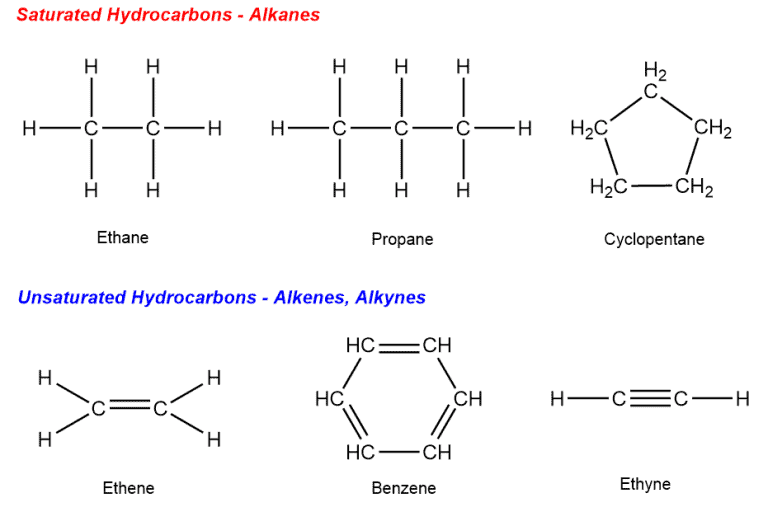
So, what’s the big deal with “saturated”? It’s like a sponge that’s soaked up as much water as it possibly can. For these hydrocarbons, it means they’ve got all the hydrogen atoms they can possibly hold onto. No empty spots, no room for anything else. And because of this happy-fullness, their typical reaction to life’s little challenges is… well, pretty much nothing! They’re incredibly stable. Imagine a comfy old armchair that just sits there, unbothered by the world. That’s our saturated hydrocarbon.
This means they’re not usually the ones jumping into exciting chemical reactions. You won’t find them doing backflips with other molecules. They’re more likely to be found lounging around, perfectly content. This is why things like methane (the main ingredient in natural gas, think warm fuzzy feelings from your furnace) and propane (the BBQ hero!) are so reliable. They don't spontaneously combust when you look at them funny. They’re dependable. They show up. They do their job. It’s almost heartwarming, isn’t it? A little molecule that’s just so… chill.
But don't mistake their chill for weakness! While they might not be the life of the chemical party, their stability is their superpower. It's why we can trust gasoline to power our adventures without worrying it'll suddenly decide to become something else entirely. It’s why plastics, many of which are built from these sturdy hydrocarbon structures, can last for ages, holding our groceries and protecting our electronics. They are the quiet guardians of our modern lives, the unsung heroes of convenience.
Now, they can be coaxed into doing something, but it usually takes a bit of persuasion. Think of it like trying to get your most introverted friend to dance at a wedding. It's not going to happen just because the music is good. You need a really special occasion, or maybe a little nudge. For saturated hydrocarbons, that nudge often involves a good amount of heat or a bit of a spark. They're not going to participate in a polite discussion with an oxidizer; they need a full-on, fiery embrace to really get going. This is how we get combustion, the spectacular burning that gives us energy. It’s their way of letting loose, a controlled explosion of happiness that fuels our world.

And when they do react under these extreme conditions, it's usually a pretty straightforward exchange. They’ll happily share their hydrogen atoms (or even carbon atoms!) with oxygen, producing water and carbon dioxide. It’s a bit like a generous soul finally deciding to share their snacks, but on a molecular level. They give up their stability, but for a grander purpose: to create energy and make things happen. It's a testament to their underlying goodness, that even in their most energetic moments, they’re still focused on providing something useful.
What’s truly fascinating is how this lack of reactivity is actually what makes them so incredibly useful. Because they don't easily break down or change, they can form the long, strong chains that make up things like polyethylene (the stuff your plastic bags are made of) and polypropylene (used in everything from car parts to carpets). These long chains are like sturdy ropes, holding everything together. If they were constantly reacting with everything around them, our plastic cups would disintegrate before we even finished our coffee!
So, next time you fill up your car, or light your gas stove, take a moment to appreciate the humble, steadfast nature of saturated hydrocarbons. They’re the reliable friends of the chemical world. They might not be flashy, but they are the bedrock upon which so much of our comfortable, modern life is built. They are the quiet, dependable backbone, always ready to provide energy when needed, and otherwise content to just be. It’s a beautiful thing, really. A whole class of molecules that are just… happy being themselves, and in doing so, make our lives a whole lot easier and warmer.
They're the quiet guardians of our modern lives, the unsung heroes of convenience.
It’s a reminder that sometimes, the greatest strength lies not in being the most reactive, but in being the most resilient. These molecules are proof that being consistently there, and consistently themselves, is a powerful force. They’re the original definition of ‘solid’ – in every sense of the word!
